ICU Management & Practice, Volume 18 - Issue 4, 2018
Expired CO2 can be easily monitored in the intensive care unit (ICU), especially in patients under invasive mechanical ventilation, using infrared measurement by sampling mainstream expiratory flow using an in-line chamber, or sidestream expiratory flow (by continuous aspiration through a sampling line connected between the intubation tube and the Y-piece of the ventilator).
Expired CO2 is determined by three parameters:
- CO2 production (CO2) mainly due to tissue metabolic activity
- CO2 transport related to cardiac output (CO) and haemoglobin level
- CO2 clearance by alveolar ventilation.
Given its high diffusive capacity, CO2 is easily eliminated by alveolar ventilation, although end-tidal CO2 partial pressure (PEtCO2) is higher than alveolar CO2 partial pressure (PACO2) due to ventilation-perfusion mismatch. The gradient between PEtCO2 and arterial CO2 partial pressure (PaCO2) is usually low (3-5 mmHg) but increases with increasing alveolar dead space, even though PEtCO2 remains highly correlated with PaCO2.
By allowing a combined analysis of respiratory, haemodynamic and metabolic status, capnography is a versatile tool with developing clinical applications in the ICU. While capnography is commonly used in the operating room, this technique may be underused in the ICU (Cook et al. 2011; Georgiou et al. 2010; Ono et al. 2016).The aim of this paper is to highlight the advances and the usefulness of expired CO2 monitoring in the specific ICU setting.
Capnography and respiratory intensive care
Airway management
Capnography is a reliable tool to confirm the correct placement of endotracheal (Guggenberger et al. 1989) or supraglottic devices, given the lack of significant CO2 production in the oesophagus. Despite a high diagnostic performance (Silvestri et al. 2005), false negatives are encountered in the cardiac arrest setting (Heradstveit et al. 2012) or as a consequence of technical pitfalls (leaks around endotracheal cuff (Dunn et al. 1990), kinking of the sampling line, ventilator failure…). False positives may happen if the stomach contains CO2 (e.g. in patients receiving noninvasive ventilation for hypercapnic respiratory failure). Finally, despite recommendations for systematic use during intubation in the ICU, the clinical impact of this strategy remains to date unknown in the ICU setting. Indeed, most of the recommendations are made from the NAP 4 audit (Cook et al. 2011), which showed that 74% of deaths related to airway issues in the ICU (tube displacement, oesophageal intubation) were associated with the lack of capnography.
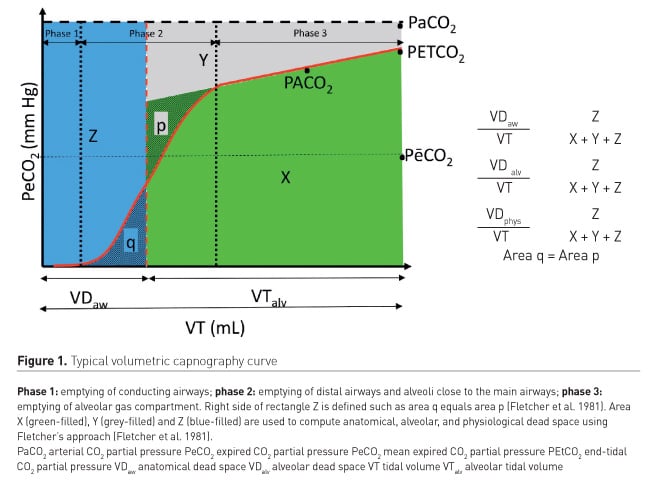
Dead space measurement and volumetric capnography
Partial pressure of expired CO2 is usually plotted against time on a capnogram, allowing assessment of PEtCO2. Partial pressure of expired CO2 may also be plotted as a function of expired tidal volume to provide volumetric capnography. An ideal volumetric capnography curve can be described in three expiratory phases (Figure 1) (Verscheure et al. 2016):
- Phase 1 - exhaled CO2 amounts to zero and reflects the lack of CO2 content in the conducting airways
- Phase 2 - CO2 increases linearly, reflecting mixing of CO2 content of distal airways and alveoli close to the main airways
- Phase 3 - CO2 reaches a slowly rising plateau (reflecting alveolar gas compartment), whose slope is an indication of ventilation-perfusion mismatch. Both anatomical (VDaw) and alveolar dead space (VDalv) can be computed by combining capnography and arterial blood sampling using graphical analysis of the volumetric capnography curve (Fletcher et al. 1981). Assuming PaCO2 approximates PACO2, physiological dead space (VDphys) can also be computed using the Enghoff modification of the Bohr equation, as follows:
VDphys Bohr-Enghoff = (PaCO2-PACO2)/PaCO2 with PACO2 = mean expired CO2 partial pressure
However, VDphys assessed with the Bohr-Enghoff equation overestimates the true VDphys, since venous admixture and low ventilation-perfusion areas increase the difference between PaCO2 and PACO2. It was recently shown that the midportion of phase 3 is a reliable estimator of PACO2 (Tusman et al. 2011), allowing a continuous assessment of true physiological dead space, without arterial blood sampling, using Bohr’s original equation, as follows:
VDphys Bohr = (PACO2-PeCO2)/PACO2
VDphys Bohr as assessed by volumetric capnography has been shown to be closely related to dead space measurement using the multiple inert gas technique (Tusman et al. 2011), and is not impacted by the effect of shunt or low ventilation-perfusion areas.
