ICU Management & Practice, Volume 21 - Issue 3, 2021
Introduction
In the intensive care unit (ICU), supplemental oxygen administration is a routine treatment administrated to a large number of patients. The rationale for this oxygen administration is the prevention and the treatment of hypoxia, which has deleterious effects and ultimately leads to cell death (O’Driscoll et al. 2017). As a reminder, hypoxia is a condition in which there is not enough oxygen available to the blood and body tissues while hypoxaemia is an abnormally low concentration of oxygen in the blood. The formula to calculate the arterial content of oxygen in blood is:
(arterial oxygen saturation.haemoglobin.1.34) + (arterial oxygen tension.0.003)
suggesting that oxygen saturation is the main determinant of oxygen content in blood. For this reason, excessive values of arterial oxygen tension (PaO2) are often tolerated and perceived by physicians as a safety net against hypoxaemia and its consequences (Suzuki et al. 2013; Young et al. 2015).
Increasing evidence suggests that exposure to hyperoxia during critical illness is associated with impaired outcome and conservative oxygen therapy should be a future recommendation in most guidelines (Barbateskovic et al. 2019; Chu et al. 2018). Nevertheless, inconsistent findings are reported with no deleterious effect or even a protective effect of hyperoxia in ICU patients (Duclos et al. 2021; Mackle et al. 2020). We briefly reviewed the current literature on the topic to discuss the benefits and harms of supplemental oxygen administration in ICU for critically ill patients.
Definitions
The term “hyperoxia” generically refers to an excess oxygen supply (administration of any inspired oxygen fraction (FiO2) higher than 0.21) leading to a raise of PaO2 above normal values.
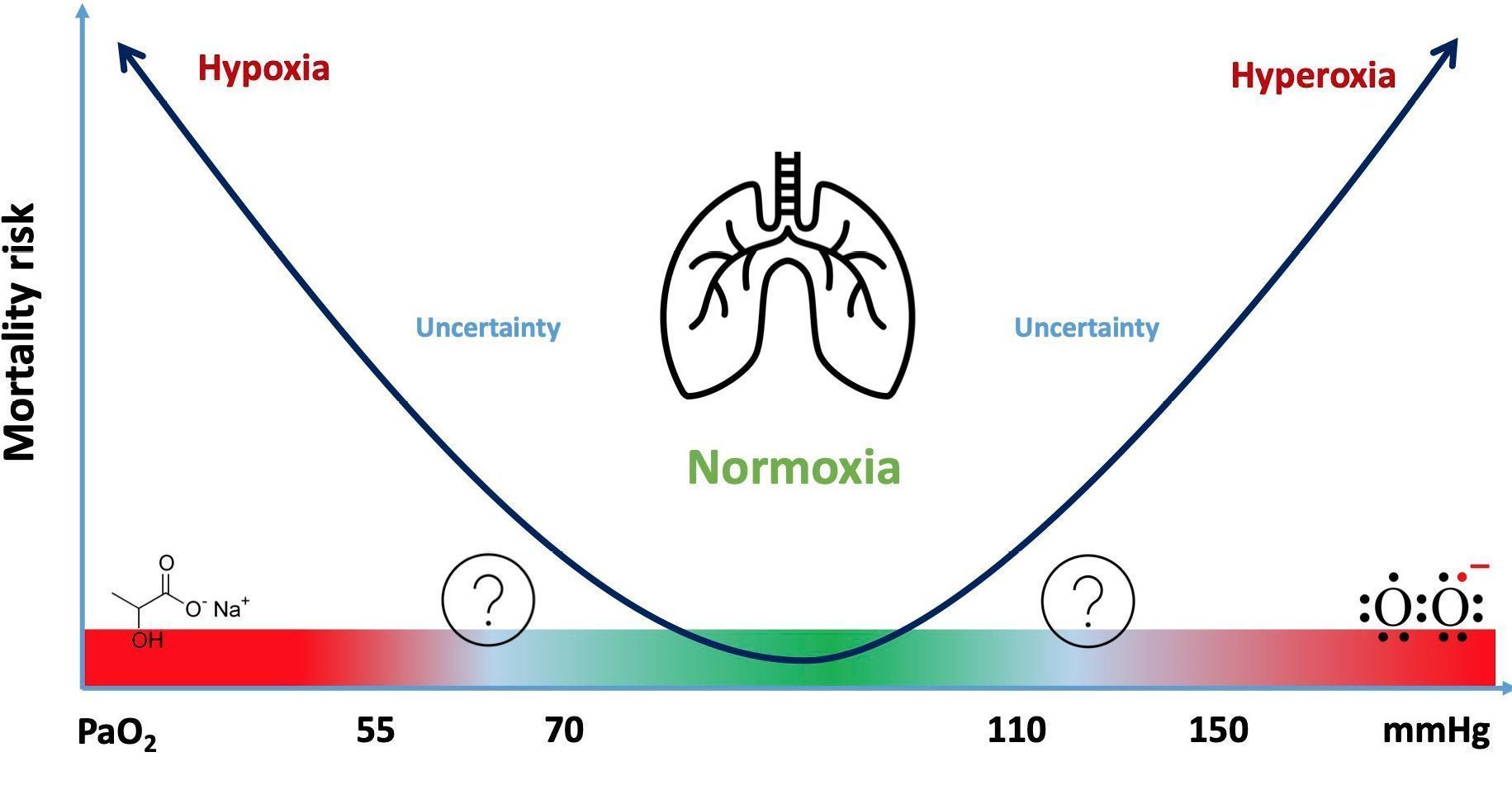
