CorMedix Receives CE Mark for Neutrolin® Catheter Lock Solution
- ICU
- 09/07/2013
CorMedix Inc. announced on July 5, 2013, that it received CE Mark approval for Neutrolin®, a catheter lock solution, for patients with central venous...
READ MORE
CorMedix Inc. announced on July 5, 2013, that it received CE Mark approval for Neutrolin®, a catheter lock solution, for patients with central venous...
READ MORE
New research from Western University unravels a novel means of communication that allows bacteria such as Burkholderia cenocepacia (B. cenocepacia)...
READ MORE
The influenza virus’ ability to mutate quickly has produced new, emerging strains that make drug discovery more critical than ever. For the first...
READ MORE
ZOLL Medical Corporation, a manufacturer of medical devices and related software solutions, announced today that it has purchased the assets of CoAxia™,
READ MORE
ImaCor Inc., the developer of the world's only hemodynamic transesophageal echocardiography (hTEE™) management device, today announced the implement
READ MORE

A remote 'eye in the sky' will help keep round-the-clock tabs on critically ill patients at Guy's and St Thomas' Hospitals thanks to an innovative...
READ MORE
When the world’s top critical and intensive care practitioners come to Durban at the end of August they will be telling a story of care that goes...
READ MORE
A single systemic dose of special immune cells prevented rejection for almost four months in a preclinical animal model of kidney transplantation,...
READ MORE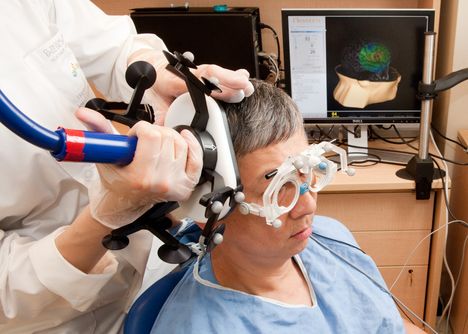
Transcranial magnetic stimulation (TMS) The TMS device is a handheld magnetic coil that delivers low intensity stimulation and elicits muscle...
READ MORE