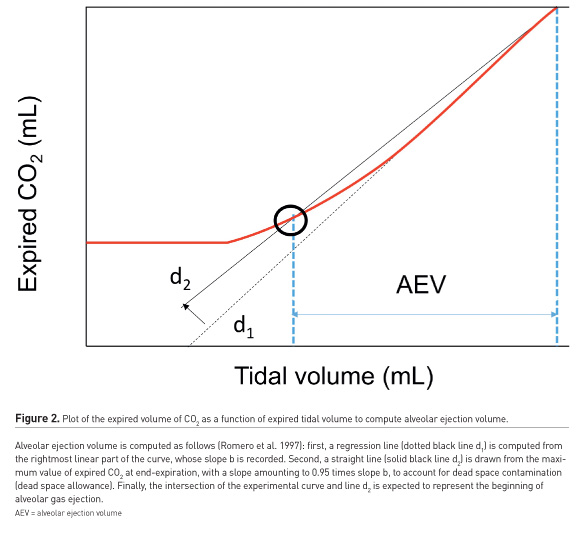
Finally, plotting the volume of expired CO2 as a function of expired tidal volume allows computation of VDalv (Fletcher et al. 1981) and alveolar ejection volume (Romero et al. 1997), defined at the predicted point where alveolar emptying begins (Figure 2), as an attempt to better quantify phase 3 of the volumetric capnography curve.
The following clinical applications of volumetric capnography have been reported:
- Nuckton et al. (2002) reported that dead space (computed from the Bohr-Enghoff equation) was strongly associated with acute respiratory distress syndrome (ARDS) mortality. In addition, Gattinoni et al. (2003) showed that decrease of dead space during prone position was associated with lower ARDS mortality. Alveolar ejection volume is also associated with ARDS mortality (Lucangelo et al. 2008), with the advantage of being independent of ventilatory settings (Romero et al. 1997). Whether strategies aiming to minimise deadspace decrease ARDS mortality remains however unknown.
- As early as 1975, Suter et al. (1975) showed that the "best positive end-expiratory pressure (PEEP)" (i.e. PEEP level associated with the highest oxygen transport) was associated with the lowest dead space (computed from the Bohr-Enghoff equation with a correction for the effect of shunt [Kuwabara et al. 1969]). Increase in dead space below and above the best PEEP level was interpreted as evidence of lung overdistension (and hence compression of alveolar vessels) at low and high lung aerated volume.
- Increased dead space fraction has been reported as an excellent predictor of extubation failure in a single study on ICU adult patients (González-Castro et al. 2011), a finding that should be confirmed before application in clinical practice.
However volumetric capnography presents the following limitations: measurement errors due to air leaks (e.g. during noninvasive ventilation) or obstruction of airway adaptor by secretions or condensation droplets, requirement of sufficient expiratory time in order to allow complete CO2 exhalation, deviation from the ideal 3 phases curve in some clinical situations, dependency to ventilatory settings etc.
