ICU Management & Practice, Volume 17 - Issue 1, 2017
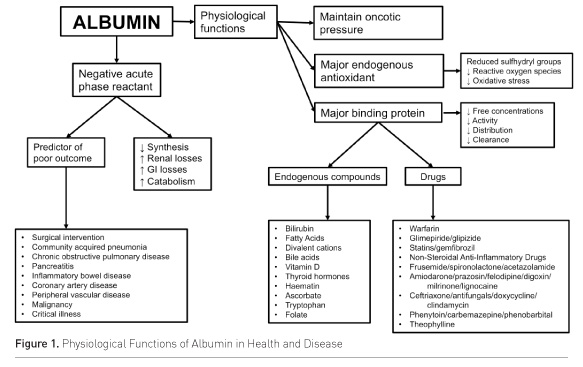
Serum albumin is an essential plasma protein, with a variety of homeostatic and predictive roles in health and disease (Figure 1). Hypoalbuminaemia is common in critical illness. Human albumin solution has been administered clinically for more than five decades, but its use has been subject to marked controversy for the last twenty years (Fanali et al. 2012). This has shaped not just day-to-day practice in the intensive care unit (ICU), but also the evolution of international, multicentre randomised controlled trials (RCTs) in critical care. The most recent data from the United States suggests that, at least in academic medical centres, albumin administration is increasing, particularly among surgical patients and those with higher illness severity scores (Suarez et al. 2017). In Australia and New Zealand, although overall artificial colloid use has recently fallen, sales of 4% and 20% albumin solutions have remained constant (Glassford et al. 2016; Hasmmon et al. 2015). However, significant regional and international variation in the acceptability of albumin for use as a resuscitation fluid has been demonstrated (Finfer et al. 2010; McIntyre et al. 2016; Glassford et al. 2016). Some centres have even instituted intervention programmes to reduce albumin administration (Lyu et al. 2016), as the financial implications of albumin use can be considerable. In Australia, for example, regulations regarding blood product processing and distribution facilitate the administration of albumin by clinicians. In other countries, such as the UK, they can make it prohibitively expensive.
Controversial Fluid or Controversial Analysis?
In 1998 a systematic review written by the Cochrane Injuries Group and published in the BMJ attempted to synthesise the extant literature on albumin administration in the critically ill (Cochrane Injuries Group Albumin Reviewers 1998). In 24 highly heterogeneous studies reporting mortality, in which a total of 1204 patients were randomised to receive albumin (or plasma protein fraction) or an alternative (no albumin or a crystalloid solution), albumin was shown to be associated with a significant increase in mortality (OR 1.68, 95% CI 1.26 to 2.23). In 14 of these studies, the patient population was surgical, in 9 the patients were included once diagnosed with hypoproteinaemia or hypoalbuminaemia, in 4 following trauma, and in 3 following burns. Sepsis was only definitively mentioned as a feature of the population of a single trial. On sub-group analysis, when albumin was given to mainly surgical or trauma patients for the correction of hypovolaemia, there was no statistically significant increase in mortality (OR 1.46, 95% CI 0.97 to 2.22). Moreover, on exclusion of the 11 trials at greatest risk of bias, the odds ratio for mortality following albumin administration to correct hypovolaemia fell (OR 1.39, 95% CI 0.8 to 2.4). The included trials were small, clinically heterogeneous, prone to bias, and many had been performed 10 to 20 years previously. The meta-analysis excluded those studies where patients were randomised but no deaths occurred. In addition, albumin was compared to a variety of different, or unrecorded fluid types. While acknowledging the limitations of their findings, the authors called for a review into the routine use of albumin and for a rigorous randomised, controlled examination of its efficacy (Cochrane Injuries Group Albumin Reviewers 1998).
This publication was met with a flurry of rapid responses and editorials (Workman 1999; Dearlove 1999; Offringa 1998a; Berger 1998; Offringa 1998b; Shwe and Bhavnani 1998; Chalmers 1998; Frame and Moiemem 1998; Goodman 1998; Beale et al. 1998; Soni 1998; Riordan et al. 1998; Nadal et al. 1998; Petros et al. 1998; Nel 1998; McClelland 1998; Lawler and Morgan 1998; Fogarty and Khan 1999; Kaag and Zoetmulder 1998), including harsh criticism of the study, and statements of support opposing further albumin use (Offringa 1998a), which were almost immediately “clarified” (Offringa 1998b). Mainstream media presented a picture of significant harm (BBC News 1998; Murray 1998; Mills 1998). One letter to the BMJ from an academic at the UK Cochrane Centre in Oxford, who claimed he would “sue anyone who gave me an albumin infusion” (Chalmers 1998), led to further incendiary media coverage (Boseley 2000). This debate may also have contributed to the subsequent widespread adoption of transparent declarations of conflicts of interest in any submissions to peer-reviewed journals, including letters and rapid responses (Dearlove 1998; Chalmers 1998; Smith 1998).