Normoxaemia is often defined as a PaO2 between 80 and 100 mmHg in a subject breathing room air at sea level. Unfortunately, clinical studies used a wide variety of definition for hyperoxia, explaining partly the inconsistency of findings (Barbateskovic et al. 2019; Damiani et al. 2014). A linear relationship was found between elevated PaO2 and mortality when PaO2 is analysed as a continuous variable, but without a strong threshold for harm (Janz et al. 2012). In a cohort study, ICU mortality appeared to increase linearly with exposure time even with mild hyperoxia [120-200 mmHg] (Helmerhorst et al. 2017a).
Hyperoxia Effects
Aerobic metabolism generates reactive oxygen species (ROS). Those are responsible for oxidative stress, which leads to cell death if not counterbalanced (Davies 1995). Thus, antioxidant systems are available in eucaryotic cells to eliminate ROS and maintain cell life. Situations of imbalance between oxidative stress and antioxidant defenses aggravate oxidative stress, cause damage to the cells with potential cell death and exacerbate the inflammatory response (Helmerhorst et al. 2015).
In mouse lungs, hyperoxia caused oxidative stress, increased inflammatory pulmonary response, increased pro-inflammatory cytokines and macrophages rates and created histological pulmonary damage (Helmerhorst et al. 2017b). Denitrogenation after high oxygen concentration breathing leads to alveolar collapse and lung atelectasis (Aboab et al. 2006).
Hyperoxia has also well-described systemic effects. Animal studies found greater spread of infection as well as altered organ perfusion and haemodynamics in models of septic and haemorrhagic shock occurring during hyperoxia (Dyson et al. 2009; Rodríguez-González et al. 2014). Whereas hypoxia may be viewed as a
natural response to injury, hyperoxia is never found except in conditions generated by human interventions. In this sense, oxygen should be assessed as a drug with possible side effects in case of overdosage.
Hyperoxia is Certainly Deleterious
Hyperoxia is a frequent event in critically ill patients. In a multicentre retrospective cohort study, Douin et al. (2021) showed that among 3,464 injured patients, 46% of ICU time was spent in hyperoxia defined by an arterial oxygen saturation (SaO2) above 96%. Hyperoxia was associated with a greater risk of in-hospital mortality.
