ICU Management & Practice, ICU Volume 13 - Issue 2 - Summer 2013
Why Use an Indirect Calorimeter in the ICU?
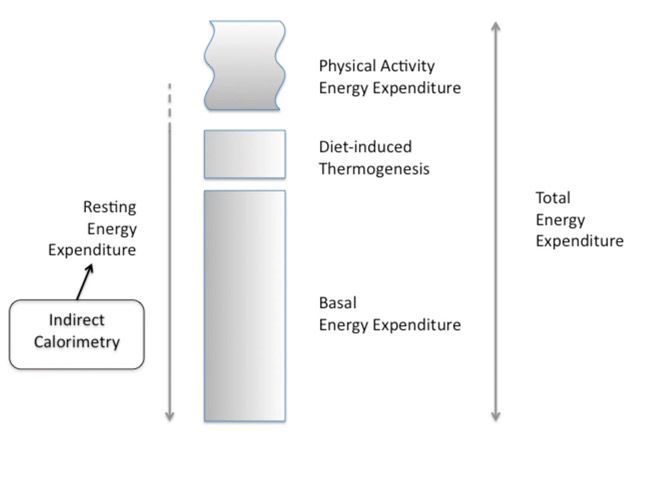
The magnitude of the caloric debt (the difference between energy expenditure (EE) and the caloric intake) has been strongly associated with the rate of complications occurring in ICU patients (Dvir et al. 2006; Faisy et al. 2009; Villet et al. 2005). A large caloric debt is partly explained by the difficulties in providing the desired caloric intake by the enteral route (Artinian et al. 2006). Conversely, a risk of overfeeding (ie caloric intake > EE) is present when EE is undefined, especially when parenteral nutrition is used (Dissanaike et al. 2007; Gramlich et al. 2004; The Veterans Affairs Total Parenteral Nutrition Cooperative Study Group, 1991). Hence, both over- and underfeeding will be prevented when the caloric intake matches the actual EE (Singer et al. 2010). However, some recent data (Arabi et al. 2011; Casaer et al. 2011; Rice et al. 2012; Vincent and Preiser, 2013) challenge the concept of EE as a guide for the prescription of calories, when a non-inhibitable endogenous production of glucose provides a large amount of calories during the early phase of critical illness. Other recent studies (Heidegger et al. 2013; Singer et al. 2011) found some clinical benefit of an IC-guided caloric intake, as compared to an exclusive enteral regimen, irrespective of the EE. In any case the accurate measurement of EE can merely help to prevent overfeeding in longstaying patients.The Total EE (TEE) includes three components (see Figure 1): the
Basal Energy Expenditure (BEE) or Basal Metabolic Rate (BMR), the energy used
during substrate metabolism, including the diet-induced thermogenesis (DIT) and
the energy required for physical activity (Haugen et al. 2007; Lev et al.
2010). The BEE is the energy required to maintain the body’s basic cellular metabolic
activity and organ functions (Haugen et al. 2007). BEE is very close to the
resting energy expenditure (REE) when a postabsorptive individual is resting,
but not in critically ill patients (Lev et al. 2010).
Figure 1. Components of the Energy Expenditure
EE is extremely difficult to predict during critical illness. Traditionally,
the EE of a critically ill patient is considered to be higher than the EE
predicted for a matched healthy subject, although some ICU patients could be
hypometabolic (Magnuson et al. 2011; McClave et al. 2013; Siirala et al. 2010).
The prediction of EE is further complicated by its large variations over a stay
in ICU (Uehara et al. 1999). Even the complex predictive equations based on
different anthropometrical and vital parameters with or without correction
stress factor (Frankenfield et al. 2009; Frankenfield et al. 2012;
Frankenfield, 2011) are inaccurate in critically ill patients (Avitzur et al.
2003; Frankenfield et al. 2009).
A calorimeter is designed to measure EE, calculated from the
amount of exhaled of carbon dioxide (VCO2) and consumed oxygen (VO2) (Haugen et
al. 2007).
