Medical imaging is uniquely positioned to play a pivotal role at many points in the cancer patient journey, from early detection as part of screening through to survivorship and/or end of life care. Reliance on imaging to contribute to decision making is substantive; a review of
Cancer Care Ontario’sLung Diagnostic Pathway revealed 22 touch points where a patient may have imaging as part of their diagnostic work-up (Cancer Care Ontario 2012a).
The value of radiology depends not only on the image produced, but also on the contribution to patient management decisions and clinical outcomes. To improve outcomes and to enhance patient care, we need to ensure that the end product of an imaging examination — the report — provides the information needed for optimum patient management. Although medicine is constantly evolving, radiological reporting has remained virtually unchanged. The format and quality of radiological reporting needs to evolve. Pathology reporting and surgical reporting are already undergoing changes (Cancer Care Ontario 2012b; Canadian Partnership Against Cancer 2012; Simpson et al, 2015). The time has come for quality improvement of radiology reporting related to cancer patients.
Radiology Report
Over the last 120 years report generation by radiologists has evolved from handwritten, to transcribed, to voice recognition formats, with little change in content and structure. The earliest known x-ray report (Reiner and Knight 2004) could easily be mistaken for a report created in 2015.
The radiology report is the primary communication tool between the radiologist and the referring physician, other radiologists who may reference the report during follow-up exams, and — increasingly — the patient.To facilitate clinical decision-making, the report must contain accurate information and address any specific clinical questions in language that is clear and understandable (American College of Radiology (ACR) 2014a). For efficiency and clarity, this information needs to be easily extracted from the report.
Current reporting standards for cancer or other indications are narrative; reports are created, stored and viewed as free text. Details are hidden within the report and can only be found by reading the report in its entirety. There is considerable variability in how radiologists report the same types of examinations (goergen 2011). This makes extraction of key clinical information difficult and time-consuming, and may lead to miscommunication of information and suboptimal patient care. Deficiencies in radiology reports have been identified and attributed to lack of organisation, clarity, succinctness and completeness (Johnson et al. 2004). Information that is necessary for decision-making can be missing (Naik et al 2001). To help alleviate some of the issues of miscommunication and/or completeness, referring physicians have indicated a preference for structured reports (Naik et al. 2001; Schwartz et al. 2011).
If the radiology report is lacking information or lacks clarity, the test cannot fulfil the functions outlined in the definition of appropriateness. The examination may be appropriately ordered, but if the needed information from the examination is not extracted and not reported than the expected impact on patient outcomes cannot be achieved.
In attempts to improve quality, various organisations have produced guidelines that outline the essential components of a report (Hall 2000; Stolberg 2002; Ridley 2002; ACR 2014a; Goergen 2011; Kahn et al. 2009). Based on these guidelines, a list of report content elements common to all radiologists’ reports has been compiled and grouped into four categories:
1. Demographics;
2. Relevant clinical information;
3. Body of the report;
4. Impression (conclusion or diagnosis).
Although these guidelines define the high-level content areas of a quality radiology report, there is limited or a complete lack of direction regarding how to most effectively convey this information to the referring physician.
Stakeholders
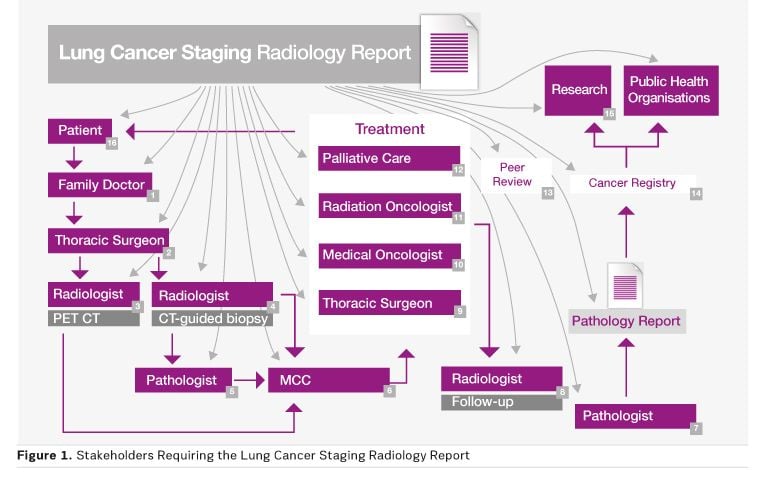
If we map what happens to a cancer imaging report we see that the contents are needed by more than just the referring physician. Stakeholders include patients and family members, family doctors, oncologists, the healthcare team, pathologists, radiologists, multidisciplinary tumour boards, collaborative staging, etc. As an example, a CT lung cancer staging report may be used by up to 16 stakeholders (see Figure 1), creating 16 opportunities for information to be erroneously interpreted. Each stakeholder may also have specific needs regarding the content.
