Author
Dan Conley
Correspondent
Key Points
- Strain elastography vs. ShearWave™ elastography
- Non-invasive alternative to biopsy
- Measure tissue stiffness in kilopascals
- Appropriate for detection, characterisation, biopsy guidance, therapeutic monitoring
Introduction
Because of the correlation between stiffness of tissue and pathological status, medical imaging modalities that assess stiffness can be instrumental in staging diseases of the liver, breast, prostate, and kidney. One such modality, ultrasound elastography, has traditionally relied on soft compression of the target tissue to achieve this evaluation, previously performed by clinical palpation. This traditional approach, called ‘strain’ elastography, relies on applied deformation of the target tissue and an estimate of the differential displacement before and after that deformation; this approach is limited, however, by the location of the target tissue and the experience of its users. ShearWave™ Elastography (SWE™), introduced in the early 2000s, is a faster, automated alternative to strain elastography, and has been shown to aid in the characterisation of focal and diffuse diseases, as well as in the guidance of tissue biopsies and in the monitoring of therapeutic applications.
How does ShearWave Elastography Work?
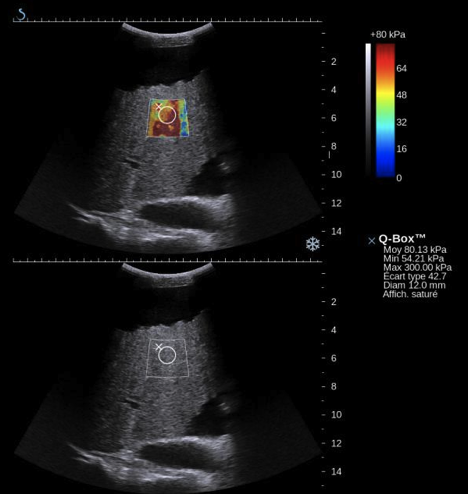
SWE measures the velocity of shear waves (or transverse waves) moving through tissue. Because shear waves travel from 1 to 10 metres per second, extremely high-speed acquisition is necessary for capturing and interpreting them. The French company SuperSonic Imagine has pioneered the UltraFast™ ultrasound technology that acquires images 200 times faster than conventional ultrasound systems, thus being able to capitalize on the high-speed shear waves. These data are displayed in a two-dimensional map, which overlays the colour-coded tissue elasticity information, measured in kilopascals, onto the B-mode image for anatomical correlation. The two-dimensional SWE image shows the extent and variations of tissue stiffness—yielding crucial information for disease management and patient care.
The clinical efficacy of SWE was established through a two year multicentre breast clinical study conducted in 17 American and European sites. The goal of this study, which enrolled 1800 cases, was to determine whether SWE could improve the accuracy of sonographic characterisation of breast masses, and therefore better orient patients towards short-term follow-up or biopsy. This data was analysed to identify how each SWE criteria, when added to greyscale criteria, could improve lesion characterisation (in sensitivity and specificity when compared to greyscale alone) and eventually improve the BI-RADS® score. The first main result of this clinical study was that SWE features are reproducible. Reproducibility assures the physician of a reliable evaluation of the lesion during an exam and for follow-up exams. The second result was that SWE features increased diagnostic accuracy and improved lesion characterisation, thereby each individual feature when added to the grayscale criteria improved the BI-RADS® classification of lesions.
Several studies on SWE have also focused on the liver, where SWE features several advantages over more conventional imaging modalities and other types of non-invasive liver stiffness assessments. As liver biopsies are associated with a non-negligible morbidity rate, and can lead to complications including infection, damage to viscera, and haemorrhaging, techniques for a non-invasive liver stiffness assessment are currently highly investigated.
SWE is a non-invasive and non-ionising modality that can be used to assess liver fibrosis. This has 4 major advantages that were recently demonstrated by publications on patients with viral Hepatitis:
- First, SWE is diagnostically sensitive, so it can be used to optimize the indications for biopsy, and to overcome several of its sampling limitations. The ability to survey and quantify an entire region of interest in real time minimizes the need for second or third biopsies, and reduces follow-ups due to variability in sampling.
- Two, SWE can be used iteratively to follow stiffness changes over time. These qualities make it an attractive option for monitoring evolving liver injuries, including cases of biliary atresia.
- Third, the excellent sensitivity and specificity of SWE can help identify cases of low-grade fibrosis, when the disease is in its earliest stages. (Early identification of liver fibrosis in children, for instance, is critical for the swift, appropriate treatment of the disease.)
- Fourth, on liver nodules, the qualities of SWE can help differentiate 2 benign types of nodules, which deserve different treatment: adenomas from atypical fibrous nodular hyperplasia. It can also distinguish hepatocellular carcinomas from cholangiocarcinomas, and more accurately identify hepatocellular carcinomas in cirrhotic livers.
From a broad efficiency perspective, the first and second of these advantages are the most compelling; eliminating biopsy-related complications not only reduces unnecessary suffering, it reduces the additional and financial costs (hospitalisation; additional interventions) associated with them.
Reducing the need for liver biopsies is one of the signal benefits of SWE. But the technology can assist in other clinical areas to similar effect. Prostate cancer, for instance, is the most common cancer in males worldwide—yet with early detection, it is also one of the most treatable. SWE has been shown to improve identification and visualisation of prostate cancer, which in turn may help target necessary biopsies. SWE is easy to use and renders quantifiable, high quality stiffness imaging with reproducible results.
Additionally, as SWE imaging is real time and extremely accurate, it can also be useful in pediatric imaging applications, wherein patients are often difficult to keep still long enough to image.. (Non-invasiveness is of course also paramount for this population.)
SWE has proven useful in many applications, including, thyroid, breast, musculoskeletal and gynaecological imaging.
Caption: Example of a patient suffering from hepatitis B infection, which had led to chronic liver fibrosis. As highlighted by the extremely high stiffness values (stiffness of normal liver parenchyma usually ranges between 4 to 6 kPa), this patient had reached to stage of hepatic cirrhosis, with ascites collection as seen on top of the greyscale image.