Investigators working to stem the spread of antibiotic-resistant bacteria have taken a major step in their efforts to develop new treatments.
Researchers at The University of Texas Health Science Center at Houston (UTHealth) report they have identified a novel mechanism that a particular superbug uses to fend off a key front-line antibiotic called daptomycin. The superbug often affects critically ill patients.
This information is helping investigators identify compounds to knock out a mechanism of resistance in order to “disarm” the superbug.
Prior to the mass production of antibiotics, a cut or strep throat could lead to serious illness or even death. Antibiotics gave doctors the ability to treat bacterial infections. But, bacteria have developed mechanisms of resistance that can make antibiotics ineffective against the most aggressive superbugs.
Thousands of people succumb to superbug-related infections worldwide annually and superbugs account for $20 billion in excess health care costs in the United States each year.
“Antibiotic resistance is one of the major public health threats of the 21st Century,” said Cesar Arias, M.D., Ph.D., the study’s senior author and associate professor in the Division of Infectious Diseases at the UTHealth Medical School. “These superbugs can make antibiotics useless, which makes certain bacterial infections virtually untreatable.”
While there are several types of antibiotic-resistant bacteria, the study focused on a hard-to-treat superbug called vancomycin-resistant enterococci or VRE. VRE usually affect patients who have a compromised immune system or who are critically ill.
The frequency of VRE recovered from hospitalized patients in the United States has increased eightfold in the last 15 years, becoming the second most common hospital-associated bacterium in the United States, Arias said.
The superbug appears to be building resistance to one of the few antibiotics that works against it – daptomycin.
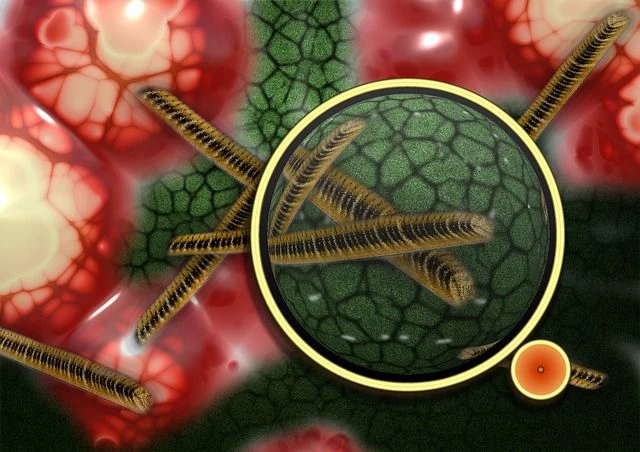
To see how VRE developed the ability to ward off daptomycin during the course of treatment, Arias’ team used fluorescent labeled daptomycin and observed the interaction between the superbug and the antibiotic with the aid of advanced microscopy techniques.
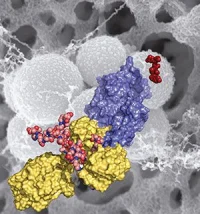
Contrary to the prevailing belief that tiny electrical charges on the surface of VRE cells repel the antibiotic, the researchers report that the VRE cells actually divert the antibiotic and “trap” it to an area where it is rendered ineffective. The mechanism of resistance is completed by changing the composition of the bacterial cell membrane. The study also provides the genetic and biochemical basis for the resistance pathway.
“The importance of this work is that an understanding of ‘how’ bacteria become resistant can then lead to a search for new antibiotics that target the resistance pathway itself, thus overcoming and preventing resistance,” said Barbara E. Murray, M.D., study co-author as well as director of the Division of Infectious Diseases and holder of the J. Ralph Meadows Professorship in Internal Medicine at the UTHealth Medical School.
“This important study is about resistance to the ‘last resort’ agent, daptomycin, one of the few antibiotics we have to treat multi-drug resistant enterococci, and it builds on Dr. Arias' previous study published in The New England Journal of Medicine,” added Murray, who is president-elect of the Infectious Diseases Society of America.
Arias’ co-authors from the UTHealth Medical School include Truc T. Tran, Pharm. D. (first author); Diana Panesso, Ph.D.; Lorena Diaz; Eugenia Mileykovskaya, Ph.D.; Jose Munita, M.D.; Jinnethe Reyes; and William Dowhan, Ph.D., holder of the John S. Dunn, Sr., Chair in Biochemistry and Molecular Biology.
Other co-authors include Nagendra N. Mishra and Arnold S. Bayer, M.D., of UCLA; Ziqianq Guan, Ph.D., of Duke University School of Medicine; Yousif Shamoo, Ph.D., of Rice University; and George M. Weinstock, Ph.D., of Washington University in St. Louis.
