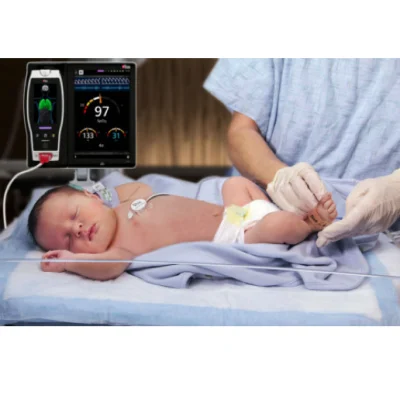
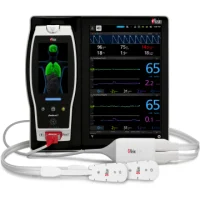
Masimo announced today FDA clearance of RAS-45, an acoustic respiration sensor for rainbow Acoustic Monitoring® (RAM®), for infant and neonatal patients. RAM could previously be used to monitor adult and pediatric patients greater than 10 kg using RAS-125c and RAS-45 sensors. With clearance of the RAS-45 sensor for infant and neonatal patients, acoustic respiration rate measurement is now, for the first time, possible for patients of all sizes, including neonates, in the United States.
RAM noninvasively and continuously measures respiration rate using an innovative adhesive sensor with an integrated acoustic transducer, the RAS-45 and RAS-125c, applied to the patient’s neck area or, for infant and neonatal patients under 10 kg, the chest. Using acoustic signal processing that leverages Masimo Signal Extraction Technology® (SET®), the respiratory signal is separated and processed to display continuous respiration rate (RRa®) and an acoustic respiration waveform, a visualization of the vibrations caused by the patient’s airflow. The acoustic sensor also allows clinicians to listen to the sound of a patient’s breathing, whether at the bedside, through a point-of-care device like the Radical-7® Pulse CO-Oximeter®, or remotely, from a Patient SafetyNet™ view station.
The RAS-45 sensor for infant and neonatal patients offers multiple benefits of particular importance for successfully monitoring these youngest and most fragile patients. With the clearance for newborns and neonates, RRa’s accuracy range has been expanded up to 120 breaths per minute, while still providing accuracy of +/- 1 breath per minute, facilitating accurate measurement of the higher respiratory rates common in this population. The sensor itself is significantly smaller than the RAS-125c sensor, and in fact with a diameter of approximately 2.2 cm without adhesive is only slightly larger than a nickel. Similarly, it weighs so little, 13 grams, that its presence may be barely noticeable, and features an adhesive that is transparent, light, and flexible. The size, weight, and adhesive advantages make it particularly suitable for the smaller stature and delicate skin of infants and neonates.
RRa has been shown not only to be accurate(1,2) and reliable(1), but also easy-to-use(1), easy-to-tolerate(1,3), and to enhance patient compliance with respiration monitoring. In a study comparing pediatric patient tolerance of sidestream capnography with a nasal cannula to respiration rate monitoring with an RAS-125c acoustic sensor, 15 out of 40 patients removed the capnography cannula, while only one removed the RAM acoustic sensor.(3) In a study of 98 patients consciously sedated during upper gastrointestinal endoscopy, researchers found that RRa monitoring with the RAS-125c sensor more accurately assessed respiration rate than impedance pneumography.(2)
Joe Kiani, Founder and CEO of Masimo, commented, “From the beginning, we have focused our R&D on neonates and children for many reasons, including our belief that helping clinicians care for children will provide more benefit to society. RAM harnesses the power of our breakthrough signal processing and sensor technology and applies it to a measurement that has either been unreliable or difficult to use, respiration measurement, the third vital sign.”
RAM is available on most rainbow SET™-ready platforms. Continuous monitoring of respiration rate can be helpful in cases such as sedation-based procedures and post-surgical patients receiving patient-controlled analgesia for pain management.(4,5)
References
- Macknet MR et al. Accuracy and Tolerance of a Novel Bioacoustic Respiratory Sensor in Pediatric Patients. Anesthesiology. 2007;107:A84 (abstract).
- Goudra BG et al. Comparison of Acoustic Respiration Rate, Impedance Pneumography and Capnometry Monitors for Respiration Rate Accuracy and Apnea Detection during GI Endoscopy Anesthesia. Open J Anesthesiol. 2013;3:74-79.
- Patino M et al. Accuracy of Acoustic Respiration Rate Monitoring in Pediatric Patients. Paediatr Anaesth. 2013 Sep 3.
- Stoelting, RK et al. APSF newsletter. 2011. www.apsf.org.
- The Joint Commission Sentinel Event Alert. Issue 49, August 8, 2012, www.jointcomission.org.
Latest Articles
Paediatrics, Neonatal, Masimo, non-invasive, Acoustic Respiration Sensor, RAS-45, rainbow Acoustic Monitoring Sensor RAS-45, FDA clearance, Paediatircs, Infant, Acosustic respiration rate, acoustic respiration rate, Respiration sensor
Masimo announces FDA clearance of the rainbow acoustic monitoring® RAS-45 sensor