ICU Management & Practice, Volume 18 - Issue 4, 2018
Dengue is a febrile illness, caused by one of the serotypes of Flavivirus (DENV1-4), transmitted by Aedes aegypti and Aedes albopictus mosquitoes during a blood meal (Simmons et al. 2012; Guzman et al. 2015; Pai-Dhungat et al. 2013). The vectors live in an urban habitat and are daytime feeders. Aedes albopictus is now spreading to North America and Europe due to international trade. The island of Madeira, Portugal, and five other European countries have witnessed an outbreak of dengue (Guzman et al 2015).
The World Health Organization (WHO) currently estimates there may be 50-200 million dengue infections worldwide every year. Half of the world’s population living in endemic areas is at risk (Pai-Dhungat et al. 2013). Recovery from infections in one serotype provides lifelong immunity against that serotype. However, cross immunity is only partial and temporary. Subsequent infection by either serotype increases the risk of developing severe dengue.
Clinical features
As per the WHO 1997 classification, dengue comprises a clinical spectrum, which consists of
- Dengue fever (breakbone fever)
- Haemorrhagic fever and
- Dengue shock syndrome (WHO 1997)
The revised WHO classification scheme (2012) consists of
- Dengue without warning signs
- Dengue with warning signs
- Severe dengue
Expanded dengue comprises unusual or atypical manifestations of dengue which can happen in the absence of plasma leakage.
Clinical features of dengue fever without warning signs
- Fever which lasts 5 to 7 days
- Myalgia/arthralgia
- Retro-orbital pain
- Rash
- Haemorrhagic manifestations – Petechie, ecchymosis, mucosal bleed, positive tourniquet test
- Leucopaenia and thrombocytopaenia
Clinical features of dengue with warning signs
- Abdominal pain
- Persistent vomiting
- Mucosal bleed
- Lethargy
- Hepatomegaly
- Rapid decline in platelet count
The differentiating features of dengue with warning signs consist of haemoconcentration with a more than 20% rise in haematocrit, pleural effusion and ascites secondary to plasma leakage because of increased vascular permeability.
Clinical features of severe dengue
- Severe plasma leakage leading to cold clammy skin, thready pulse, narrowed pulse pressure, shock, fluid accumulation with respiratory distress
- Severe bleeding
- Organ involvement
- Liver: aspartate aminotransferase (AST) or alanine aminotransferase (ALT) ≥ 1000
- Central nervous system (CNS): Impaired consciousness
- Heart and other organ involvement
Expanded dengue
Additional manifestations of dengue virus infection may include liver failure, CNS involvement, myocardial dysfunction, acute kidney injury and other organ involvement. Neurological manifestations include encephalopathy and seizures, stroke, Guillain-Barre syndrome, transverse myelitis, mono and poly neuropathies (Solomon et al. 2000). Diagnosis is by serological testing, culture and detection by polymerase chain reaction (PCR) in cerebral spinal fluid. Cardiovascular manifestations include myocardial impairment, arrhythmias and fulminant myocarditis (Miranda et al. 2013). Haemophagocytic lymphohistiocytosis is described in association with dengue virus infection.
Pathogenesis
After the virus is introduced into the skin by an infected mosquito, viraemia is detected from 6 to 18 hours before onset of symptoms and ends as the fever results. Both innate and adaptive immune responses play a role in clearance of infection. Virus-specific T lymphocytes response and antibody-dependent enhancement of infection decide severity of the infection. Plasma leakage, haemoconcentration and abnormalities in homeostasis characterise severe dengue.
A transient and reversible imbalance of inflammatory mediators, cytokines and chemokines occurs during severe dengue, probably driven by a high early viral burden, and leading to dysfunction of vascular endothelial cells, derangement of the haemocoagulation system, then to plasma leakage, shock and bleeding.