An international attempt to document the annual use of colloids in industrialised countries showed a significant reduction in the use of albumin between 1995 and 2006, with a concomitant increase in the use of synthetic colloids over the same period. However, the data was difficult to obtain, from fragmentary sources, and in many cases incomplete (Jones et al. 2010). An industry-sponsored report suggests a non-statistically significant 19% reduction in the volume of albumin supplied between 1998 and 2000, with an average of 5.4 million litre equivalents of 4% albumin being sold each year (Vincent et al. 2003). Although it is impossible to assign causation, a survey of British ICU directors indicated that the use of albumin in more than half of UK ICUs had been influenced by this systematic review (Brown et al. 2001)
Establishing a SAFE Starting Point
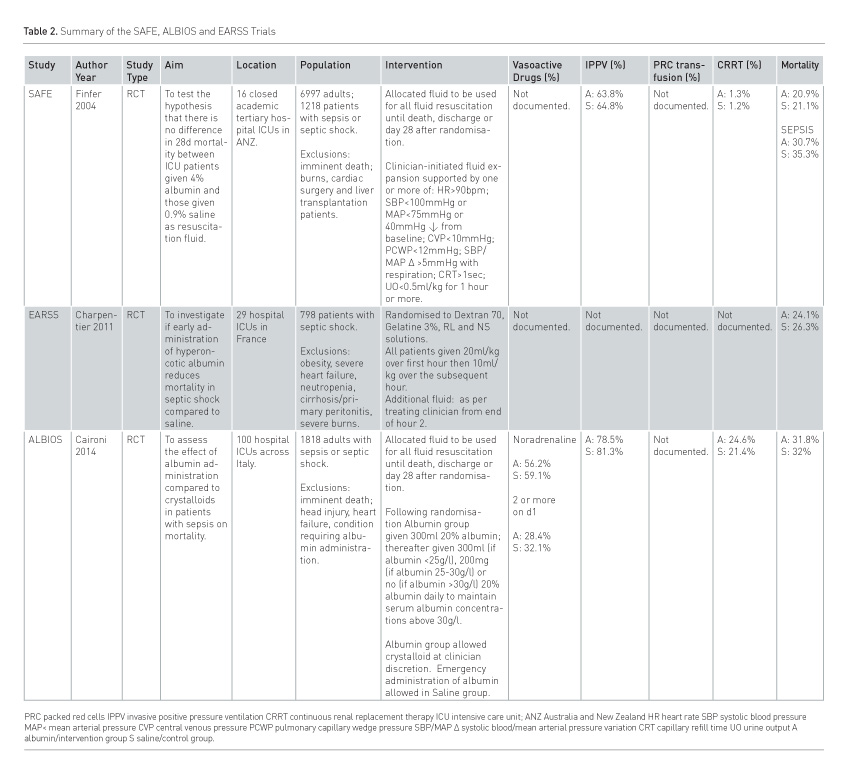
As a response to the continued uncertainty regarding the use of albumin, in 2004 the Australia and New Zealand Intensive Care Clinical Trials Group published the Saline versus Albumin Fluid Evaluation (SAFE) Study.
SAFE was the first high-level, randomised, double-blind controlled evidence in 6,997 patients from 16 ICUs that 4% albumin administration was, well, safe. No differences were reported in 28-day all-cause mortality, need for mechanical ventilation or renal replacement therapy, and length of hospital or ICU stay between those critically ill patients requiring intravascular volume expansion given saline and those given albumin (Finfer et al. 2004). However, the SAFE trial was neither designed nor powered to demonstrate superiority to saline in different groups of critically ill patients—merely that its use was safe in the heterogeneous population of the ICU. Thus, albumin may be the fluid of choice in certain groups of patients, or under certain circumstances contra-indicated. A non-statistically significantly increased risk of mortality with albumin administration in trauma patients, and a similarly non-statistically significant reduction in mortality in patients with sepsis were observed in the trial, and further analyses of these subgroups were made (Myburgh et al. 2007; Finfer et al. 2011).
Is Albumin SAFE in Sepsis?
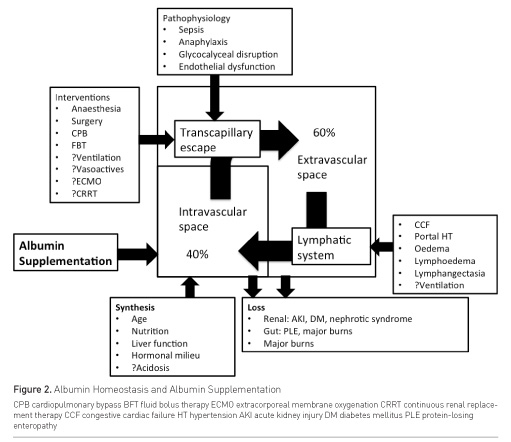
In septic patients, human albumin solution can be given for two broad indications—to restore or protect or expand intravascular volume, or to supplement serum albumin in an attempt to ameliorate the perceived deleterious effects of hypoalbuminaemia often associated with sepsis and/or critical illness. Although physiological reasoning suggests that albumin supplementation in the critically ill would be biologically logical, and the benefits of albumin use for fluid bolus therapy may be thought to be greatest among hypoalbuminaemic patients, the interaction between endogenous albumin concentrations and exogenous supplementation appears to be more complex (Figure 2).
In the 1,218 SAFE patients with sepsis there were no significant demographic differences between the saline and albumin groups at baseline. However, patients receiving albumin were administered significantly less study fluid with no differences in transfusion requirements, vasopressor use or need for mechanical ventilation over the first three days of the study, and significantly less fluid overall over the first two days (Finfer et al. 2011). On unadjusted estimate there was no difference in the risk of death between those patients randomised to albumin and those to saline (OR 0.87; 95% CI 0.74–1.02; p=0.09). When adjusted for potential baseline confounding and in the 75.5% of patients with sufficient information, albumin administration was independently associated with a reduction in the odds ratio for death at 28 days (OR 0.71, 95% CI 0.52–0.97, p=0.03) in a model accounting for illness severity, gender, age, postoperative admission, source of sepsis and serum albumin (Finfer et al. 2011).
Importance
of Age
The largest trial to date comparing the efficacy of albumin to saline resuscitation in sepsis was not performed in adults, but in critically ill children in sub-Saharan Africa (Maitland et al. 2011a). The Fluid Expansion as Supportive Therapy (FEAST) trial was a two-stratum, multicentre, open, randomised controlled study comparing the effects of albumin or saline resuscitation with maintenance therapy only on mortality in more than 3000 children with clinical evidence of impaired perfusion. Children without severe hypotension were randomised to receive 20ml/kg fluid bolus therapy (FBT) of 5% albumin, or 0.9% saline, or no bolus at all, with no invasive ventilation, renal replacement therapy or vasoactive medications available due to the resource-poor setting of the study. Approximately 20% of the study population was recruited following a protocol amendment increasing FBT volumes to 40ml/kg because of concern regarding under-resuscitation compared to international guidelines.
Of the 2097 children randomised to receive FBT, 1050 were assigned to albumin and 1047 to saline, with the groups being well balanced regarding baseline demographics, haemodynamic and clinical characteristics. There was no significant difference in the median volume of all fluid administered over the first, or second, or cumulatively by the end of the eighth hour from the start of the study (Table 1). With reported mortality rates of 10.6% vs 10.5% respectively at 48h (RR 1, 95% CI 0.78-1.29, p=0.96), and 12.2% vs 12% at 4 weeks (RR 1.01, 95% CI 0.8-1.28, p=0.91), and no difference in the incidence of pulmonary oedema, increased intracranial pressure or both at 48h (RR 1.17, 95% CI 0.68-2.03, p=0.49), or the incidence of neurological sequelae or death at 4 weeks (RR 1.04, 95% CI 0.84-1.28, p=0.71), there appears to be no statistically significant difference comparing albumin to saline FBT in this patient group.