PaCO2 surrogate
Capnography and haemodynamic intensive care
Cardiac arrest
The use of capnography in the cardiac arrest setting has been well documented and is recommended in both American and European resuscitation guidelines (Link et al. 2015; Soar et al. 2015). In this setting, capnography is a versatile tool that can help the management of cardiac arrest patients (Heradstveit et al. 2014).
First, as described above, it can confirm the correct placement of an endotracheal tube. Second, capnography may assess the quality of cardiac resuscitation, which is correlated with the PEtCO2 level, since PEtCO2 is related to cardiac output. Hence it is recommended (Paiva et al. 2018) to achieve a PEtCO2>20 mmHg during resuscitation. Third, capnography may help to detect the return to a spontaneous circulation if a sudden rise of PEtCO2 occurs. Fourth, initial low values or failure to maintain “correct” values of PEtCO2 are associated with worse outcome (Levine et al. 1997), and may help the decision to terminate resuscitation or help to triage patients with refractory cardiac arrest eligible for extracorporeal life support (Conseil français de réanimation cardiopulmonaire et al. 2009).
Cardiac output surrogate
As PEtCO2 is highly correlated to CO and monitored on a breath-by-breath basis (Weil et al. 1985), it may be used as a surrogate for continuous cardiac output monitoring over short periods, assuming CO2 production and elimination remain constant. In this connection, some authors recently investigated whether PEtCO2 variations could be used to track CO changes related to change in cardiac loading conditions (Monnet et al. 2012). In a study on 65 mechanically ventilated patients with acute circulatory failure, PEtCO2 increased by at least 5% during a passive leg raising manoeuvre predicted fluid responsiveness with 100% specificity and a sensitivity of 71%. For patients under veno-arterial extracorporeal membrane oxygenation (ECMO), PEtCO2 might reflect transpulmonary (or native) cardiac output. Naruke et al. (2010) reported that patients that were successfully weaned from veno-arterial ECMO exhibited a rise of PEtCO2 of at least 5 mmHg after reduction of ECMO flow, which was interpreted as a rise of native CO. If confirmed, this method could allow a more precise screening of patients who could be safely weaned from ECMO.
CO can be measured noninvasively with the differential Fick method, using measurements of CO2 elimination (VCO2) and PEtCO2 on a breath-by-breath basis before and during a CO2 rebreathing manoeuvre (Jaffe 1999). Since cardiac output is computed from expired CO2, blood flow from non-ventilated lung regions is not accounted for and a correction for shunt fraction has to be performed using arterial oxygen saturation assessed by a pulse oximeter and an iso-shunt diagram (Rocco et al. 2004). The NICO® system is a commercially available device based on this technique, using a rebreathing loop controlled by a pneumatic valve inserted at the Y-piece level. This technique has an acceptable reliability (Gueret et al. 2006) in cardiac surgery patients, but a lack of reliability in conditions of high intrapulmonary shunt (Rocco et al. 2004). In addition, the technique is hampered by several additional limitations: CO monitoring is not continuous (1 measurement every 3 minutes) making the technique unsuitable to assess fluid responsiveness by the passive leg raising test or other postural tests (Yonis et al. 2017); the technique is contraindicated in patients requiring strict control of PaCO2 (e.g. brain-injured patients); haemodynamic and respiratory instability (with rapidly changing VCO2 between the basal and rebreathing phase) may decrease the reliability of the CO measurement. The ideal patient for this technique would be mechanically ventilated, with no active breathing and no pulmonary disease, which probably makes the technique suitable for intraoperative monitoring.
Capnography for metabolic intensive care
Capnography for intrahospital transport
Conclusion
Conflicts of interest
Abbreviations
ARDS acute respiratory distress syndrome
CO cardiac output
CO2 carbon dioxide
ECMO extracorporeal membrane oxygenation
ICU intensive care unit
PACO2 CO2 alveolar partial pressure
PaCO2 CO2 arterial partial pressure
PeCO2 mean expired CO2 partial pressure
PEEP positive end-expiratory pressure
PEtCO2 end-tidal CO2 partial pressure
VCO2 CO2 production
VDalv alveolar dead space
VDaw anatomical dead space
VDphys physiological dead space
VO2 O2 consumption








