ICU Management & Practice, Volume 16 - Issue 3, 2016
Even though systemic haemodynamic variables may be normalised, there could be regions with inadequate regional oxygenation at the tissue level. The most recent developments of noninvasive monitoring of the peripheral circulation have helped physicians to early identify patients at high risk for tissue hypoperfusion, organ failure and poor outcome.
Why Might Clinical Assessment of Peripheral Circulation be Helpful?
Examination of peripheral circulation is easily done by touching the skin, measuring capillary refill time (CRT) or even by observing the skin mottling pattern. The cutaneous vascular bed plays an important role in thermoregulation of the body, and this process can result in skin circulation alterations that have direct effects on skin temperature and colour, i.e., a cold, clammy, white and mottled skin. There are different methods to clinically assess the peripheral perfusion.
Mottle Score
Pallor, mottling and cyanosis are key visual indicators of reduced skin circulation, which can be scored by just looking at the skin. Mottling is the result of heterogenic small vessel vasoconstriction and is thought to reflect abnormal skin perfusion. It is defined as a bluish skin discolouration that typically manifests near the elbows or knees and has a distinct patchy pattern.
Capillary Refill Time
Capillary refill time (CRT) is defined as the time required for a distal capillary bed (e.g. nail bed, forehead or knee) to regain its colour after pressure has been applied to cause blanching. Over the past 30 years, the definition of a delayed CRT has been debated in the literature. Very few studies have addressed the CRT normal range in adults and its relation to body site, effect of ambient or skin temperature, and its reliability among examiners. Compelling recent studies have demonstrated that the interrater reliability for CRT measurement between examiners showed substantial agreement for the strategy of subjective CRT evaluation at the bedside (Ait-Oufella et al. 2014; van Genderen et al. 2014b). Assuming normal core temperature, decreased skin blood flow as the cause of delayed CRT can be estimated by measuring skin temperature, since cold extremities reflect constriction of cutaneous vessels that ultimately decreases the amount of blood volume within peripheral vasculature. By contrast, peripheral vasodilation has the opposite effect. Inducing peripheral vasodilation with nitroglycerin infusion in patients with shock after haemodynamic stabilisation and with much delayed CRT resulted in significant decrease in CRT by 51% toward normal compared with baseline values (Lima et al. 2014).
Skin temperature
One should pay attention to how to evaluate skin temperature. A temperature gradient can better reflect changes in cutaneous blood flow than the absolute skin temperature itself. As peripheral temperature may be influenced by ambient temperature, a gradient between forearm and finger temperature may be a more reliable measurement, as the two skin temperatures are exposed to the same ambient temperature. Assessing skin temperature by touching the extremities or measuring a body temperature gradient can assist the physician to recognise a clinically acceptable CRT, which is more predictive in warm extremities conditions. Because of a conditional effect, cold extremities will often be related to a delayed CRT. Therefore, if the extremities are cold, one should expect a delayed CRT and CRT will not be much help for the clinician. On the other hand, warm extremities indicate adequate cutaneous blood flow and one should expect a normal CRT, and a delayed CRT in this condition suggests cutaneous microcirculatory derangement (Lima et al. 2011).
See Also:Study: Hyperoxia Alters Microcirculation in Healthy Volunteers
Minimally
Invasive Technologies to Assess Peripheral
Circulation
Despite all the technological innovation in monitoring peripheral circulation, there has been little success in incorporating these technologies in clinical practice. Many factors contribute to this, but their clinical use still faces some hurdles for adoption. The signal from these devices is accessible at regional level and is often unfamiliar to the doctor, who is unable to contextualise its use in order to influence critical care. This is a barrier against their acceptance, as is the high cost of these devices. To be successful, the technique should be feasible for routine use at the bedside, robust, easy to use and to integrate into care. Minimally invasive technologies that cover some of those criteria include optical monitoring devices and transcutaneous measure- ment of oxygen tension (Table 1) (Lima and Bakker 2005). Optical monitoring utilises the optical properties of haemoglobin to measure partial pressure of oxygen and haemoglobin saturation. Commonly used optical methods in the clinical setting that are able to monitor tissue oxygenation at the bedside include nearinfrared spectroscopy and direct visualisation of the sublingual microcirculation. Continuous transcutaneous measurement of oxygen tension is based on the electrochemical properties of noble metals to measure the oxygen content of the tissue.