However, the most provocative findings of the FEAST trial do not relate to the comparison of albumin with saline, but to the comparison of FBT with no resuscitation, and fully challenge the current paradigm of paediatric FBT-based fluid resuscitation. In this population, FBT with albumin or saline increased the absolute risk of death by 3.3% in children with suspected severe infection (RR 1.45, 95% CI 1.13 to 1.86; p = 0.003). Most deaths occurred within the first 24h, and the majority within 48h. This mortality difference persisted across all pre-specified, pathophysiologically logical sub-groups, with no heterogeneity between centres or across age groups (Maitland et al. 2011a; 2011b).
While it may be difficult to apply the findings of the FEAST trial directly to paediatric patients in ICUs in the developed world, it represents the purest examination of the effects of FBT in isolation in critically ill children to date, and suggests it may be harmful. Despite age-dependent differences in physiology (Gamble et al. 2000), it may be that continuing to compare albumin to other forms of resuscitation fluid in adults is conceptually wrong. Given that the peak haemodynamic effects of such FBT appear to be of limited clinical significance and duration (Aya et al. 2016; Bihari et al. 2013; Bihari et al. 2016; Glassford et al. 2014), perhaps future studies should focus instead on comparisons of any FBT in the critically ill with alternative interventions such as delayed FBT administration or early vasopressor therapy.
Resuscitation vs Supplementation: Does Intent Matter?
As opposed to purely using albumin as a resuscitation fluid, two large RCTs in adults have investigated the role of albumin supplementation and maintenance serum albumin concentration in sepsis (Table 2). The Albumin Italian Outcome in Sepsis (ALBIOS) study was a large multicentre, open-label, randomised, controlled trial designed to examine the effects of albumin supplementation in more than 1800 patients with sepsis or septic shock across 100 Italian ICUs (Caironi et al. 2014). Patients were randomised to either 20% albumin and crystalloid, or crystalloids alone. Those in the albumin group received 300ml of 20% albumin on randomisation, and subsequent infusions as required to maintain a serum albumin concentration >30g/l. No difference in mortality was observed between groups at either 28 days (RR 1.0; 95% CI 0.87 to 1.14; p=0.94) or 90 days (RR 0.94; 95% CI 0.85 to 1.05; p=0.29), although patients given albumin did have a shorter time to cessation of vasopressor agents (3, IQR:1 to 6 days vs 4, IQR:2 to 7 days; p=0.007).
In a post-hoc analysis of 1121 patients with septic shock, as defined by the Sequential Organ Failure Assessment (SOFA) score, there was a trend towards a reduction in 90-day mortality with albumin administration (RR 0.87; 95% CI 0.77 to 0.99; p=0.049). This persisted when corrected for baseline differences between groups, but not when corrected for what the investigators deemed clinically relevant variables, although the p-value for heterogeneity between patients with and without shock remained significant (Caironi et al. 2014).
The second study, a multicentre, randomised, controlled trial of Early Albumin Resuscitation during Septic Shock (the EARSS study) has not been published in its entirety and only an abstract is available (Charpentier and Mira 2011). This is a Stage 4 prospective, multicentre, randomised controlled trial comparing early albumin administration versus saline on 28-day survival in patients with septic shock (Early Albumin Resuscitation During Septic Shock, NCT00327704, clinicaltrials. gov/ct2/show/NCT00327704). Those randomised to the albumin group were to receive 100ml of 20% albumin every 8h for 72h. Initial findings were reported from 798 patients with septic shock recruited from 29 French centres. No significant difference in mortality was demonstrated between groups (24.1% in the albumin group and 26.3% in the saline group) (Charpentier and Mira 2011). Information regarding the patients in the EARSS trial is limited, and while systematic reviews must account for grey literature (Cook et al. 1993), in the absence of transparent methodology these data must be considered to be at high risk of bias. It is difficult to say how robust the findings of the EARSS study are, without them having been presented in their entirety, or subjected to peer review, but there seems to be little evidence for albumin supplementation improving mortality in sepsis considering these trials in isolation.
In SAFE and ALBIOS the interventions being investigated were quite different. In the SAFE trial, a heterogeneous group of critically ill patients was randomised to albumin or crystalloids for the purposes of volume expansion. In ALBIOS and EARSS patients with sepsis and septic shock were randomised to albumin or crystalloids for the purposes of maintaining serum albumin concentrations above an arbitrary level. All three studies examine albumin administration post-primary resuscitation—patients are enrolled either on admission to the ICU or 6-24h following the development of sepsis within the ICU. Events and exposures in the emergency department, or event in the prehospital setting, may confound the results of these studies. In established sepsis, where endothelial dysfunction and glycocalyceal disruption result in increased extravasation of albumin with subsequent tissue oedema, post-primary resuscitation with albumin may not be helpful (Kupr et al. 2007; Woodcock and Woodcock 2012; Margarson and Soni 2004).
A brief report in the New England Journal of Medicine suggested that there appeared to be a reduction in mortality among patients receiving albumin that was of borderline statistical significance (RR 0.92; 95% CI 0.84 to 1.00; p=0.046), when the results of EARSS, ALBIOS and SAFE are considered together (Wiedermann and Joannidis 2014). However, it fails to account for this methodological heterogeneity in its pooling of their results. A formal systematic review of 16 trials of human albumin use in adults with sepsis using traditional meta-analytic methodology with the addition of Trial Sequential Analysis (TSA) found no difference in the relative risk of death between albumin and control groups, with no evidence of statistical heterogeneity (RR 0.94; 95% CI 0.87 to 1.01; p=0.11, I2=0%) (Patel et al. 2014). TSA is similar to the sequential interim analysis employed in large phase II clinical studies to account for the increasing risk of type I error with repeated hypothesis testing (Todd et al. 2001) but applied to the repeated testing of significance with the addition of each trial to a metaanalysis (Wettersley et al. 2008). Trials were included if they compared the administration of albumin to a control fluid and presented all-cause mortality data. Published criticisms of this meta-analysis centre on the inclusion of the figures for 90-day mortality from the EARSS trial instead of the 28-day mortality presented in abstract form (Wiedermann 2014a), the inclusion of trials by a group demonstrating a consistent pattern of fraudulent research (Shafer and Wilkes 2014), and the possibility of the inclusion of the same patients from multiple studies (Wiedermann 2014b). However, the meta-analysis appears robust, with multiple sensitivity and sub-group analyses, clearly presented methodology and extensive meta-regression that aim to account for these features (Patel et al. 2014b; 2014c).
