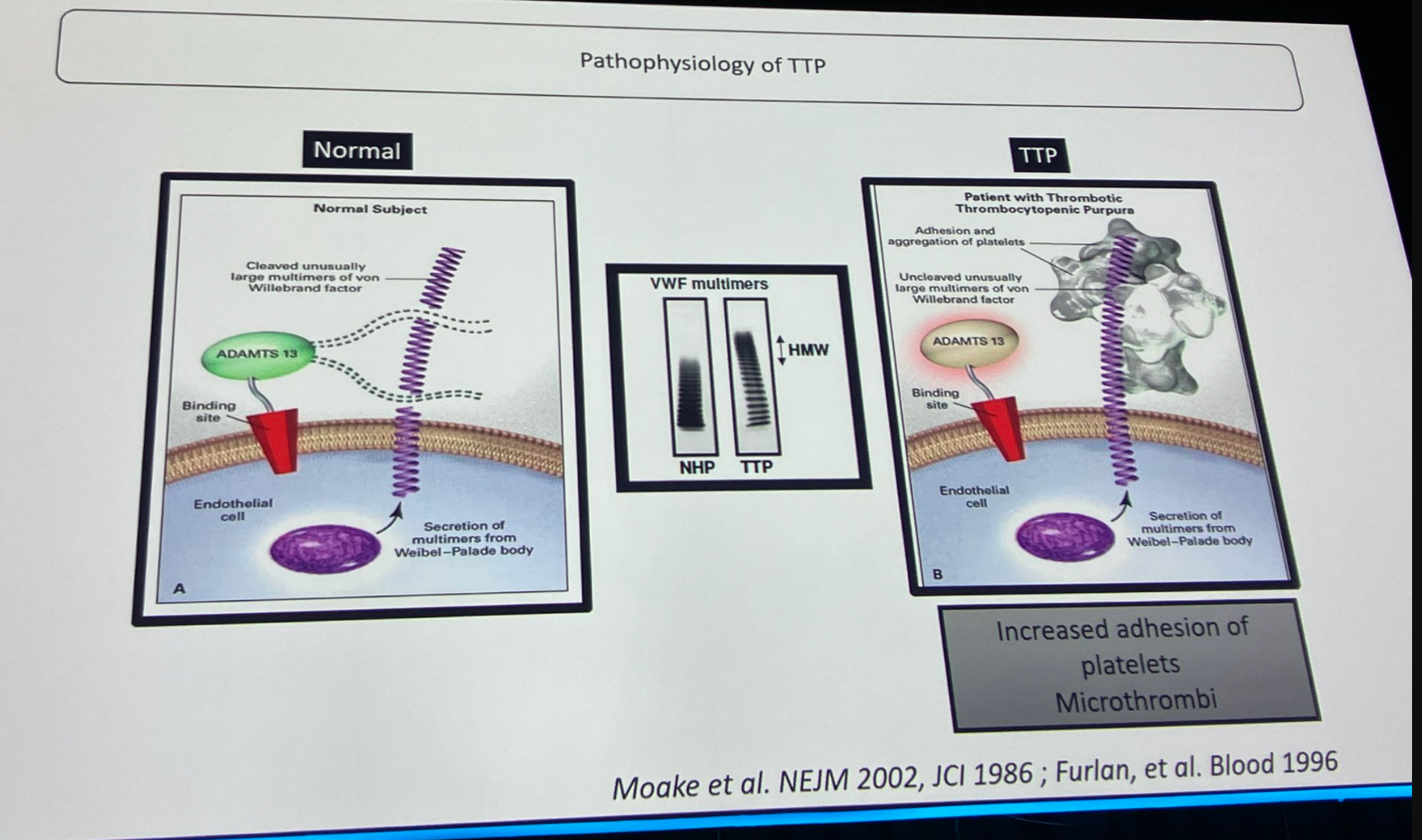
Previous research has indicated the potential benefits of using magnesium sulphate in thrombotic thrombocytopaenic purpura (TTP) treatment. The goal of the Magnesium sulphate in patients with thrombotic thrombocytopenic purpura (MAGMAT) study was to assess the impact of magnesium sulphate supplementation on the recovery of patients with TTP.
This was a multicentre study involving adult patients diagnosed with TTP. Participants were randomly assigned to receive either intravenous magnesium sulphate (initial dose of 6 g followed by a continuous 6 g/24 h infusion for 3 days) or a placebo alongside standard TTP treatment. The primary outcome of the study was the median time required for platelet levels to return to normal (platelet count ≥ 150 G/L). The study assessed both the effectiveness and safety of magnesium sulphate using an intention-to-treat analysis.
The study included 73 participants. Among these, 35 received magnesium sulfate, and 38 received placebo. In both groups, the median time for platelet levels to return to normal was four days. The study found that the likelihood of response (platelet normalisation) was similar between the two groups, with no significant difference. The incidence of serious adverse reactions was comparable in both groups. By day 90, four patients in the magnesium sulphate group and two in the placebo group had died. The most common side effect was low blood pressure, occurring in 34% of the magnesium sulphate group and 29% of the placebo group, with no significant difference in occurrence between the two groups.
Overall, these findings show that in patients with TTP, adding magnesium sulphate to the standard treatment did not lead to a significant improvement in time to platelet normalisation.
Source: Intensive Care Medicine; #LIVES2023
Slide credit: Presentation @LIVES2023
Image Credit: ESICM