Principles of Calorimetry (Table 1)

Calorimetry is a measure of the caloric expenditure of a patient through the production of heat by metabolic process (Haugen et al. 2007). As direct calorimetry cannot be used at the bedside, indirect calorimetry (IC) was elaborated by measuring respiratory gases (Haugen et al. 2007). The VO2 and VCO2 are measured by analysis of the gas exchanged, and the modified Weir equation allows the calculation of TEE (see Table 1, equation 1) (Weir, 1949). The measurement of VO2 and VCO2 is calculated as the difference in the volume of inspired and expired air multiplied by the concentration of the content of O2 and CO2 respectively (see Table 1, equation 3). Because the small difference between inspiratory (Vi) and expiratory volumes (Ve) is difficult to measure, Vi is commonly calculated using the Haldane transformation, which assumes that inert gas nitrogen (N2) is constant in both inspired and expired gases (see Table 1, equation 4). In this simplification the risk of error on the denominator increases as FiO2 increases, especially when FiO2 is above 60% or when anaesthetic gases or nitric oxide are inhaled (Walsh, 2003). REE can be derived from VO2 alone (see Table 1, equation 5) if the patient does not have respiratory failure and is in a stable condition. In conditions of acute lung injury this equation does not take into account the VO2 of the lung, which may reach 15-20% of the total VO2 (Bursztein et al. 1989). The urinary nitrogen content (uN2) is often neglected because it only accounts for less than 4% of the TEE (see Table 1, equation 2) (Bursztein et al. 1989; Haugen et al. 2007). The Respiratory Quotient (RQ) is the ratio between VCO2 and VO2 (VCO2/VO2), and represents the relative metabolism of the different macronutrients. The carbohydrate and fat oxidation could be calculated from these measures (see Table 1, equations 6) (Bursztein et al. 1989). A value of 1.0 corresponds to a complete glucose oxidation. RQ of the proteins is less and lower for the lipid. The physiologic human range is 0.67-1.2. If the value is outside a range of 0.7 to 1.0, the quality of the measurement should be questioned (Compher et al. 2006; Haugen et al. 2007).
IC was validated compared to reference techniques (direct calorimetry,
double labelled water technique) (Lev et al. 2010). IC was largely used in
various categories of ICU patients (Kemper et al. 1992; Kyle et al. 2012). The
ICU nutrition guidelines proposed the use of IC instead of predictive equations
when it is available, particularly for lean and obese patients, and in patients
with burn injuries and liver diseases (Kreymann et al. 2006; McClave et al.
2009; Singer et al. 2009). The integration of the IC in a computerised
information system allows automatic calculation of the caloric debt (Dvir et al.
2006; Berger et al. 2006). The respiratory quotient (RQ) could also be used to
assess the presence of under- (RQ < 0.85) or overfeeding (RQ > 1.0), but
with a low sensitivity and specificity (McClave et al. 2003a). RQ >= 1.0 may
be associated with reduced tolerance and mild respiratory compromise (McClave
et al. 2003a).
The devices are described as closed-circuit indirect calorimeters
if the inspired air comes from an air or oxygen tank inside the calorimeter. However,
open-circuit indirect calorimeters (inspired air source is room air or
ventilator) are the most widely used in ICU (Lev et al. 2010). The expensive,
cumbersome and oldest machines were composed of a Douglas bag in which the expiratory
gases were blended (Schoeller 2007). The commercial metabolic cart contains a
mixing chamber inside and gas analysers. The expired gas is mixed with a flow
of room air, large enough to ensure constant total flow. The Deltatrac II
(Datex-Ohmeda, General Electrics, Finland) was the most frequently used and
classical IC, but is no longer marketed (Schoeller 2007). Technical expertise
and regular calibration were needed, such as warming delay (about 20-30
minutes) (Lev et al. 2010). Only a few dedicated centres still use these devices.