Baseline Values: Does Endogenous Serum Albumin Concentration Matter?
In the SAFE trial, no difference was found in mortality between resuscitation with albumin or saline in patients with serum albumin concentrations above or below 25g/l, nor was serum albumin concentration found to interact significantly with the effect of saline or albumin on mortality when considered as a continuous variable (Finfer et al. 2006). Hypoalbuminaemic patients were older, more likely to have undergone surgery, have acute respiratory distress syndrome or sepsis, and less likely to have had traumatic brain injury, though illness severity scoring was similar. The unadjusted ratio of odds ratios between treatment groups when comparing patients with a baseline serum albumin concentration <25g/l and those with >25g/l was 0.80 (95% CI 0.63 to 1.02), with the ratio of odds ratios falling to 0.73 (95% CI 0.55 to 0.97) after adjustment for baseline risk factors for death. However, serum albumin concentration as a continuous variable demonstrated no significant interaction with treatment allocation for 28-day mortality on multivariate analysis. Despite the suggestion of benefit on appropriate adjustment in the binary albumin concentration group, the authors felt that overall this subgroup analysis neither provided significant evidence of a difference in treatment effect of albumin compared to saline resuscitation, irrespective of baseline serum albumin concentration, nor did it suggest that hypoalbuminaemic patients were at an increased risk of death.
While separation in serum albumin concentration was achieved between groups in the ALBIOS study, it aimed to maintain serum albumin concentrations of 30g/l or more throughout admission in the intervention group; this was not achieved until after day 8 (Caironi et al. 2014). Both groups presented with median serum albumin concentrations of 24g/l, though with interquartile ranges suggesting that similar analyses to those performed in the SAFE cohort could have been performed. Baseline albumin concentration is not used to adjust outcomes, nor is it reported as a sensitivity analysis. No post-hoc analyses of ALBIOS study data have been published to date. Unfortunately, no information is available regarding the disposition of the patients in the EARSS cohort as regards baseline serum albumin concentration.
See Also links: Fluid Choices in Trauma
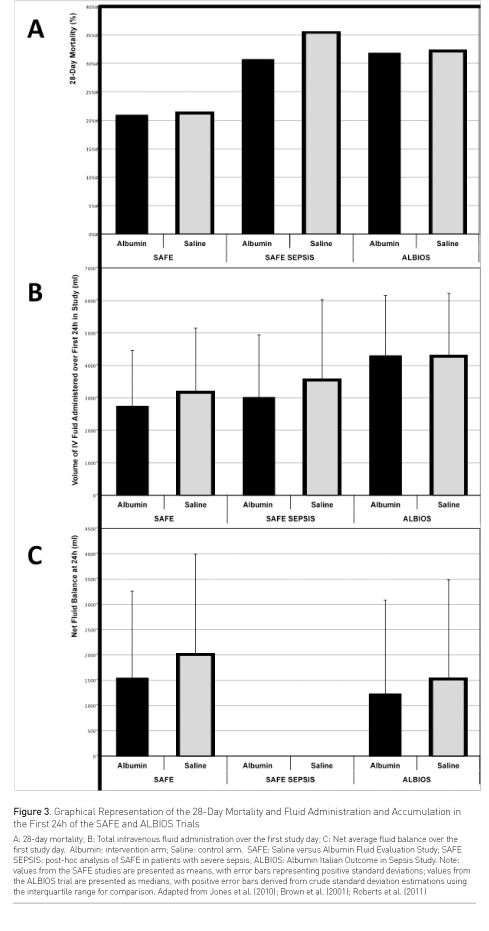
Given the established and increasing concerns regarding fluid accumulation and poor outcomes in a variety of critically ill populations, including those with sepsis (Bouchard et al. 2009; Grams et al. 2011; National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network 2006; Payen et al. 2008; Rosenberg et al. 2009; Toraman et al. 2004), the volume of product administered, and the volume of fluids co-administered must be considered as potential confounders when assessing the effects of albumin administration or resuscitation on mortality (Bagshaw and Bellomo 2007). Indeed, the potential to limit the volume of fluid resuscitation is one of the attractive features of albumin resuscitation, and colloid use in general. Standard teaching that 3 times the volume of crystalloid is required to achieve the same effect as for a given volume of colloid has been shown to be incorrect, with the SAFE trial suggesting that, over the first 4 days of the study, 1:1.4 times the amount of albumin to saline was administered. Those in the albumin group of the SAFE trial received approximately 2700ml of intravenous fluid over their first 24 hours in ICU, while those in the saline group received approximately 3100ml. Net mean positive fluid balances at 24h were approximately 1540ml and 1990ml respectively (Finfer et al. 2004). In the 1,218 patients with sepsis, net fluid balance was not reported, but less fluid was given over the first 24h and 48h of the study in the albumin group (603 patients) than the saline group (615 patients) (Figure 3) (Finfer et al. 2011).
In the ALBIOS study, both groups received approximately 4300ml of intravenous fluid over their first 24 hours in ICU, with 20% albumin only accounting for approximately 7% of the total fluid administered in the albumin group. Patients in the albumin group reported a net median fluid balance of 1229ml at 24h and 350ml at 48h, while those in the crystalloid group received approximately 4250ml of fluid over the first 24h and had net median positive fluid balances of 1504ml at 24h and 620ml at 48h (Figure 3) (Caironi et al. 2014).
Compared to those in the SAFE trial, the patients in the ALBIOS study were older, had higher illness severity scores, and were more likely to be ventilated, to have received prerandomisation colloid, and to die within 28 days. These differences may partially explain the differences in volume status. While not significant, lower mortality rates were reported in the intervention (albumin) groups, as were lower fluid balances and/or lower volumes of fluid administered (Figure 3). In neither study were mortality results adjusted for measures of volume status.
Of the four most recent systematic reviews of albumin administration in sepsis, none account for total volume of fluid administered or fluid accumulation as potential confounding covariates (Patel et al. 2014a; Jiang et al. 2014; Rochwerg et al. 2014; Xu et al. 2014). Meta-regression techniques allow the effect of potentially confounding variables on effect sizes to be explored, much as logistic or linear regression does at a trial level (Baker et al. 2009). While several studies examined dose, strength, or concentration of albumin in sensitivity analyses (Jiang et al. 2014; Rochwerg et al. 2014; Xu et al. 2014), one meta-analysis performed extensive meta-regression with a variety of covariates, including volume of albumin administered (Patel et al. 2014a). However, more global measures of fluid administration and accumulation remain unaccounted for.