The study titled “Daptomycin-Resistant Enterococcus faecalis Diverts the Antibiotic Molecule from the Division Septum and Remodels Cell Membrane Phospholipids” received support from the National Institute of Allergy and Infectious Diseases (R01 AI093749), part of the National Institutes of Health.
Arias leads the UTHealth Medical School Laboratory for Antimicrobial Research and the Universidad El Bosque, Molecular Genetics and Antimicrobial Resistance Unit in Colombia. Arias’ laboratories have collaborations with several Latin American countries to study specific types of antibiotic-resistant bacteria.
Source: The University of Texas Health Science Center at Houston
Researchers at The University of Texas Health Science Center at Houston (UTHealth) report they have identified a novel mechanism that a particular superbug uses to fend off a key front-line antibiotic called daptomycin. The superbug often affects critically ill patients.
This information is helping investigators identify compounds to knock out a mechanism of resistance in order to “disarm” the superbug.
Prior to the mass production of antibiotics, a cut or strep throat could lead to serious illness or even death. Antibiotics gave doctors the ability to treat bacterial infections. But, bacteria have developed mechanisms of resistance that can make antibiotics ineffective against the most aggressive superbugs.
Thousands of people succumb to superbug-related infections worldwide annually and superbugs account for $20 billion in excess health care costs in the United States each year.
“Antibiotic resistance is one of the major public health threats of the 21st Century,” said Cesar Arias, M.D., Ph.D., the study’s senior author and associate professor in the Division of Infectious Diseases at the UTHealth Medical School. “These superbugs can make antibiotics useless, which makes certain bacterial infections virtually untreatable.”
While there are several types of antibiotic-resistant bacteria, the study focused on a hard-to-treat superbug called vancomycin-resistant enterococci or VRE. VRE usually affect patients who have a compromised immune system or who are critically ill.
The frequency of VRE recovered from hospitalized patients in the United States has increased eightfold in the last 15 years, becoming the second most common hospital-associated bacterium in the United States, Arias said.
The superbug appears to be building resistance to one of the few antibiotics that works against it – daptomycin.
To see how VRE developed the ability to ward off daptomycin during the course of treatment, Arias’ team used fluorescent labeled daptomycin and observed the interaction between the superbug and the antibiotic with the aid of advanced microscopy techniques.
Contrary to the prevailing belief that tiny electrical charges on the surface of VRE cells repel the antibiotic, the researchers report that the VRE cells actually divert the antibiotic and “trap” it to an area where it is rendered ineffective. The mechanism of resistance is completed by changing the composition of the bacterial cell membrane. The study also provides the genetic and biochemical basis for the resistance pathway.
“The importance of this work is that an understanding of ‘how’ bacteria become resistant can then lead to a search for new antibiotics that target the resistance pathway itself, thus overcoming and preventing resistance,” said Barbara E. Murray, M.D., study co-author as well as director of the Division of Infectious Diseases and holder of the J. Ralph Meadows Professorship in Internal Medicine at the UTHealth Medical School.
“This important study is about resistance to the ‘last resort’ agent, daptomycin, one of the few antibiotics we have to treat multi-drug resistant enterococci, and it builds on Dr. Arias' previous study published in The New England Journal of Medicine,” added Murray, who is president-elect of the Infectious Diseases Society of America.
Arias’ co-authors from the UTHealth Medical School include Truc T. Tran, Pharm. D. (first author); Diana Panesso, Ph.D.; Lorena Diaz; Eugenia Mileykovskaya, Ph.D.; Jose Munita, M.D.; Jinnethe Reyes; and William Dowhan, Ph.D., holder of the John S. Dunn, Sr., Chair in Biochemistry and Molecular Biology.
Other co-authors include Nagendra N. Mishra and Arnold S. Bayer, M.D., of UCLA; Ziqianq Guan, Ph.D., of Duke University School of Medicine; Yousif Shamoo, Ph.D., of Rice University; and George M. Weinstock, Ph.D., of Washington University in St. Louis.
The study titled “Daptomycin-Resistant Enterococcus faecalis Diverts the Antibiotic Molecule from the Division Septum and Remodels Cell Membrane Phospholipids” received support from the National Institute of Allergy and Infectious Diseases (R01 AI093749), part of the National Institutes of Health.
Arias leads the UTHealth Medical School Laboratory for Antimicrobial Research and the Universidad El Bosque, Molecular Genetics and Antimicrobial Resistance Unit in Colombia. Arias’ laboratories have collaborations with several Latin American countries to study specific types of antibiotic-resistant bacteria.
Source: The University of Texas Health Science Center at Houston
Latest Articles
antibiotic resistance
Investigators working to stem the spread of antibiotic-resistant bacteria have taken a major step in their efforts to develop new treatments. Researchers...