Several randomised controlled trials reported deleterious effects of prolonged hyperoxia. Girardis et al. (2016) found an increased ICU mortality in 218 patients treated with a liberal oxygen therapy compared with 216 patients treated with a conservative strategy, defined by a PaO2 between 70 and 100 mmHg (20.2% vs. 11.6%, absolute risk reduction at 0.086 [95% confidence interval (CI) from 0.017 to 0.150], p = 0.01). Of note, in this study, the patients were hospitalised in the ICU for at least three days and received mechanical ventilation. Those findings were confirmed in a meta-analysis reporting a deleterious effect of prolonged hyperoxia exposure over time in a heterogeneous patient population (Chu et al. 2018).
Sepsis is associated with excessive radical oxygen species (ROS) formation from the nitric oxide (NO) pathway (Rodríguez-González et al. 2014). Additional ROS formation from hyperoxia could further worsen organ dysfunction. The HYPERS2S randomised controlled trial showed that hyperoxia, defined by a group treated with FiO2 at 1, increased the risk of 28-day mortality in patients with septic shock: 93 (43%) of 217 patients were non-survivors in the hyperoxia group versus 77 (35%) of 217 patients in the normoxia group [hazard ratio 1.27, 95% CI 0.94-1.72; p = 0.12] (Asfar et al. 2017). The authors reported a significant difference in the overall incidence of serious adverse events between the hyperoxia (185 [85%]) and normoxia groups (165 [76%]; p = 0.02), with a clinically relevant doubling in the hyperoxia group of the number of patients with ICU-acquired weakness (24 [11%] vs. 13 [6%]; p = 0·06) and atelectasis (26 [12%] vs 13 [6%]; p=0·04) compared with the normoxia group. This higher rate of complications resulted in the premature interruption of this trial.
A meta-analysis of the Cochrane group assessed the supplemental use of oxygen in patients with acute myocardial infarction. The authors concluded that randomised controlled trials did not support the routine use of inhaled oxygen in patients with acute myocardial injury, and a harmful effect of hyperoxia cannot be ruled out (Cabello et al. 2013). In cardiac arrest, high concentration of inhaled oxygen is routinely used during the resuscitation phases to increase presumably the tissular oxygen availability. Nevertheless, a randomised controlled trial showed an increase at 24 hours of neuronal injury markers in post-cardiac arrest patients ventilated with a FiO2 at 1 rather than those ventilated with a FiO2 at 0.3 (Kuisma et al. 2006). The 2021 European Resuscitation Council guidelines on post-resuscitation care recommend normoxia to improve recovery after cardiac arrest (Nolan et al. 2021).
Limitations and Questions
Despite this body of evidence, several studies did not confirm the deleterious effects of hyperoxia. In 205 patients with acute respiratory distress syndrome, a randomised controlled trial found that early exposure to a conservative oxygen therapy defined by a target arterial oxygen tension from 55 to 80 mmHg did not increase survival at day 28 (Barrot et al. 2020). Same findings were reported in another multicentre randomised controlled trial including 2928 patients with acute hypoxaemic respiratory failure (Schjørring et al. 2021). The ICU-ROX investigators randomised 1000 patients undergoing mechanical ventilation to receive conservative or usual oxygen therapy. The primary endpoint was the number of ventilator free-days, which was not significantly different between the two groups (Mackle et al. 2020). In a post-hoc analysis including 251 patients with sepsis, although the difference of 90-day mortality did not reach a significant level, the trend was in favour of the usual group, i.e. that with high target of PaO2 [36.2% vs. 29.2%, p = 0.24] (Young et al. 2020).
In brain injured patients, observational studies suggest a beneficial effect of early hyperoxia (Crawford et al. 2017; Jeon et al. 2014). However, high quality studies found no effect or even deleterious effects in the hyperoxia groups (Damiani et al. 2014; Roffe et al. 2017). In severe blunt chest trauma patients, a cohort study with a propensity score-based analysis found that early hyperoxia was not associated with more complications, including death and/or hospital-acquired pneumonia and/or acute respiratory distress syndrome. Hyperoxia was also associated with an increase in hospital-acquired pneumonia-free, mechanical ventilation-free, and ICU-free days (Duclos et al. 2021).