In a systematic review of trials comparing colloids with crystalloids given for the purposes of resuscitation designed to evaluate the reported crystalloid: colloid ratio, a volume ratio of 1.5 (95% CI 1.36 to 1.65) was identified on meta-analysis of 36 cohorts across 24 trials (Orbegozo Cortes et al. 2015). On meta-regression, the volume ratio significantly decreased each decade from the 1990s, and significantly increased with increasing concentration of albumin solution, in the relevant sub-group analysis. While SAFE was included, the majority of the reported trials are small, clinically and methodologically heterogeneous, and have been reported over a span of four decades. No account was made for the co-administration of crystalloid in the colloid groups, though this degree of detail is often very hard to obtain from papers more than 35 years old. This interesting study demonstrates the facility with which meta-regression can be used to parse the complexities of data synthesis, but then fails to address the topic of mortality. An alternative approach to assessing the impact of volume status on the relationship between albumin administration and mortality in patients with sepsis may be individual patient data meta-analysis. However, obtaining patient-level data from multiple research groups, from studies performed over decades, in a contentious area of research, would be challenging.
Carrier Fluid and Concurrent Administration: Is It a Matter of Co-Exposure?
The effects of albumin, irrespective of the reason for administration, on patient outcome are further confounded by the concurrent administration of the sodium- and chloriderich carrier fluid of dilute solutions, making comparisons not between different fluids but between essentially similar fluids and additional colloid. However, the sodium and chloride content of various albumin solutions varies with albumin concentration and country of origin, as does the inclusion of other organic compounds such as octanoate (Kaplan and Kellum 2010). In the ALBIOS and EARSS studies patients received a concentrated solution of 20% albumin (Caironi et al. 2014; Charpentier and Mira 2011), while patients in the SAFE study received 4% albumin (Finfer et al. 2004). The constituents and tonicity are not presented in either the papers or any supplemental material. In a nested cohort study of more than 600 patients from three ICUs involved in the SAFE trial, the volume rather than type of fluid administered was a much stronger predictor of the acid-base and biochemical changes resulting from resuscitation with albumin or saline (Bellomo et al. 2006). It may be that the risks associated with excess sodium (Bihari et al. 2010) and chloride (Yunos et al. 2014; Yunos et al. 2012) administration obscure any benefits of albumin delivery. One potential alternative would be to administer concentrated salt-poor albumin solution as a form of low-volume haemodynamic resuscitation. In a retrospective observational study of 202 patients in an Australian tertiary intensive care unit, 100ml of 20% albumin solution delivered the same haemodynamic improvement as 500ml of 4% albumin solution, but in a volume-, chloride- and sodium-sparing manner (Bannard-Smith et al. 2015).
Concurrent administration of other fluids is potentially an important consideration in any study involving a head-to-head comparison of fluids. Other debates rage within the fluid space: the emerging preference for balanced crystalloid solutions over non-physiological “normal” saline, for example (Glassford et al. 2016). However, the signal for harm observed in critically ill patients with sepsis treated with hydroxyethyl starches (HES) across multiple studies is of particular interest, given the process of care observed in the ALBIOS study (Caironi et al. 2014). Overall, 31.9% of patients in the albumin group were exposed to HES in the 24h prior to randomisation, and 17.5% were exposed to a median of 750ml over at least 24h during the study. Moreover, 33.6% of patients in the saline group were exposed to HES in the 24h prior to randomisation, and 17% were exposed to a median of 1000ml over at least 24h during the study. 90-day mortality (Albumin: 47.8% vs 39.7%; Saline: 52.6% vs 41.7%) and the incidence of acute kidney injury (Albumin: 27.5% vs 20.7%; Saline: 27.7% vs 21.6%) were higher in both treatment groups in those exposed to HES during the study. A proposed individual patient comparative analysis of data from the SAFE and CHEST (Crystalloid v Hydroxyethyl Starch Trial) studies is forthcoming, and may offer some insights into the impact of starch exposure in the setting of albumin administration (Hammond et al. 2014), but given these results, post-hoc analyses of the ALBIOS data would provide additional relevant data.
Alternative Approaches to Analysing the Evidence
Bayesian frameworks offer an alternative method by which to analyse the relative comparative effectiveness of albumin as a resuscitation fluid in sepsis.
Network meta-analysis is a non-frequentist method of comparing multiple treatments directly within and indirectly across RCTs. The process is similar to bootstrapping, with a number of iterations being modelled. Techniques to minimise the effect of initial values on the posterior inference and reduce sample autocorrelation are applied (Hamra et al. 2013). In a recent network meta-analysis of fluid resuscitation in 19,000 septic patients across 14 studies with 15 direct comparisons, a lower mortality was associated with albumin use than with crystalloid or starch use in a 4-node model, and with albumin compared to saline in a 6-node model with moderate confidence in all albumin estimates (Rochwerg et al. 2013). Beyond the difficulty of interpreting these results in a frequentist paradigm, bias is of even more significance in network studies than in conventional meta-analyses as it affects not only direct comparisons, but also any indirect comparison made. Clinical, statistical and methodological heterogeneity, and inconsistency, or a discrepancy between direct and indirect comparisons, may affect different regions of a network to a greater or lesser extent (Li et al. 2011). More research is required before the conclusions of network meta-analysis form the basis of changes in clinical practice, but they offer an exciting alternative to traditional models of evidence integration.
Conclusions
Although albumin has been used clinically for more than seventy years, and more than a decade has passed since the publication of high-level evidence of its safety, its administration remains controversial in the critically ill. Randomised, controlled trials demonstrate no benefit from the routine correction of hypoalbuminaemia in this population. It is unlikely that subsequent large frequentist RCTs of albumin-based FBT in specific patient populations will be performed, given the difficulty in identifying such patients, the number of patients to be recruited to power such a study, and the prohibitive cost of providing and packaging solutions and placebos on such a scale. In Australia, where albumin is provided to hospitals as blood product at no cost by the Australian Red Cross Blood Services, a possible solution would be to employ a Bayesian platform trial (Berry et al. 2015). With this methodology it may be possible to compare different concentrations of albumin, in different sub-groups of patients, with different colloid and crystalloid solutions. As with network meta-analysis, this is a novel and attractive approach to trial design, but it is likely to be several years before the suitability of such a methodology can be assessed, with the first such trial only just receiving funding (Monash University School of Public Health and Preventive Medicine 2015).