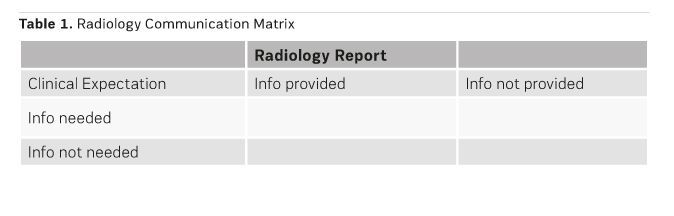
Radiology Communication Matrix
The clinical expectations and the information provided or not provided in the radiology report can be presented in a communication matrix (see Table 1). One dimension is the clinical expectation — what information stakeholders do or do not need. The second dimension is the information provided or not provided in the radiology report.
Ideally the radiology report would provide all the information needed by the stakeholder. Information provided in the report but not needed is relative and can vary between stakeholders.
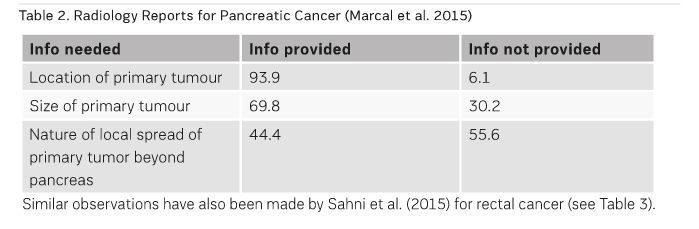
The radiology report is expected to provide the information that will impact on the clinical management decision at every point of the cancer journey in which imaging is required. However, recent literature demonstrates that this is not occurring a substantial portion of the time. Marcal et al. (2015) assessed the content of radiology reports for pancreatic cancer and noted many instances where the required information was not provided (see Table 2).
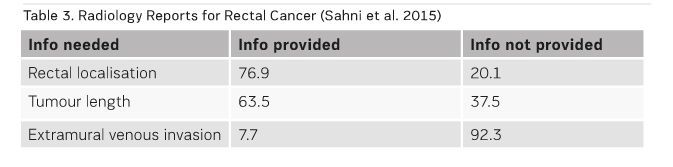
Similar observations have also been made by Sahni et al. (2015) for rectal cancer (see Table 3).
Structured Reporting
To decrease variability and improve quality of radiology reports, structured reporting is advocated (Schwartz et al 2011). The terms structured and standardised reporting have been used interchangeably in the literature and are a source of confusion. Even more confusing is the use of the following additional terms: standardised structured reporting, standardised electronic reporting, synoptic, proforma, itemised reporting, checklist reporting and template reporting.
Structured reporting uses elements arranged in a pattern of organisation, headers, that form the foundation of the report (Weiss et al 2008). These can be elementary to use — only subject headers — or be more advanced to include subsections, ie organs and measurements. To the structured reports can be added standardised elements – components of the report that can be expected to be reported in the same way across radiologists.
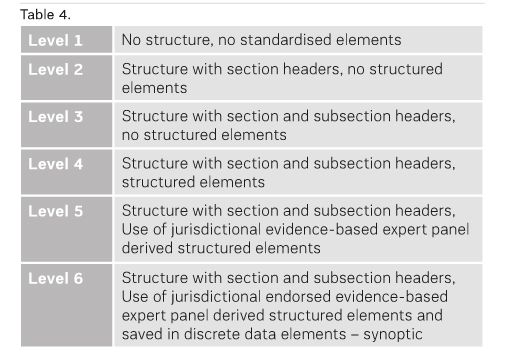
In the absence of consensus-based definitions for levels of structured reporting in radiology, we propose the following as working definitions (see Table 4). Having a spectrum from Level 1 to Level 6 with clearly distinguishing characteristics (describing content structure and content governance) is helpful in understanding the current status of reporting and in planning for the future.
The implementation of voice recognition software has allowed for the development and use of structured elements or templates. These templates allow for consistent format. At this time there are limitations with these systems with respect to moving from ‘structured’ to ‘synoptic’. However, enhanced data usage to benefit patient care (eg, real-time clinical staging or the contribution of specific imaging results in ‘personalised’ approaches to care) and interest in quality improvement activities such as peer review or correlation with synoptic pathology data is helping to move the field forward.