Management
Step I − Overall assessment
- History, onset of symptoms, past medical and family history
- Physical examination, including full physical and mental assessment
- Investigation, including routine laboratory tests and dengue-specific laboratory tests
Step II − Diagnosis, differentials, assessment of disease phase and severity
Step III − Management
- Disease notification
- Management decisions: Depending on the clinical manifestations and other circumstances, patients may either be:
- Sent home (Group A) or
- Referred for in-hospital management (Group B) or
- Require emergency treatment and urgent referral (Group C)
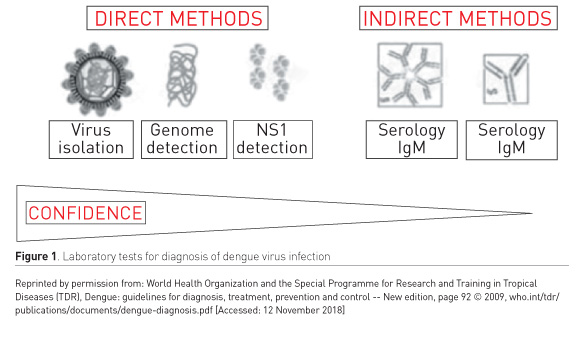
Laboratory tests
See Figure 1.
- Directly by detection of viral components in serum—high specificity but labour-intensive and costly
- Indirectly by serology—lower specificity but more accessible and less costly
1. Direct methods
During the first week of illness:
- Reverse transcription polymerase chain reaction (RT-PCR) assay for detection of viral nucleic acid
- Nonstructural protein 1 antigen test (NS1Ag): sensitivity 90% in primary infection and 60-80% in secondary infection
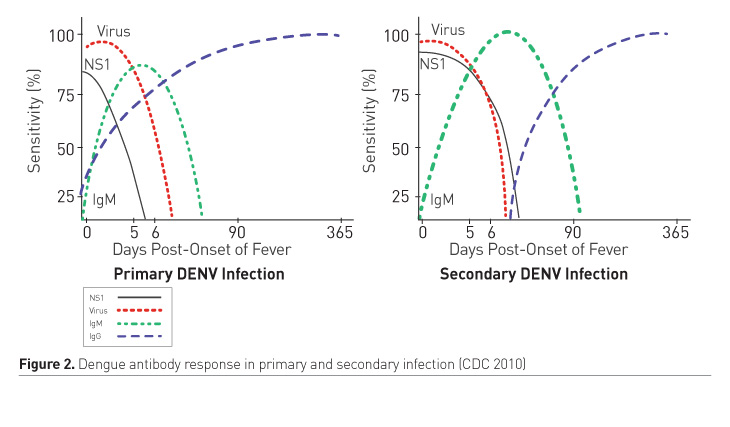
2. Indirect methods (Figure 2)
- Immunoglobin M antibody (IgM Ab): Detected as early as 4 days after onset of illness
- Immunoglobin G antibody (IgG Ab): In primary infection there is slow rise and low titres, detected after 7 days of onset of illness. In secondary infection, there is rapid rise in titres beginning 4 days after onset of illness.
The ratio of IgG: IgM >1:1.10 on day 2 of illness helps to diagnose secondary dengue infection (Changal et al. 2016).
3. Direct virus isolation through cultures with delayed results
4. Dengue viral proteins detected in tissue samples using immunohistochemical staining
Liver tissues have highest yield. This is rarely indicated.
Differential diagnoses
- Viral haemorrhagic fevers like Ebola, Lassa, Yellow fever, Hanta, Crimean congo, Severe fever with thrombocytopaenia syndrome virus (SFTSV)
- Chikungunya
- Malaria
- Typhoid fever
- Leptospirosis
- Rickettsial infection
- Zika virus
- Sepsis due to bacteraemia
Dengue case management
Treatment according to groups A to C:
Group A: Patients who may be sent home (outpatient management)
- Bed rest and frequent oral liquids
- Paracetamol (10mg/kg/dose) not more than 3-4 times/day in chidren and not more than 3g/day in adults. Tepid water sponging if high grade fever persists
- Patients with more than 3 days of illness need to be reviewed daily for disease progression (Look for leukopaenia, thrombocytopaenia, increasing Hct, defervescence and warning signs).