Outside of those populations in which its use is contraindicated, such as TBI or burns, the current evidence base demonstrates that the administration of albumin as resuscitation fluid to critically ill patients is safe, and may be beneficial in patients with severe sepsis. If any benefit is to be seen with albumin use, it will be in subgroups of critically ill patients, not in the undifferentiated population of the ICU, and use will be dependent on regional and local guidelines, economic concerns and clinician preference. At present, clinical judgement and physiological reasoning, rather than strength of evidence, remain the primary drivers for the administration of albumin in the critically ill.
Acknowledgements
The authors would like to thank Dr. Craig J.
French, Dr. Maurice Le Guen, Dr. Stephen Thornhill, and Dr. Mark Kubicki for
their advice with the preparation of the final manuscript.
Financial Disclosures
RB has received unrestricted research grants from Baxter Pty Ltd.
Funding/Support
The Avant Doctors-In-Training Research Scholarship supports Dr Glassford. This study was supported by an unrestricted educational grant from the Austin Hospital Anaesthesia and Intensive Care Trust Fund.
Role
of the Sponsor
Avant and the Austin Hospital Anaesthesia and Intensive Care Trust Fund played no further role in the preparation of this manuscript, nor had any role in the design and conduct of the study; collection, management, analysis, or interpretation of the data beyond that described; nor any role in preparation, review, or approval of the manuscript.
Competing interests
The Avant Doctors-In-Training Scholarship funds
NJG’s research salary. RB has received unrestricted research grants from Baxter
Pty Ltd.
References:
Afinogenova Y, Tapper EB (2015) The efficacy and safety profile of albumin administration for patients with cirrhosis at high risk of hepatorenal syndrome is dose dependent. Gastroenterol Rep, 3(3): 216-21.
Alderson P, Bunn F, Lefebvre C et al. (2004) Human albumin solution for resuscitation and volume expansion in critically ill patients. Cochrane Database Syst Rev, (4):CD001208.
ANZIC-RC wins $4.4 million for ground-breaking pneumonia trial. In: School of Public Health and Preventative Medicine Blog, 21 December 2016..
Aya HD, Ster IC, Fletcher N et al. (2016) Pharmacodynamic analysis of a fluid challenge. Crit Care Med, 44(5): 880-91.
Bagshaw SM, Bellomo R (2007) The influence of volume management on outcome.
Curr Opin Criti Care, 13(5): 541-8.
Baker WL, White CM, Cappelleri JC et al.; Health Outcomes, Policy, and Economics (HOPE) Collaborative Group (2009) Understanding heterogeneity in meta-analysis: the role of meta-regression. Int J Clin Pract, 63(10): 1426-34.
Bannard-Smith J, Alexander P, Glassford N et al. (2015) Haemodynamic and biochemical responses to fluid bolus therapy with human albumin solution, 4% versus 20%, in critically ill adults. Crit Care Resusc, 17(2): 122-8.
Bartels K, Thiele RH, Gan TJ (2013) Rational fluid management in today's ICU practice. Crit Care, 17 Suppl 1: S6.
BBC News (1998) Blood product may have 'killed thousands', 23 July.
Beale RJ, Wyncoll DL, McLuckie A (1998)
Human albumin administration in critically ill patients. Analysis is
superficial and conclusions exaggerated. BMJ, 317(7162):884. PubMed ![]()
Bellomo R, Morimatsu H, French C et al.; Safe Study Investigators (2006) The effects of saline or albumin resuscitation on acid-base status and serum electrolytes. Crit Care Med, 34(12): 2891-7.
Berger A (1998) Why albumin may not work. BMJ, 317(7153): 240.
Berry SM, Connor JT, Lewis RJ (2015) The platform trial: an efficient strategy for evaluating multiple treatments. JAMA, 313(16): 1619-20.
Bihari S, Peake SL, Prakash S et al. (2015) Sodium balance, not fluid balance, is associated with respiratory dysfunction in mechanically ventilated patients: a prospective, multicentre study. Criti Care Resusc, 17(1): 23-8.
Bihari S, Prakash S, Bersten AD (2013) Post resusicitation fluid boluses in severe sepsis or septic shock: prevalence and efficacy (price study). Shock, 40(1): 28-34.
Bihari S, Teubner DJ, Prakash S et al. (2016) Fluid bolus therapy in emergency department patients: indications and physiological changes. Emerg Med Australas, 28(5): 531-7.
Boseley S (2000) Blood product promoted despite reports of patient deaths. The Guardian, 25 February.
Bouchard J, Soroko SB, Chertow GM et al.; Program to Improve Care in Acute Renal Disease (PICARD) Study Group (2009) Fluid accumulation, survival and recovery of kidney function in critically ill patients with acute kidney injury. Kidney Int, 76(4): 422-7.
Brown JM, McCormick BA, Vella K et al. (2001) Use of albumin in intensive care units in the United kingdom. Crit Care Resusc, 3(1): 19-21.
Caironi P, Tognoni G, Masson S et al. (2014) Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med, 370(15): 1412-21.
Chalmers I (1998) What is going on behind the scenes in the debate about albumin? BMJ, 317(7162): 235.
Charpentier J, Mira J (2011) Efficacy and tolerance of hyperoncotic albumin administration in septic shock patients: the EARSS study [abstract]. Intensive care medicine, 37(Supplement 2): S115-438.
Cochrane Injuries Group Albumin Reviewers (1998) Human albumin administration in critically ill patients: systematic review of randomised controlled trials. BMJ, 317(7153): 235-40.
Cook DJ, Guyatt GH, Ryan G et al. (1993)
Should unpublished data be included in meta-analyses? Current convictions and
controversies. JAMA, 269(21): 2749-53. PubMed ![]()
Dearlove O (1999) Beyond conflict of
interest. What is truth as it relates to albumin? BMJ, 318(7181) :464. Article ![]()
Early albumin resuscitation during septic shock
Eljaiek R, Heylbroeck C, Dubois MJ (2016) Albumin administration for fluid resuscitation in burn patients: a systematic review and meta-analysis. Burns.
Fanali G, di Masi A, Trezza V et al. (2012) Human serum albumin: from bench to bedside. Mol Aspects Med, 33(3): 209-90.
Finfer S, Bellomo R, Boyce N et al.; SAFE Study Investigators (2004) A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med, 350(22): 2247-56.
