Researchers from the Mayo Clinic (Jacksonville, FA, USA) have found that the use of two new oral medications is effective in fighting hepatitis C infection amongst liver transplant patients. Given the clinical importance of the study, results have been presented in a plenary session of the American Association for the Study of Liver Diseases (The Liver Meeting® 2014) being held between 7 and 11 November in Boston, Massachusetts.
"This is the first study to examine the use of these two new drugs — simeprevir and sofosbuvir — in liver transplant recipients, and, based on this large study, we find it to be a better option than current treatment," said the study's lead author, Surakit Pungpapong, MD, a transplant hepatologist and an associate professor of medicine at Mayo Clinic in Florida.
Current standard pre- and post-transplant treatment requires the use of interferon injections, along with ribavirin. As interferon engages the human immune system to fight the hepatitis C virus, this immune response can also lead to organ rejection, Dr. Pungpapong explained. Interferon can cause a variety of side effects including insomnia, anaemia, depression, flu symptoms, irritability, and hair loss, amongst others, the doctor said.
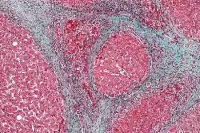
Chronic hepatitis C virus is the most common chronic bloodborne infection in the United States, affecting more than three million individuals. In most cases, infected people show no symptoms of the disease until liver damage from chronic inflammation occurs decades later — which happens to 5-30 percent of infected individuals. Data show that hepatitis C infection accounts for two-thirds of newly diagnosed chronic liver disease cases and 40 percent of liver transplants.
When patients with hepatitis C receive a liver transplant, their new livers will eventually become infected. These transplanted organs then require anti-viral treatment before they become severely damaged. However, traditional post-transplant hepatitis C therapy can take up to a year, is potentially toxic and can lead to organ rejection, the researchers said.
For this study, Mayo Clinic researchers enrolled post-transplant patients at their three US sites, including Rochester (MN), Scottsdale (AZ), and the Mayo Clinic Transplant Center in Jacksonville (FL) which has one of the five most active liver transplants programmes nationwide. The research team will report on outcomes of more than 100 patients, but the study is still ongoing.
These patients were too sick to be treated for hepatitis C before their transplants, according to Dr. Pungpapong. "By the time liver cirrhosis occurs, it could be too late to use anti-viral drugs."
Simeprevir and sofosbuvir were approved for pretransplant use last year by the Food and Drug Administration (FDA), but not as a combined therapy. The FDA also required that the two drugs be combined with interferon and ribavirin.
However, in a large clinical trial, researchers tested simeprevir and sofosbuvir without interferon — an off-label use — in pre-transplantation patients and found the combination to be a brief and effective therapy. In the new study, the Mayo team extended the idea of using these drugs together without interferon in post-transplantation patients.
The Mayo team observed that eradication of the virus in the patients was excellent — more effective than the use of interferon and ribavirin — and with far fewer side effects. "We believe use of these drugs, both pre- and post-transplant, represents a substantial clinical advance," Dr. Pungpapong noted.
Source: EurekAlert.org
Image Credit: hepcomm.org
"This is the first study to examine the use of these two new drugs — simeprevir and sofosbuvir — in liver transplant recipients, and, based on this large study, we find it to be a better option than current treatment," said the study's lead author, Surakit Pungpapong, MD, a transplant hepatologist and an associate professor of medicine at Mayo Clinic in Florida.
Current standard pre- and post-transplant treatment requires the use of interferon injections, along with ribavirin. As interferon engages the human immune system to fight the hepatitis C virus, this immune response can also lead to organ rejection, Dr. Pungpapong explained. Interferon can cause a variety of side effects including insomnia, anaemia, depression, flu symptoms, irritability, and hair loss, amongst others, the doctor said.
Chronic hepatitis C virus is the most common chronic bloodborne infection in the United States, affecting more than three million individuals. In most cases, infected people show no symptoms of the disease until liver damage from chronic inflammation occurs decades later — which happens to 5-30 percent of infected individuals. Data show that hepatitis C infection accounts for two-thirds of newly diagnosed chronic liver disease cases and 40 percent of liver transplants.
When patients with hepatitis C receive a liver transplant, their new livers will eventually become infected. These transplanted organs then require anti-viral treatment before they become severely damaged. However, traditional post-transplant hepatitis C therapy can take up to a year, is potentially toxic and can lead to organ rejection, the researchers said.
For this study, Mayo Clinic researchers enrolled post-transplant patients at their three US sites, including Rochester (MN), Scottsdale (AZ), and the Mayo Clinic Transplant Center in Jacksonville (FL) which has one of the five most active liver transplants programmes nationwide. The research team will report on outcomes of more than 100 patients, but the study is still ongoing.
These patients were too sick to be treated for hepatitis C before their transplants, according to Dr. Pungpapong. "By the time liver cirrhosis occurs, it could be too late to use anti-viral drugs."
Simeprevir and sofosbuvir were approved for pretransplant use last year by the Food and Drug Administration (FDA), but not as a combined therapy. The FDA also required that the two drugs be combined with interferon and ribavirin.
However, in a large clinical trial, researchers tested simeprevir and sofosbuvir without interferon — an off-label use — in pre-transplantation patients and found the combination to be a brief and effective therapy. In the new study, the Mayo team extended the idea of using these drugs together without interferon in post-transplantation patients.
The Mayo team observed that eradication of the virus in the patients was excellent — more effective than the use of interferon and ribavirin — and with far fewer side effects. "We believe use of these drugs, both pre- and post-transplant, represents a substantial clinical advance," Dr. Pungpapong noted.
Source: EurekAlert.org
Image Credit: hepcomm.org
Latest Articles
cirrhosis, Mayo Clinic, liver transplant, Hepatitis C
Researchers from the Mayo Clinic (Jacksonville, FA, USA) have found that the use of two new oral medications is effective in fighting hepatitis C infection...