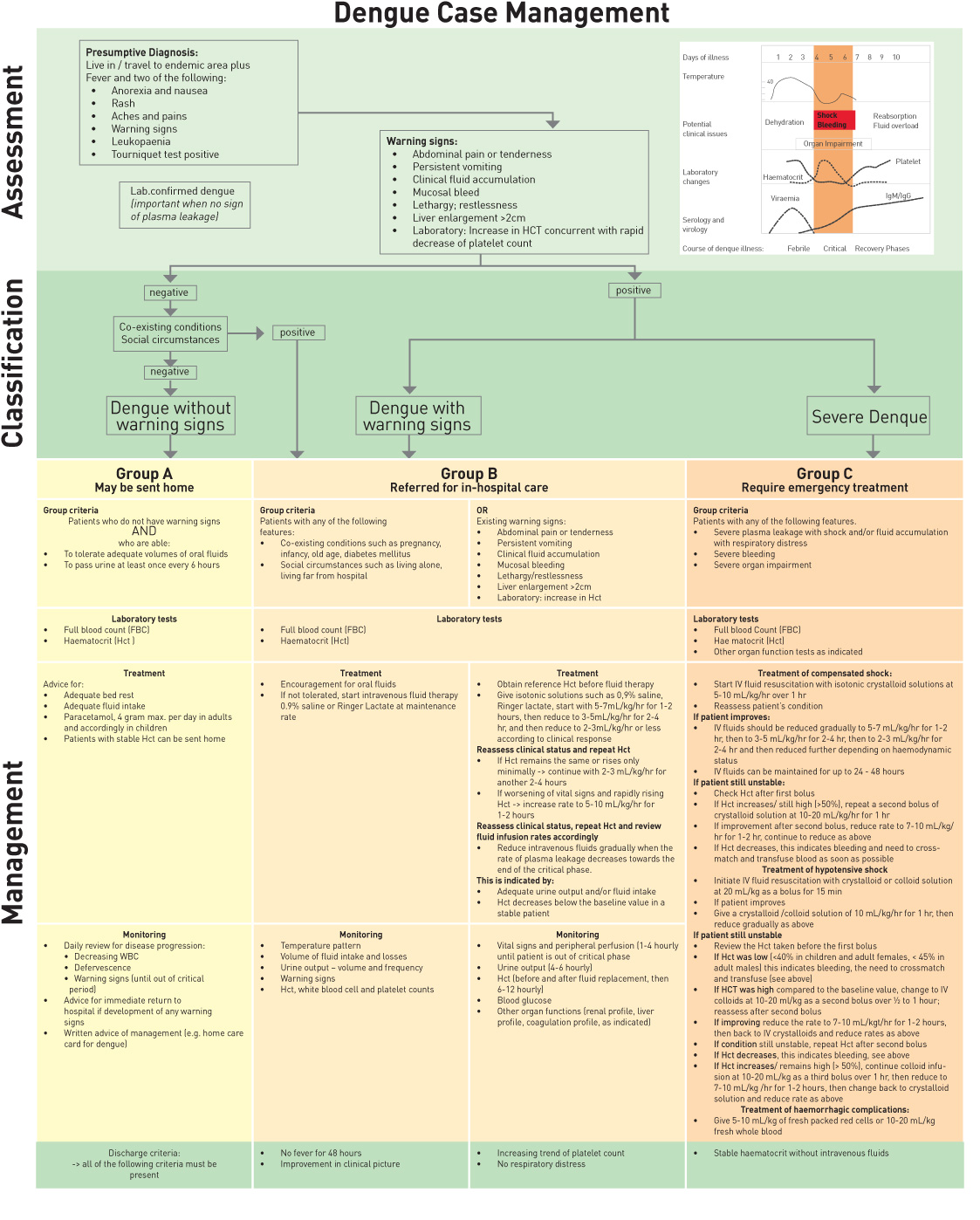
Group B: Patients with warning signs, co-existing conditions and those with social circumstances need inhospital management for close observation.
Warning signs: no clinical improvement, deterioration around the time of defervescence, abdominal pain, vomiting, cold peripheries, obtundation, bleeding, breathlessness, oligoanuria. Refer to Figure 3 (previous page) for fluid management of Group B patients.
Group C:
- Patients with severe plasma leakage causing shock, third spacing, respiratory distress
- Severe haemorrhages
- Severe organ impairment
Further divided into patients with compensated and uncompensated shock (Figures 4-5).