Finfer S, Bellomo R, McEvoy S et al. (2006) Effect of baseline serum albumin concentration on outcome of resuscitation with albumin or saline in patients in intensive care units: analysis of data from the saline versus albumin fluid evaluation (SAFE) study. BMJ, 333(7577): 1044.
Finfer S, Liu B, Taylor C et al.; SAFE Trips Investigators (2010) Resuscitation fluid use in critically ill adults: an international cross-sectional study in 391 intensive care units. Critical Care, 14(5): R185.
Finfer S, McEvoy S, Bellomo R et al. (2011) Impact of albumin compared to saline on organ function and mortality of patients with severe sepsis. Intensive Care Med, 37(1): 86-96.
Fogarty BJ, Khan K (1999) More on albumin. Multicentre randomised controlled trail is needed before changing resuscitation formulas for major burns. BMJ, 318(7192): 1215.
Frame JD, Moiemem N (1998) Human albumin administration in critically ill patients. Statisticians not trained in burns care should not evaluate data. BMJ, 317(7162): 884-5.
Gamble J, Bethell D, Day NP et al. (2000) Age-related changes in microvascular permeability: a significant factor in the susceptibility of children to shock? Clin Sci (Lond) 2000, 98(2): 211-6.
Glassford N, Eastwood G, Bellomo R (2014)
Physiological changes after fluid bolus therapy in sepsis: a systematic review
of contemporary data. Crit Care, 18(6): 696. PubMed ![]()
Glassford NJ, French CJ, Bailey M et al. (2016) Changes in intravenous fluid use patterns in Australia and New Zealand: evidence of research translating into practice. Crit Care Resus, 18(2): 78-88.
Glassford NJ, Mårtensson J, Eastwood GM et al.; GLobal OBservational Evaluations in the ICU (GLOBE-ICU) investigators (2016) Defining the characteristics and expectations of fluid bolus therapy: a worldwide perspective. J Crit Care, 35: 126-32.
Goodman NW (1998) Human albumin administration in critically ill patients. Paper failed to mention earlier review. BMJ, 317(7162): 884.
Grams ME, Estrella MM, Coresh J et al.; National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome Clinical Trials Network (2011) Fluid balance, diuretic use, and mortality in acute kidney injury. Clin J Am Soc Nephrol, 6(5): 966-73.
Hammond NE, Billot L, Finfer S et al. (2014) Individual patient data comparative analysis of hydroxyethyl starch 130/0.4 v 4% albumin for fluid resuscitation in critically ill patients: statistical analysis plan. Critical Care Resusc, 16(3): 206-13.
Hammond NE, Taylor C, Saxena M et al. (2015) Resuscitation fluid use in Australian and New Zealand Intensive Care Units between 2007 and 2013. Intensive Care Med, 41(9):1611-9.
Hamra G, MacLehose R, Richardson D (2013) Markov chain Monte Carlo: an introduction for epidemiologists. Int J Epidemiol, 42(2): 627-34.
Jiang L, Jiang S, Zhang M et al. (2014) Albumin versus other fluids for fluid resuscitation in patients with sepsis: a meta-analysis. PLoS One, 9(12): e114666.
Jones D, McEvoy S, Merz TM et al. (2010) International albumin use: 1995 to 2006. Anaesth Intensive Care, 38(2): 266-73.
Kaag M, Zoetmulder FA (1998) Human albumin
administration in critically ill patients. More research into proper use of
albumin is needed. BMJ, 317(7162): 883. PubMed ![]()
Kaplan LJ, Kellum JA (2010) Fluids, pH, ions and electrolytes. Curr Opin Crit Care, 16(4): 323-31.
Kuper M, Gunning MP, Halder S et al. (2007)
The short-term effect of hyperoncotic albumin, given alone or with furosemide,
on oxygenation in sepsis-induced acute respiratory distress syndrome.
Anaesthesia, 62(3): 259-63. PubMed ![]()
Kwok CS, Krupa L, Mahtani A et al. (2013) Albumin reduces paracentesis-induced circulatory dysfunction and reduces death and renal impairment among patients with cirrhosis and infection: a systematic review and meta-analysis. Biomed Res Int, 2013: 295153.
Lawler PG, Morgan GA (1998) Human albumin administration in critically ill patients. Modified editorial might have restrained media response. BMJ, 317(7162):885.
Li T, Puhan MA, Vedula SS et al.; Ad Hoc Network Meta-analysis Methods Meeting Working Group (2011) Network meta-analysis-highly attractive but more methodological research is needed. BMC Med, 9: 79.
Lyu PF, Hockenberry JM, Gaydos LM et al. (2016) Impact of a sequential intervention on albumin utilization in critical care. Crit Care Med, 44(7):1307-13.
Maitland K, Akech SO, Russell EC (2011) Mortality after fluid bolus in African children with severe infection: the authors reply. N Engl J Med, 365(14): 1351-3.
Maitland K, Kiguli S, Opoka RO et al. (2011) Mortality after fluid bolus in African children with severe infection. N Engl J Med, 364(26): 2483-95.
Margarson MP, Soni NC (2004) Changes in serum albumin concentration and volume expanding effects following a bolus of albumin 20% in septic patients. Br J Anaesth, 92(6): 821-6.
McClelland B (1998) Albumin: don't confuse us with the facts. Rather than fulminating, seek to answer the questions raised. BMJ, 317(7162): 829-30.
McIntyre L, Rowe BH, Walsh TS et al (2016) Multicountry survey of emergency and critical care medicine physicians' fluid resuscitation practices for adult patients with early septic shock. BMJ Open, 6(7) :e010041.
Mills H (1998) 300 die as health chiefs dither. Observer (London), 26 July.
Murray I (1998) Burns treatment 'has killed 30,000', The Times (London)
Myburgh J, Cooper DJ, Finfer S et al. (2007) Saline or albumin for fluid resuscitation in patients with traumatic brain injury. N Engl J Med, 357(9): 874-84.
Nadel S, Marriage S, De Munter C et al. (1998) Human albumin administration in critically ill patients. Review did not provide recommendations for alternative treatment. BMJ, 317(7162): 882-3.
National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome Clinical Trials Network, Wiedemann HP, Wheeler AP et al. (2006) Comparison of two fluid-management strategies in acute lung injury. N Engl J Med, 354(24): 2564-75.
Navickis RJ, Greenhalgh DG, Wilkes MM
(2016) Albumin in burn shock resuscitation: a meta-analysis of controlled
clinical studies. J Burn Care Res, 37(3): e268-78. PubMed ![]()
Nel MR (1998) Human albumin administration in critically ill patients. Critical analysis of original studies has to take place. BMJ, 317(7162): 882.