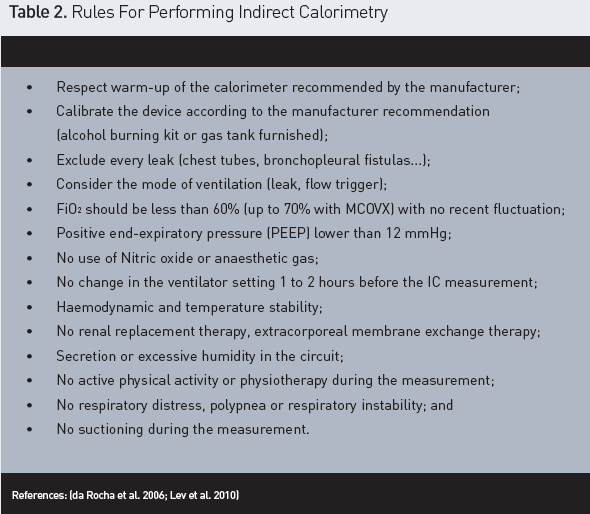
How Should We Use an Indirect Calorimeter? (Figure 2 and Table 2)
In a systematic review, Compher et al. define best practice
methods to measure the REE in adults. A minimum rest of 10 to 20 minutes, and a
room temperature of 20 to 25°C are requested (Compher et al. 2006).
Measurements recorded during the first five minutes are discarded. A five
minute period with <= 10% coefficient of variation for VO2 and VCO2 would be suitable (Compher et al.
2006; Lev et al. 2010). If steady state is not achieved, two or three
non-consecutive measures improve accuracy. Smyrnios et al. compared
measurements of 24-hour duration with a 30-minute duration and found no major
differences (Smyrnios et al. 1997). The best time was found to be around noon.
For unstable patients, a two-hour measurement was demonstrated to minimise the
rate of error (Smyrnios et al. 1997; Zijlstra et al. 2007). McClave et al.
showed that taking the mean REE from a 5-minute steady state (10% variation) provides
the most accurate value, which can be extrapolated to represent the 24-hour
total REE (McClave et al. 2003b). In a population of critically ill children, a
reduction of the interval of steady state resulted in a significantly greater number of
patients reaching steady state without significantly changing accuracy
(Smallwood and Mehta 2012). For a patient not ventilated, a canopy system could
be used but required a high gas flow to avoid carbon dioxide accumulation and
the need to detect very small differences in gas concentration (Walsh 2003).
Any air leak in the circuit (endotracheal tube cuff leak, leaking
chest tubes or bronchopleural fistulas) (Compher et al. 2006; Lev et al. 2010) will
affect the measure as will any procedure that affects gas exchange
(extracorporeal CO2removal or extracorporeal membrane oxygenator or renal replacement
therapy) (Lev et al. 2010).
Connecting the indirect calorimeter to ventilators with large bias
flow (flow-triggering) could be a problem (Lev et al. 2010).
An increase of the energy consumption occurs also when the patient
was agitated, suffered from pain or had fever or shivering (Chiolero et al.
1997) Lastly, attention must be paid to the presence of secretions or
condensation retained in the tubing of the ventilatory circuit (McClave et al.
2013). A high or low RQ could also be calculated if the proportion of fat and
glucose is too high or too low (Haugen et al. 2007). Finally, the device must
be adequately and regularly calibrated (Compher et al. 2006; Haugen et al.
2007).
Performance of Currently Available Indirect Calorimeters: Pitfalls and Limitations
Some handheld indirect calorimeters have been developed by the
industry with breath-to-breath analysis (Haugen et al. 2007). These devices
collect expired air via a face mask or canopy or directly from the exit port of
the ventilator (Lev et al. 2010). The performance of the MedGem (HealtheTech,
Golden, CO), a handheld device placed inside the canopy was validated against the
reference Deltatrac (Datex-Ohmeda, Madison, WI) device in a healthy population
(Stewart et al. 2005).