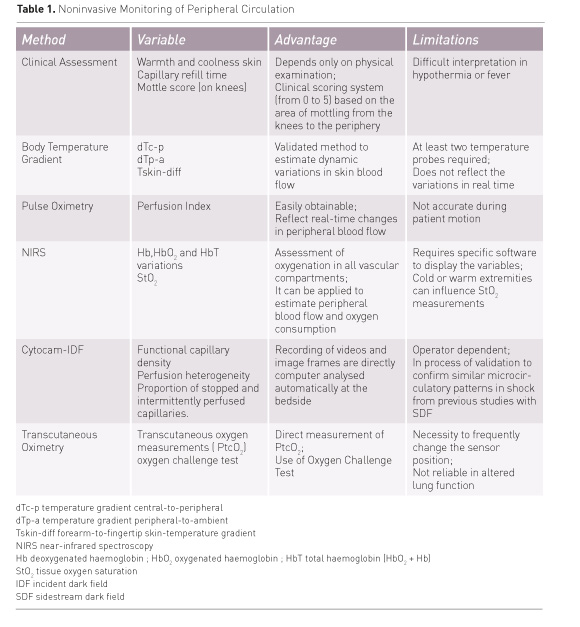
Near-Infrared Spectroscopy (NIRS)
The utility of NIRS for managing critically ill patients remains a matter of debate (Macdonald and Brown 2015). A new trend for the NIRSderived StO2 application is in predicting complications and early identification of septic patients at high risk for microcirculatory failure during specific haemodynamic therapeutic interventions, such as vasopressor and blood transfusion therapy (Conrad et al. 2015; Damiani et al. 2015). These studies outline the new trend of the clinical application of NIRS as a potential candidate to evaluate tissue monitoring during clinical treatment of those diseases that impact microvascular function, such as sepsis and hypovolaemia. However, there are some drawbacks of NIRS technology that still have to be addressed in future studies. Changes in StO2 values may be confounded by factors other than the true marker of thenar muscle oxygenation, and StO2 values may mislead the bedside clinician to assume that tissue hypoxia is present, when this change may merely reflect low blood flow to the superficial layers above the muscle capillary beds. In addition, the technique for using vascular occlusion test (VOT) has not been standardised. Currently, various types and degrees of deflation thresholds (StO2 of 10% or 40%; duration of 3 or 5 minutes) are used and no supporting evidence in the literature shows which of the methods is superior and more reliable to assess the VOT-derived StO2 slopes. These highlight a necessary further step in evaluating the NIRS clinical utility and its possible use in predicting complications and early identification of patients at risk for microcirculatory failure.
Direct Visualisation of the Sublingual Microcirculation
Due to rapid advances in technology, microcirculation evaluation has been dynamic work, and new devices have been introduced that improve microcirculatory image acquisition. Recently, a more advanced version of handheld microscopes (CytoCam, Braedius Medical, Naarden, The Netherlands), based on Incident Dark Field (IDF), has been introduced to overcome persistent limitations of the earlier devices (Hutchings et al. 2015). The main technological improvements of IDF include higher optical resolution, lower weight of the device and digital signal allowing more vessels to be observed with larger detail. In addition, recording of videos and image frames are directly computer analysed automatically at the bedside. Whether IDF measurements can reproduce and confirm similar microcirculatory patterns in shock from the previous studies with SDF needs to be confirmed. Nevertheless, some studies have compared SDF imaging and CytoCam IDF imaging in healthy subjects and neonates with promising results (Aykut et al. 2015; Gilbert-Kawai et al. 2016; van Elteren et al. 2015). Other studies have focused on the feasibility of monitoring and analysing sublingual microcirculation by nurses at the bedside using the Cytocam IDF (Tanaka et al. 2015). Bringing to the bedside the complete package of microcirculation analysis is an initial step towards the incorporation of physicians and nurses into the measurement and interpretation of microcirculation status at the bedside (Lima et al. 2015).