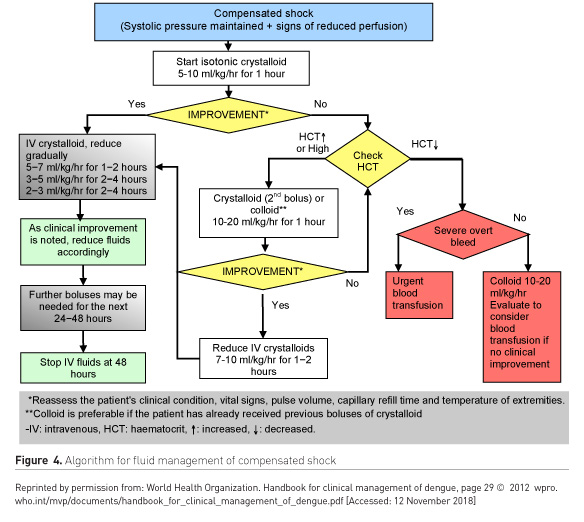
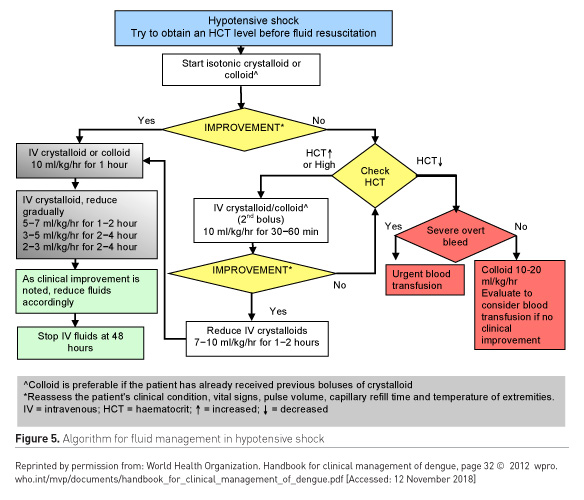
When to stop IV fluids
- Signs of cessation of plasma leakage
- Stable haemodynamics
- Decreasing haematocrit with stable haemodynamics
- Apyrexia for > 24-48 hours
- Resolving abdominal symptoms
- Improving urine output
Prophylactic transfusion of platelet concentrates in absence of bleeding, even if profound thrombocytopaenia and use of steroids, unless proven haemophagocytic syndrome, has not been shown to improve outcome (Dondorp 2016).
Treatment of haemorrhagic complications
- Strict bed rest and protection from trauma
- Avoid intramuscular injections
- Cautious insertion of urinary/nasogastric tubes/central line insertion under ultrasound guidance
- Stop the bleeding
- 5-10mL/kg of fresh packed red blood cells (PRBCs)
- Platelet transfusion (if <10,000 or active bleeding) and occasionally fresh frozen plasma (FFP).
- Vitamin K if international normalised ratio (INR) deranged or liver disease
- In gastrointestinal bleeds, H2 blockers/proton pump inhibitors (PPIs) but doubtful efficacy
Risk factors
- Patients with refractory shock
- Patients on non-steroidal anti-inflammatory agents, anticoagulant therapy
- Patients with pre-existing peptic ulcer disease
Glucose control
Both hypoglycaemia and hyperglycaemia can occur in the same patient during critical phases.
Electrolyte and acid base imbalances
- Hyponatraemia secondary to GI losses or use of hypotonic solutions
- Hypo- or hyperkalaemia in association with acute kidney injury or gastrointestinal losses or stress-induced hypercortisol state.
- Hypocalcaemia: after massive blood transfusions or use of sodium bicarbonate
- Hypophosphataemia
- Hyperuricaemia
- Lactic acidosis secondary to tissue hypoxia and hypoperfusion
- Hyperchloraemic metabolic acidosis due to administration of large volume of normal saline
Complications
- Prolonged shock
- Severe bleeding with disseminated intravascular coagulation
- Fluid overload
- Respiratory distress and failure
- Multiorgan failure (MOF)
- Abdominal compartment syndrome
- Irreversible shock and death
- Co-infections and nosocomial infection: Gram-negative sepsis, co-existing tropical diseases like malaria, leptospira, typhus,enteric fever, chikungunya
- Haemophagocytic syndrome: Manifested as persistent high-grade fever, cytopaenia and MOF associated with macrophage activation, haemophagocytosis and hypercytokinaemia with raised serum ferritin levels.