Benefits and Potential of Structured Reporting
Structured reporting has benefits for various stakeholders:
Patients
- Facilitates decision-making for treatment, ensuring communication is complete and efficient.<
- Important information is highlighted, facilitating direct, meaningful discussion with the referring physician in an environment where time is often limited.
Referring Physicians
- Improved communication (Al-Sukhni et al. 2013) allows for quick and accurate review of the complete radiological information needed for decision making.
- Provides a consistent format for discussion at multidisciplinary case conferences and comparisons over time (eg, treatment response assessment, monitoring progression, etc).
Radiologists
- Improves report completeness.
- Increases efficiency by decreasing the frequency of interaction with referring physicians due to “missing information” and decreasing the need for second reads.
- Easier to compare to previous reports and to follow effectiveness of treatment.
System
- Provides a foundation to facilitate system-level quality improvement oversight — eg, identification of patterns of care, outcomes, and data-driven system oversight (eg, appropriateness, system-level peer review).
- Discrete (synoptic) data fields enable tailored reporting to meet the needs of individual stakeholders; a single report can look different depending on the end user.
- Educational guidance through predefined standardised elements; structured reports provide a resource to radiologists to guide in the reporting of less common pathology, as well as a consistent standardised format for radiology residents to learn about and to understand the important elements of the radiology report.
- Additional benefits can be gained by linking radiology, pathology and surgical standardised (synoptic) reports. Creating a common, automated, interoperable informatics data model would allow for near real-time staging with timely decisions for patient management. Enhancing the electronic automation of staging data collection and combining radiologic and pathologic data leads to timely surveillance and planning analysis within the cancer system. Linking the data will result in improved assessment of tumour burden, tumour aggressiveness and tumour characteristics. Comparing linked fields will identify discrepancies and allow for feedback to both radiologists and pathologists. An automated, interoperable data model enables the development of radiology and radiology/pathology quality indicators, ie comparison of clinical and final pathological stages as a quality indicator (does it correlate with time interval between suspicion and treatment?; is the radiological staging appropriate?)
- More accessible data, facilitating secondary use of clinical data and data mining: increases the efficiency of central cancer registry data collection; improves population-based research, and supporting informatics assessments of quality of care, practice variations, response rates, outcomes, costs, and various demographic and genetic effects.
Cancer Care Ontario Cancer Imaging Structured Report Development
In 2009 the
Cancer Imaging Program (CIP) was established at Cancer Care Ontario to improve the quality of cancer imaging. The opportunity to improve quality through better reporting and communication by the use of synoptic radiology reports led to the addition of “
Synoptic Radiology Reporting” as a programme priority. The initiative is leveraging the knowledge gained from the development and implementation of provincial synoptic reporting for pathology, as well as the development of a synoptic report for pre-surgical staging of rectal cancer with MRI, a collaborative initiative between Cancer Care Ontario and Canadian Cancer Society (Spiegle et al. 2009).
To provide expert, interdisciplinary guidance regarding clinical content standards, the Synoptic Radiology Reporting Clinical Advisory Panel was established in 2013. This panel determined the need for the creation of a document that would be used by disease-site and/or modality-specific expert subcommittees to guide the creation of cancer imaging synoptic reports. Two white papers were developed on the architecture of the synoptic report (Cancer Care Ontario 2014a) and on governance (Cancer Care Ontario 2014b).
The Synoptic Radiology Reporting Clinical Advisory Panel recommends that the synoptic reports should:
- Be created by multidisciplinary expert groups.
- Have content informed by evidence where this evidence is available.
- Be aligned with appropriate overall clinical practice, as identified in disease pathways where they exist (eg, Cancer Care Ontario's disease pathways)
- Contain minimum mandatory elements needed to support clinical decision-making. Optional elements may also be recommended, but should be identified as such.
- Be clear and usable, and consider cross-referencing of data elements where applicable (eg, previous imaging studies or pathology synoptic reports
Radiology Structured Reporting Initiatives
Structured reports are not new to radiology. Modern radiology reporting is adopting more structured organisation and language, led by breast imaging reporting:
- Breast imaging: The BI-RADS® Atlas provides standardised breast imaging findings terminology, report organisation, assessment structure and a classification system for mammography, ultrasound and MRI of the breast. Breast imaging reporting quality has improved through the use of the Breast Imaging Reporting and Data Systems (BI-RADS®) reporting format and lexicon (ACR 2013a);
- Liver imaging: The Liver Imaging Reporting and Data System (LI-RADS) was created to standardise the reporting and data collection of CT and MR imaging for hepatocellular carcinoma (HCC) lexicon (ACR 2013b);
- Prostate imaging: Reporting and Data System (PI-RADS) for prostate cancer (ACR 2015);
- Lung imaging: The ACR Lung Imaging Reporting and Data System (Lung-RADS™) (ACR 2014b) for lung cancer screening.