Recently, some devices were proposed for critically ill patients. M-COVX (metabolic monitor in S/5 Critical Care Monitor, Datex- Ohmeda) was compared to Deltatrac II in twenty mechanically ventilated, critically ill patients and was found to yield an acceptable intra-patient reproducibility (less than 2% for VO2, VCO2 and REE) (McLellan et al. 2002). In contrast, a comparison of the Deltatrac II to the same MCOVX and the Evita 4 monitoring devices in 43 ventilated critically ill patients found poor agreement despite good correlation between measurements (Singer et al. 2006). Deltatrac MBM 100 (Datex) was compared with Vmax Encore 29n (SensorMedics) in healthy subjects and a lack of accuracy, comparability and transferability of the results was shown, but could be improved by a post-calorimetric calibration (Schadewaldt et al. 2013). However, this timeconsuming procedure was unrealistic in a common clinical setting. Similar disappointing results were found in healthy subjects with the Deltatrac II compared to five other devices: MedGraphics CPX Ultima, MedGem, Vmax Encore 29 System, TrueOne 2400, and Korr ReeVue (Cooper et al. 2009). Sundström et al. compared Deltatrac to more recent calorimeters in mechanically ventilated patients, the Quark RMR (Cosmed, Italy) and the CCM Express (MedGraphics Corp., St Paul, Minneapolis, USA) and found conflicting estimationsof EE (Sundstrom et al. 2013). Anothercomparative study found the same results (Grafet al. 2011). In these two last studies, the QuarkRMR device showed better results than the CCMexpress but with unacceptable variations for theclinical purpose.
The
devices are usually expensive (25,000 to 40,000) and the manpower cost is high (Cooney and Frankenfield,
2012). The period of measurement is long and a warming time has to be awaited
before starting the process. The calibration time and the cost of the tank of gas
for calibration could also be barriers. Davis et al. calculated that omitting
IC could save $33,000 in their surgical ICU, but in their study the predictive
equation performed well in contrast to other studies (Avitzur et al. 2003;
Cutts et al. 1997; Davis et al. 2006; Frankenfield et al. 2009). The
limitations cited above of such ventilation with no more than 60 % FiO2, no high pressure
(PEEP above 12 mmHg), no othergas inhaled or
gas exchange therapy are alsomajor drawbacks
limiting the number of patientsthat could
benefit from the technique.
A recent validation of IC (Siirala et al. 2012) in healthy
volunteers undergoing pressure controlled non-invasive ventilation support was published,
but numerous flaws exist and other confirmatory studies are needed (Esquinas
and Koksal 2013).
The IC measures the REE that is extrapolated to the rest of the
day but physical activity is not included. In clinical practice the experts recommend
to add 10 to 15% to the RMR measured to approach the TEE (Schoeller, 2007). However,
this activity factor is difficult to estimate in ICU. Physiotherapy, the
patient’s agitation and the early mobilisation that is recommended nowadays are
difficult to evaluate. Even if a continuous recording is made, the pitfalls secondary
to a higher respiratory rate (Walsh, 2003) and respiratory instability
precludes a precise evaluation of the TEE.
What’s Next?
Definitely more appropriate devices are needed to assess the
clinical relevance of using an indirect calorimetry-guided approach for the management
of caloric prescription in critically ill patients. A task force of experts has
been launched by the European Society of Clinical Nutrition and Metabolism
(ESPEN) to cooperate with industrial partners on the development of reliable
indirect calorimeters useable in ICUs. In parallel the TICACOS study (NCT01479673)
aims to assess the necessity for measuring daily resting energy expenditure as
a guide for nutritional support (Singer et al. in progress). The research
hypothesis of this study is that tight caloric control will reduce the rate of
new infections.
Abbreviations
BEE : Basal Energy Expenditure
BMR : Basal Metabolic Rate
DIT : Diet-induced thermogenesis
EE : Energy Expenditure
FeO2 : fraction of expired oxygen;
FiO2 : fraction of inspired oxygen;
IC : Indirect Calorimetry
REE : Resting Energy Expenditure
RMR: Resting Metabolic Rate
RQ : Respiratory Quotient
TEE : Total Energy Expenditure
uN2 : urinary nitrogen component;
VCO2 : production of CO2;
Ve : Expiratory Volume;
Vi : Inspiratory Volume;
VO2 : Consumption of O2.