Several reasons may explain these conflicting findings. First, in observational studies, the most severe patients often develop hypoxia due to shock and acute respiratory failure. Then, the effects of oxygen threshold setting can be misleading by the weight of covariates. Second, it was now well-recognised that administration of high concentration oxygen is deleterious. Hence, the recent randomised controlled trials set the level of PaO2 in their control group around 90 mmHg (Barrot et al., 2020; Mackle et al. 2020; Schjørring et al. 2021). Even if this variable is not reported in the protocol, the medical teams in charge of patients pay attention to avoid hyperoxia. In the ICU-ROX, in both groups, the patients had a PaO2 below 100 mmHg from day 2 (Mackle et al. 2020). Actually, the control groups received a normoxic ventilation. The third explanation is the duration and timing of intervention. The studies in which hyperoxia was associated with improved outcome used high concentration of oxygen for a short duration at an early time-point after the injury (Duclos et al. 2021; Rockswold et al. 2013). In patients with traumatic brain injury, the administration of oxygen at a FiO2 set at 1 was performed only for three hours (Rockswold et al. 2013), while those with chest trauma were assessed for their oxygenation levels only during the first 24 hours (Duclos et al. 2021). Fourth, oxygen toxicity is probably increased among the patients receiving invasive mechanical ventilation (Girardis et al. 2016). Fifth, factors not related to oxygen administration can interfere with the oxygen metabolism. The effects of bringing more oxygen to tissue has been challenged during the nineties by increasing the cardiac index. A seminal randomised controlled trial showed an increased in mortality among patients receiving supra-therapeutic goals of oxygen delivery, as compared with those receiving standard of care (Hayes et al. 1994).
Conclusion
In critically ill patients, the risks associated with hypoxaemia have led to the administration of high quantity of oxygen (Suzuki et al. 2013; Young et al. 2015). Results from the more recent studies suggest that hyperoxia was associated with poor outcomes. The actual trend is to navigate towards a more conservative approach of oxygen administration. Nevertheless, even if a definitive threshold has not been discovered yet, a target of PaO2 ranging from 70 to 90 mmHg seems safe. However, the best timing of interventions on oxygen and the best monitoring of oxygen metabolism remains to be determined to develop personalised approaches within the next years in order to maintain critically ill patients within the safe zone of the U-shaped curve of hypoxaemia and hyperoxia (Figure 1).
Conflict of Interest
Pr. Leone reports personal fees from MSD, Pfizer, Octopharma, Aspen, Orion, Amomed, Aguettant outside the submitted work. Dr Pastene has nothing to disclose.
References:
Davies KJ (1995) Oxidative stress: The paradox of aerobic life. Biochemical Society Symposium, 61:1-31.
Douin DJ, Anderson EL, Dylla L et al. (2021) Association Between Hyperoxia, Supplemental Oxygen, and Mortality in Critically Injured Patients. Critical Care Explorations, 3(5):e0418.
Duclos G, Rivory A, Rességuier N et al. (2021) Effect of early hyperoxemia on the outcome in servere blunt chest trauma : A propensity score-based analysis of a single-center retrospective cohort. Journal of Critical Care, 63:179‑186.
Dyson A, Stidwill R, Taylor V, Singer M (2009) The impact of inspired oxygen concentration on tissue oxygenation during progressive haemorrhage. Intensive Care Medicine, 35(10):1783‑1791.
Girardis M, Busani S, Damiani E et al. (2016) Effect of Conservative vs Conventional Oxygen Therapy on Mortality Among Patients in an Intensive Care Unit : The Oxygen-ICU Randomized Clinical Trial. JAMA, 316(15):1583‑1589.
Hayes MA, Timmins AC, Yau EH et al. (1994) Elevation of systemic oxygen delivery in the treatment of critically ill patients. The New England Journal of Medicine, 330(24):1717‑1722.