Continuous PtcO2 transcutaneous measurement
Oxygen sensors for transcutaneous
electrochemical measurements are based on polarography: a typical
amperometric transducer in which the rate of a chemical reaction is detected
by the current drained through an electrode. The sensor heats the skin to
43-45ºC, and as a result the skin surface oxygen tension is increased. These
transcutaneous sensors enable us to directly estimate arterial oxygen pressure
(PaO2). However, in adults the skin is thick, and differences in the skin
cause the be lower than PaO2. The correlation between PtcO2 and PaO2 also
depends on the adequacy of blood flow. The low blood flow caused by vasoconstriction
during shock overcomes the vasodilatory effect of PtcO2 sensor. This causes a
mild tissue hypoxia beneath the PtcO2 sensor. The lack of the PtcO2 ability to
accurately reflect the PaO2 in low flow shock enables us to estimate cutaneous
blood flow through the relationship between the two variables. The PtcO2 and
PaO2 values are almost equal when the blood flow is adequate. During low flow
shock, however, the PtcO2 will drop and becomes dependent on the PaO2 value.
Some studies have suggested the use of an oxygen challenge test, which refers to
the lack of PtcO2 rise in response to high oxygen inspired fraction in
patients with normal lung function, and has shown good predictive value for
unfavourable outcome of septic shock patients (Mari et al. 2014; Schlager et
al. 2014). Some technical aspects should be considered when performing the
oxygen challenge test, such as microcirculatory modifications due to heat-induced
vasodilation by the electrode, and altered cutaneous vasomotor reactivity due
to the transient hyperoxia. Altered lung function might have an influence on
PtcO2 and thus affect the oxygen challenge test. For example, a PaO2 at 100%
inspired oxygen fraction can remain low with no subsequent increase of PtcO2 during
the oxygen challenge test (Mari et al. 2014).
Clinical Implications
Recent advances in diagnostic and monitoring technologies have helped intensivists to better understand the complex pathophysiology of acute circulatory failure. The power and objectivity provided by these new technologies might cause us to think that peripheral circulation examination in the intensive care setting has become obsolete. Much emphasis is given to the global variables of perfusion, whereas relatively little is said about less vital organs, like skin and/or muscle. One may argue about the clinical significance of monitoring circulation of these non-vital organs in which blood flow is not crucial for the immediate survival. Abnormalities in peripheral circulation may still persist although systemic haemodynamic stability has been reached. Moreover, the persistence of these alterations has been associated with worse outcomes (Chien et al. 2007; Poeze et al. 2005). Therefore, some argue that following normalisation of circulation parameters, global systemic parameters are of less importance (Dunser et al. 2013). In fact physicians often lose sight of this important point, placing too much emphasis on systemic haemodynamic variables while failing to take the time to perform a simple physical examination of peripheral circulation. The absence of cold extremities, delayed CRT or mottled skin after initial resuscitation identifies patients with a more favourable outcome (Ait-Oufella et al. 2011, Lima and Takala, 2014). One next logical step, therefore, would be incorporating therapeutic strategies into resuscitation protocols that aim at normalising (peripheral) circulation parameters to investigate the impact of peripheral circulation target resuscitation in the survival of critically ill patients (van Genderen et al. 2014a). Thoughtfully integrated with the new technology, the clinical assessment of peripheral circulation should and will continue to be central to intensive care clinical practice.
Conflict of Interest
Alexandre Lima declares that he has no conflict of interest. Michel van Genderen declares that he has no conflict of interest.