- Report templates: Société Française de Radiologie (SFR) has published cancer reporting templates (SFR 2015). The Radiological Society of North America (RSNA) Reporting Initiative is an online library of bestpractices radiology report templates including cancer-related reports (RSNA 2015).
Governance is an important consideration for quality report generation. To ensure structured radiology reports guide radiologists to provide all information required to meet the needs of all stakeholders, templates should be developed by multidisciplinary expert panels, guided by the best-available evidence. Breadth of jurisdictional representation (eg, regional to — potentially — international) is also a consideration, depending on disease site and intended usage.
The list above provides examples of various initiatives intended to improve the quality of radiology reporting for select sites. However, fundamentally, the quality of all radiology reporting related to all cancer patients needs to improve.
Moving Forward with Cancer Imaging Structured Reporting
For structured reporting to gain acceptance in the broader radiology community will depend on governance, technology and mandatory use of structured reports.
Structured reporting of cancer imaging needs a home where there are clear guidelines for development of structured reports and clear mechanisms for updating of the reports as new evidence emerges. In pathology, Cancer Care Ontario's success can be attributed in part to the development and maintenance of the cancer checklists by the College of American Pathologists (Simpson et al 2015). Radiology needs to find a similar mechanism of creation and maintenance of evidence-based structured reports.
Reporting technology also needs to evolve, with seamless, effective integration of structured reporting into the radiologist workflow. This is key to ensuring success. Although many systems support templates, constructs such as conditional logic (eg, hiding options if not relevant) are not available, greatly impacting radiologist workflow. Additionally, the full benefit of synoptic reporting and data capture cannot be realised, as regardless of report ‘input,’ downstream systems such as the radiology information system (RIS) or hospital information system (HIS) only accept narrative-style reports.
The willingness of hospitals, health organisations, and/or government to mandate the use of structured reporting will also impact success. Sahni found that voluntary implementation of structured reporting resulted in a 80% uptake (Sahni et al. 2015 ). Voluntary implementation of the rectal cancer staging MRI structured report resulted in a 60 percent uptake (Sahni et al. 2015). Cancer patients require 100 percent uptake.
Conclusion
Structured reporting is not new in medical imaging and its use to improve quality is gaining momentum. Widespread acceptance of structured reporting will not be easy with the current barriers. As more high quality evidence-based structured reports are created and disseminated, as technology evolves, and as appropriate governance structures are developed the pressures for use of structured reporting will continue to grow. Radiologists can take the lead and work with industry to design reporting systems that are more efficient and systems that integrate the reporting process with the picture archiving and communication system (PACS) and the image review process.
It is time for the radiology community, in collaboration with referring physicians, to move towards reporting that meets the needs of the ‘report reader’, who is making decisions regarding patient care, versus the ‘report generator’. This is our opportunity to improve the quality of care and make a real difference in patient outcomes.
Acknowledgements
We gratefully acknowledge the helpful discussions and technical contributions of the Structured Data Team and the Pathology Electronic Reporting Committee at the College of American Pathologists (CAP). In particular, we acknowledge the contributions of Drs. Jaleh Mirza, Richard Moldwin and Samantha Spencer at the CAP. We also gratefully acknowledge the contribution of our Cancer Care Ontario team including Deanna Langer, Colleen Bedford, Priyanka Jain, David Kwan and Gemma Lee.
Al-Sukhni E, Messenger DE, Charles Victor J et al. (2013) Do MRI reports contain adequate preoperative staging information for end users to make appropriate treatment decisions for rectal cancer? Ann Surg Oncol, 20: 1148–55.