Helmerhorst HJF, Arts DL, Schultz MJ et al. (2017) Metrics of Arterial Hyperoxia and Associated Outcomes in Critical Care. Critical Care Medicine, 45(2):187‑195.
Helmerhorst HJF, Schouten LRA, Wagenaar GTM et al. (2017) Hyperoxia provokes a time- and dose-dependent inflammatory response in mechanically ventilated mice, irrespective of tidal volumes. Intensive Care Medicine Experimental, 5(1): 27.
Helmerhorst HJF, Schultz MJ, van der Voort PHJ et al. (2015) Bench-to-bedside review : The effects of hyperoxia during critical illness. Critical Care (London, England), 19:284.
Janz DR, Hollenbeck RD, Pollock JS et al. (2012) Hyperoxia is associated with increased mortality in patients treated with mild therapeutic hypothermia after sudden cardiac arrest. Critical Care Medicine, 40(12):3135‑3139.
Jeon S.-B, Choi HA, Badjatia N et al. (2014) Hyperoxia may be related to delayed cerebral ischemia and poor outcome after subarachnoid haemorrhage. Journal of Neurology, Neurosurgery, and Psychiatry, 85(12):1301‑1307.
Kuisma M, Boyd J, Voipio V et al. (2006) Comparison of 30 and the 100% inspired oxygen concentrations during early post-resuscitation period : A randomised controlled pilot study. Resuscitation, 69(2):199‑206.
Mackle D, Bellomo R, Bailey M et al. (2020) Conservative Oxygen Therapy during Mechanical Ventilation in the ICU. The New England Journal of Medicine, 382(11):989‑998.
Nolan JP, Sandroni C, Böttiger BW et al. (2021) European Resuscitation Council and European Society of Intensive Care Medicine guidelines 2021 : Post-resuscitation care. Intensive Care Medicine, 47(4):369‑421.
O’Driscoll BR, Howard LS, Earis J et al. (2017) BTS guideline for oxygen use in adults in healthcare and emergency settings. Thorax, 72(Suppl 1):ii1‑ii90.
Rockswold SB, Rockswold GL, Zaun DA, Liu J. (2013) A prospective, randomized Phase II clinical trial to evaluate the effect of combined hyperbaric and normobaric hyperoxia on cerebral metabolism, intracranial pressure, oxygen toxicity, and clinical outcome in severe traumatic brain injury. Journal of Neurosurgery, 118(6):1317‑1328.
Rodríguez-González R, Martín-Barrasa JL, Ramos-Nuez Á et al. (2014) Multiple system organ response induced by hyperoxia in a clinically relevant animal model of sepsis. Shock, 42(2):148‑153.
Roffe C, Nevatte T, Sim J et al. (2017) Effect of Routine Low-Dose Oxygen Supplementation on Death and Disability in Adults With Acute Stroke : The Stroke Oxygen Study Randomized Clinical Trial. JAMA, 318(12):1125‑1135.
Schjørring OL, Klitgaard TL, Perner A et al. (2021) Lower or Higher Oxygenation Targets for Acute Hypoxemic Respiratory Failure. The New England Journal of Medicine, 384(14):1301‑1311.
Suzuki S, Eastwood GM, Peck L et al. (2013) Current oxygen management in mechanically ventilated patients : A prospective observational cohort study. Journal of Critical Care, 28(5):647‑654.
Young PJ, Beasley RW, Capellier G, Eastwood GM et al. (2015) Oxygenation targets, monitoring in the critically ill : A point prevalence study of clinical practice in Australia and New Zealand. Critical Care and Resuscitation: Journal of the Australasian Academy of Critical Care Medicine, 17(3):202‑207.
Young P, Mackle D, Bellomo R et al. (2020) Conservative oxygen therapy for mechanically ventilated adults with sepsis : A post hoc analysis of data from the intensive care unit randomized trial comparing two approaches to oxygen therapy (ICU-ROX). Intensive Care Medicine, 46(1):17‑26.