Offringa M (1998a) Excess mortality after human albumin administration in critically ill patients. Clinical and pathophysiological evidence suggests albumin is harmful. BMJ, 317(7153): 223-4.
Offringa M (1998b) Consider validity, clinical relevance, and applicability of albumin for critically ill patients. BMJ, 317(7154): 343.
Orbegozo Cortés D, Gamarano Barros T, Njimi H et al. (2015) Crystalloids versus colloids: exploring differences in fluid requirements by systematic review and meta-regression. Anesth Analg, 120(2): 389-402.
Patel A, Laffan MA, Waheed U, Brett SJ (2014a) Randomised trials of human albumin for adults with sepsis: systematic review and meta-analysis with trial sequential analysis of all-cause mortality. BMJ, 349: g4561.
Patel A, Laffan MA, Waheed U et al. (2014b) Exclusion of Boldt et al Articles does not alter the conclusion of no survival benefit with albumin. BMJ, 349: g4561.
Patel A, Laffan MA, Waheed U et al. (2014c) Exclusion of Haupt et al 1982 and or Rackow et al 1983 does not alter the conclusion of no survival benefit with albumin. Re: Randomised trials of human albumin for adults with sepsis: systematic review and meta-analysis with trial sequential analysis of all-cause mortality. BMJ, 349: g4561.
Payen D, de Pont AC, Sakr Y et al.; Sepsis
Occurrence in Acutely Ill Patients (SOAP) Investigators (2008) A positive fluid
balance is associated with a worse outcome in patients with acute renal
failure. Crit Care, 12(3): R74. PubMed ![]()
Petros A, Schindler M, Pierce C et al.
(1998) Human albumin administration in critically ill patients. Evidence needs
to be shown in paediatrics. BMJ, 317(7162): 882. PubMed ![]()
Riordan FA, Williams A, Thomson AP (1998) Human albumin administration in critically ill patients. Albumin has been used in meningococcal disease. BMJ, 317(7162): 883.
Roberts I, Blackhall K, Alderson P et al. (2011) Human albumin solution for resuscitation and volume expansion in critically ill patients. Cochrane Database Syst Rev, (11): CD001208.
Rochwerg B, Alhazzani W, Sindi A et al. (2014) Fluid resuscitation in sepsis: a systematic review and network meta-analysis. Ann Intern Med, 161(5): 347-55.
Rosenberg AL, Dechert RE, Park PK et al. (2009) Review of a large clinical series: association of cumulative fluid balance on outcome in acute lung injury: a retrospective review of the ARDSnet tidal volume study cohort. J Intensive Care Medicine, 24(1): 35-46.
Shafer SL, Wilkes MM (2014) Fraudulent data should be excluded from meta-analyses. BMJ, 349: g4561.
Shafer SL, Wilkes MM (2014) Re: Randomised trials of human albumin for adults with sepsis: systematic review and meta-analysis with trial sequential analysis of all-cause mortality. BMJ 2014;349 :g4561.
Shwe KH, Bhavnani M (1998) Human albumin administration in critically ill patients. Some patients may benefit. BMJ, 317(7162): 885-6.
Smith R (1998) Beyond conflict of interest. Transparency is the key. BMJ, 317(7154): 291-2.
Soni N (1998) Human albumin administration in critically ill patients. Validity of review methods must be assessed. BMJ, 317(7162): 883-4.
Sort P, Navasa M, Arroyo V et al. (1999) Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med, 341(6): 403-9.
Suarez JI, Martin RH, Hohmann SF et al. (2017) Human albumin use in adults in U.S. academic medical centers. Crit Care Med, 45(1): e16-22.
Todd S, Whitehead A, Stallard N et al. (2001) Interim analyses and sequential designs in phase III studies. Br J Clin Pharmacol, 51(5): 394-9.
Toraman F, Evrenkaya S, Yuce M et al. (2004) Highly positive intraoperative fluid balance during cardiac surgery is associated with adverse outcome. Perfusion, 19(2): 85-91.
Vincent JL, Wilkes MM, Navickis RJ (2003)
Safety of human albumin--serious adverse events reported worldwide in
1998-2000. Bri J Anaesth, 91(5): 625-30. PubMed ![]()
Wetterslev J, Thorlund K, Brok J et al. (2008) Trial sequential analysis may establish when firm evidence is reached in cumulative meta-analysis. J Clin Epidemiol, 61(1): 64-75.
Wiedermann CJ (2014a) Attrition bias in the 2014 meta-analysis of Patel and co-workers. Re: Randomised trials of human albumin for adults with sepsis: systematic review and meta-analysis with trial sequential analysis of all-cause mortality. BMJ;349: g4561.
Wiedermann CJ (2014b) Double-counted mortality data bias in the 2014 meta-analysis of Patel and co-workers. Re: Randomised trials of human albumin for adults with sepsis: systematic review and meta-analysis with trial sequential analysis of all-cause mortality. BMJ 2014;349:g4561.
Wiedermann CJ, Joannidis M (2014) Albumin replacement in severe sepsis or septic shock. N Engl J Med, 371(1): 83.
Woodcock TE, Woodcock TM (2012) Revised
Starling equation and the glycocalyx model of transvascular fluid exchange: an
improved paradigm for prescribing intravenous fluid therapy. Br J Anaesth,
108(3): 384-94. PubMed ![]()
Workman S (1999) Albumin controversy continues. Lack of efficacy shows that treatments de not work. BMJ, 318(7181): 464.
Xu JY, Chen QH, Xie JF et al. (2014)
Comparison of the effects of albumin and crystalloid on mortality in adult
patients with severe sepsis and septic shock: a meta-analysis of randomized
clinical trials. Crit Care, 18(6): 702. PubMed ![]()
Xue HP, LIin B, Mo JZ et al. (2002) Effect of albumin infusion on preventing the deterioration of renal function in patients with spontaneous bacterial peritonitis. Chin J Dig Dis, 3: 32-4.
Yunos NM, Bellomo R, Glassford N et al. (2014) Chloride-liberal vs. chloride-restrictive intravenous fluid administration and acute kidney injury: an extended analysis. Intensive Care Med, 41(2): 257-64.
Yunos NM, Bellomo R, Hegarty C et al. (2012) Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. JAMA, 308(15): 1566-72.