American College of Radiology (2013a) ACR BI-RADS® atlas. [Accessed: 9 November 2015] Available from acr.org/Quality-Safety/Resources/BIRADS
American College of Radiology (2013b) Updated version of LI-RADS™ is now available. Inside Quality and Safety eNews, March 2013. [Accessed: 9 November 2015] Available from acr.org/Quality-Safety/eNews/Issue-01-March-2013/LIRADS
American College of Radiology (2014a) The ACR practice parameter for communication of diagnostic imaging findings. Revised 2014, Resolution 11. Reston, VA: American College of Radiology. [Accessed: 9 November 2015] Available from acr.org/~/media/C5D1443C9EA4424AA12477D1AD1D927D.pdf
American College of Radiology (2014b) Lung CT screening reporting and data system (Lung-RADS™. [Accessed: 9 November 2015] Available from http://www.acr.org/Quality-Safety/Resources/LungRADS
American College of Radiology (2015) Now available: PI-RADS™ version 2. Inside Quality and Safety eNews, March 2015. [Accessed: 9 November 2015] Available from acr.org/Quality-Safety/eNews/Issue-09-March-2015/PIRADS-Version-2
Canadian Partnership Against Cancer (2012) Synoptic reporting (surgery) [Accessed: 3 April 2014] Available from www.partnershipagainstcancer.ca/priorities/2007-2012-initiatives/cancer-guidelines-2007-2012-strategic-initiatives/synoptic-surgical-reporting-2/
Cancer Care Ontario (2012a) Lung cancer diagnosis pathway. Version 2012.02. [Accessed: 9 November 2015] Available from cancercare.on.ca/common/pages/UserFile.aspx?fileId=298429
Cancer Care Ontario (2012b) Synoptic pathology reporting. [Accessed: 3 April 2014] Available from cancercare.on.ca/ocs/clinicalprogs/pathnlabmed/pathproj_prof
Cancer Care Ontario (2014a) Synoptic radiology report for cancer imaging: establishing the minimum elements required for a quality synoptic report. [Accessed: 9 November 2015] Available from cancercare.on.ca/common/pages/UserFile.aspx?fileId=324328)
Cancer Care Ontario (2014b) Synoptic radiology reporting: clinical checklist development governance. [Accessed: 9 November 2015] Available from cancercare.on.ca/common/pages/UserFile.aspx?fileId=324322
Goergen SK (2011) Radiology written report guideline, version 5. Sydney, NSW: Royal Australian and New Zealand College of Radiologists. [Accessed: 9 November 2015] Available from ranzcr.edu.au
Hall FM (2000) Language of the radiology report: primer for residents and wayward radiologists. AJR Am J Roentgenol, 175(5): 1239-42.
Johnson AJ, Ying J, Swan JS et al. (2004) Improving the quality of radiology reporting: a physician survey to define the target. J Am Coll Radiol, 1(7): 497-505.
Kahn CE Jr, Langlotz CP, Burnside ES et al. (2009) Toward best practices in radiology reporting. Radiology, 252(3): 852-6.
Marcal LP, Fox PS, Evans DB et al. (2015) Analysis of free-form radiology dictations for completeness and clarity for pancreatic cancer staging. Abdom Imaging, 40(7): 2391-7.
Naik SS, Hanbidge A, Wilson SR (2001) Radiology reports: examining radiologist and clinician preferences regarding style and content. AJR Am J Roentgenol, 176(3): 591–8.
Radiological Society of North America (2015) RSNA informatics™ reporting. [Accessed: 9 November 2015] Available from radreport.org/
Reiner BI, N. Knight N (2004) Reinventing the radiology report: part I, a history. Imaging Economics, November/December. [Accessed: 15 October 2015] Available from old.imagingeconomics.com/2004/11/reinventing-the-radiology-report-part-i-a-history
Ridley LJ (2002) Guide to the radiology report. Australas Radiol, 46(4): 366-9.
Sahni VA, Silveira PC, Sainani NI et al. (2015) Impact of a structured report template on the quality of MRI reports for rectal cancer staging. AJR Am J Roentgenol, 205(3): 584-8.
Schwartz LH, Panicek DM, Berk AR et al. (2011) Improving communication of diagnostic radiology findings through structured reporting. Radiology, 260(1): 174–81.
Simpson RW, Berman MA, Foulis PR et al. (2015) Cancer biomarkers: the role of structured data reporting. Arch Pathol Lab Med, 139(5): 587-93.
Société Française de Radiologie (2015) Comptes rendus normalisés. [Accessed: 9 November 2015] Available from sfrnet.org/sfr/societe/6-groupes-et-societes-specialises/gica/comptes-rendus-normalises/index.phtml
Spiegle G, Leon-Carlyle M, Schmocker S et al. (2009) Development of a synoptic MRI report for primary rectal cancer. Implement Sci, 4: 79.
Stolberg HO (2002) Radiology reporting handbook. Can Assoc Radiol J, 53 (2): 63-72.
Weiss DL, Langlotz CP (2008) Structured reporting: patient care enhancement or productivity nightmare? Radiology, 249(3): 739–47.