Abbreviations
CRT capillary refill time
NIRS near infrared spectroscopy
VOT vascular occlusion test
References:
Ait-Oufella H, Bige N, Boelle PY et al. 2014. Capillary refill time exploration during septic shock. Intensive Care Med, 40(7): 958-64.
PubMed ↗
Aykut G, Veenstra G, Scorcella C et al. (2015) Cytocam-Idf (incident dark field illumination) imaging for bedside monitoring of the microcirculation. Intensive Care Med Exp, 3(1): 40.
PubMed ↗
Conrad M, Perez, P, Thivilier C et al. (2015) Early prediction of norepinephrine dependency and refractory septic shock with a multimodal approach of vascular failure. J Crit Care, 30: 739-43.
PubMed ↗
Damiani E, Adrario E, Luchetti MM et al. (2015) Plasma free hemoglobin and microcirculatory response to fresh or old blood transfusions in sepsis. PLoS One, 10(5): e0122655.
PubMed ↗
Gilbert-Kawai E, Coppel J, Bountziouka V et al. (2016) A comparison of the quality of image acquisition between the incident dark field and sidestream dark field video-microscopes. BMC Med Imaging, 16: 10.
PubMed ↗
Hutchings S, Watts S, Kirkman E (2015) The Cytocam video microscope. A new method for visualising the microcirculation using Incident Dark Field technology. Clin Hemorheol Microcirc, 62(3): 261-71.
PubMed ↗
Lima A, Bakker J (2005) Noninvasive monitoring of peripheral perfusion. Intensive Care Med, 31(10): 1316-26.
PubMed ↗
Lima A, López A, Van Genderen ME et al. (2015) Interrater reliability and diagnostic performance of subjective evaluation of sublingual microcirculation images by physicians and nurses: a multicenter observational study. Shock, 44(3): 239-44.
PubMed ↗
Lima A, Van Bommel J, Sikorska K et al. (2011) The relation of near-infrared spectroscopy with changes in peripheral circulation in critically ill patients. Crit Care Med, 39(7): 1649-54.
PubMed ↗
Lima A, Van Genderen ME, Van Bommel J et al. (2014) Nitroglycerin reverts clinical manifestations of poor peripheral perfusion in patients with circulatory shock. Crit Care, 18(3): R126.
PubMed ↗
Macdonald SP, Brown SG (2015) Near-infrared spectroscopy in the assessment of suspected sepsis in the emergency department. Emerg Med J, 32(5): 404-8.
PubMed ↗
Mari A, Vallée F, Bedel J et al. (2014) Oxygen challenge test in septic shock patients: prognostic value and influence of respiratory status. Shock, 41(6): 504-9.
PubMed ↗
Schlager O, Gschwandtner ME, Willfort-Ehringer A et al. (2014) Transcutaneous oxygen tension monitoring in critically ill patients receiving packed red blood cells. J Crit Care, 29(6): 1057-62.
PubMed ↗
Tanaka S, Harrois A, Nicolaï C et al. (2015) Qualitative real-time analysis by nurses of sublingual microcirculation in intensive care unit: the MICRONURSE study. Crit Care, 19: 388. doi: 10.1186/s13054-015-1106-3.
PubMed ↗
Van Elteren HA, Ince C, Tibboel D et al. (2015) Cutaneous microcirculation in preterm neonates: comparison between sidestream dark field (SDF) and incident dark field (IDF) imaging. J Clin Monit Comput, 29(5): 543-8.
PubMed ↗
van Genderen ME, Klijn E, Lima A, et al. (2014a) Microvascular perfusion as a target for fluid resuscitation in experimental circulatory shock. Crit Care Med, 42, e96-e105.
PubMed ↗
van Genderen ME, Paauwe J, de Jonge J et al. (2014b) Clinical assessment of peripheral perfusion to predict postoperative complications after major abdominal surgery early: a prospective observational study in adults. Crit Care, 18(3): R114.
PubMed ↗








