FDA Approves Octaplas® Expanding Octapharma U.S. Transfusion Medicine
- ICU
- 30/01/2013
Octapharma USA announced on January 22, that the U.S. Food and Drug Administration (FDA) approved Octaplas®, its solvent/detergent treated pooled...
READ MORE
Octapharma USA announced on January 22, that the U.S. Food and Drug Administration (FDA) approved Octaplas®, its solvent/detergent treated pooled...
READ MORE
At least one in five people in countries for which data are available were infected with influenza during the first year of the 2009 H1N1 pandemic,...
READ MORE
Central line associated bloodstream infections (CLABSIs) in newborns were reduced by 58 percent in less than a year in hospital neonatal intensive...
READ MORE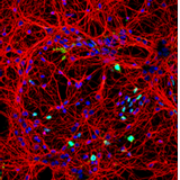
Despite recent advances in understanding the mechanisms of nerve injury, tissue-engineering solutions for repairing damage in the central nervous...
READ MORE
Gambro announced on January 24, that the U.S. Food and Drug Administration (FDA) expanded the cleared indications for its Molecular Adsorbent Recircula
READ MORE
RP-VITA Auto Drive and iPad® App extend remote care with unprecedented ease of use InTouch Health, the leader in acute care telemedicine, an
READ MORE
In recent weeks the intensive critical care units at University Health Network’s Toronto General Hospital have used Extra Corporeal Lung Support...
READ MORE
Thermo Scientific QMS Everolimus is CE Marked for use in the European Union with Liver Transplant Drug Monitoring Thermo Fisher Scientific...
READ MORE
A revised and updated edition of "Surviving Sepsis Campaign: International Guidelines for Management of Severe Sepsis and Septic Shock: 2012" were...
READ MORE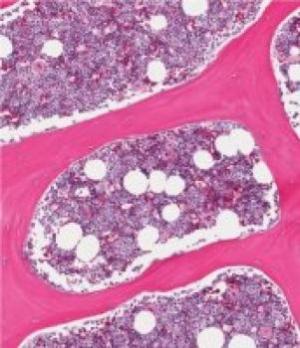
Instead of focusing on the elimination of cancer cells, maintaining a stable population of healthy blood cells in the bone marrow could be the most...
READ MORE