- Diagnosed by bone marrow biopsy demonstrating haemophagocytic activity
Treatment mainly with steroids (Methylprednisolone) and IV immunoglobulin
Dengue prevention
Prevention includes mosquito control, personal protective measures and vaccination.
- Mosquito control:
- Reducing breeding sites
- Larva control
- Use of insecticide
- Endosymbiotic control: a novel strategy consists of releasing mosquitoes infected with the intracellular endosymbiotic bacterium Wolbachia, thereby reducing mosquito lifespan and inhibiting viral replication.
- Personal protective measures: A combination of chemically-treated gear and clothing and a strong chemical repellent may be necessary in areas with high concentrations of disease-carrying arthropods.
- Vaccine: CYD-TDV is a formulation of four chimeric yellow fever 17D-dengue vaccine viruses. In April 2018 the World Health Organization (WHO) advised that it should be administered only to individuals with a history of previous dengue virus infection or laboratory evidence of previous dengue virus infection. It is administered in three doses at months 0, 6, and 12.
Conclusion
Dengue, as a mosquito-borne disease, can be prevented by effective mosquito control measures and vaccine development, which is under evaluation. Suspected dengue cases should be assessed carefully and directed to the appropriate care setting. Early recognition of severe dengue infection is essential as it needs prompt initiation of more aggressive therapy. Outpatient management is appropriate for patients without warning signs, with plenty of fluids and advice to watch for signs of dehydration, clinical deterioration or lack of improvement with defervescence or impending shock. Inpatient management is warranted for patients with dengue with warning signs, severe dengue infection or with coexisting conditions. They should be closely watched for signs of bleeding or worsening shock. Thus, high clinical suspicion, appropriate diagnostic measures and treatment at the right time is the key to achieve good outcomes in these patients and reduce morbidity as well as mortality.
References:
Centers for Disease Control and Prevention (CDC) (2010) Dengue: Laboratory guidance and diagnostic testing. [Accessed: 29 August 2018] Available from cdc.gov/dengue/clinicallab/laboratory.html
Changal KH, Raina AH, Raina A et al. (2016) Differentiating secondary from primary dengue using IgG to IgM ratio in early dengue: an observational hospital based clinico-serological study from North India. BMC Infectious Diseases, 16: 715.
Dondorp AM (2016). Other tropical diseases in the ICU. In: Webb A, Angus D, Finfer S et al., eds. (2016) Oxford textbook of critical care. 2nd edition, pp. 1404-7.
Guzman MG, Harris E (2015) Dengue. Lancet, 385: 453-65.
Kularatne SA (2015) Dengue fever. BMJ, 351: h4661.
Miranda CH, Borges Mde C, Matsuno AK et al. (2013) Evaluation of cardiac involvement during dengue viral infection. Clin Infect Dis, 57: 812-9.
Pai-Dhungat JV, Parikh F (2013) Medical philately. Dengue an escalating problem. J Assoc Physicians India, 61(8): 582.
Simmons CP, Farrar JJ, Nguyen vV et al. (2012) Dengue. New Engl J Med, 366: 1423-32.
Solomon T, Dung NM, Vaughn DW et al. (2000) Neurological manifestations of dengue infection. Lancet, 355:1053-9.
World Health Organization (1997) Dengue haemorrhagic fever: diagnosis, treatment, prevention and control. 2nd edition. Geneva: WHO. [Accessed: 29 August 2018] Available from who.int/csr/resources/publications/dengue/Denguepublication/en
World Health Organization (2012) Handbook for clinical management of dengue. Geneva: World Health Organization. [Accessed: 12 November 2018] Available from wpro.who.int/mvp/documents/handbook_for_clinical_management_of_dengue.pdf
World Health Organization (1997) Dengue haemorrhagic fever: diagnosis , treatment, prevention and control. 2nd Edition. Geneva: WHO. [Accessed: 29 August 2018] Available from who.int/csr/resources/publications/dengue/Denguepublication/en
World Health Organization and the Special Programme for Research and Training in Tropical Diseases (TDR) (2009) Dengue guidelines for diagnosis, treatment, prevention and control: new edition. [Accessed: 12 November 2018] Available from who.int/rpc/guidelines/9789241547871/en









