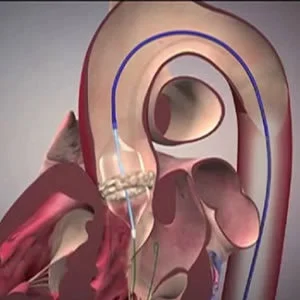
This year 2017 marks the 15th anniversary of the first transcatheter aortic valve replacement (TAVR) performed in Rouen in April 2002. The innovative procedure, which profoundly altered the landscape of cardiovascular medicine, can be considered as among the major clinical breakthroughs of the past decade, says an Editorial in the Journal of the American College of Cardiology.
You might also like: TAVI or Not TAVI — in Low Risk Patients? That Is the Question
The editorial, written by the French pioneers of TAVR themselves, highlights two recent studies published in the same issue of JACC that confirm the fascinating expansion of TAVR (both in the U.S. and France) and the progressive improvement of results and safety due to better patient selection, refinements in technology and procedural approaches, as well as improved operator experience.
While the two studies covered different topics on TAVR, both were based on the analysis of data from nationwide registries over a similar period of time.
"For the pioneers who remember the beginning of the story, marked by the fierce opposition of all experts toward a so-called totally unrealistic and stupid idea, and who also had to face the incredibly challenging clinical pathway imposed by health authorities for clinical evaluation, such important real-world registries are particularly meaningful," states the editorial.
The retrospective observational study by Carroll et al. assessed the correlation between TAVR site volume and in-hospital adjusted and unadjusted outcomes in high-risk elderly patients (mean 83 years of age), with data from the Society of Thoracic Surgeons/American College of Cardiology TVT (Transcatheter Valve Therapy) Registry. Carroll's team used the TVT registry database from 42,988 straightforward procedures (of a total of 47,270 TAVRs performed) at 395 sites from 2011 to 2015. Two-thirds of the patients underwent TAVR using the transfemoral access.
Carroll et al. estimated at 100 patients the steep learning curve necessary to achieve a plateau of procedural success and complications, beyond which procedural safety and outcome might continue to more gradually improve. "This number of 100 cases seems surprisingly high in regard to our personal appraisal and is much higher than in other reports, more particularly when the transfemoral approach is concerned. However, the information from this report might help updating recommendations of institutional and operator requirements when planning for programmatic expansion in the United States," the editorial points out.
Meanwhile, Auffret et al. report and compare the procedural changes and 30-day clinical outcomes from two nationwide French TAVR registries – FRANCE TAVI (French Transcatheter Aortic Valve Implantation) registry and FRANCE 2 (French Aortic National CoreValve and Edwards 2) registry – over a six-year period. Their report, the editorial says, clearly reflects the evolving experience of TAVR in France and offers an interesting overview of the trends in patient selection, TAVR devices used, procedural issues, and mortality and morbidity associated with the procedure in real life, including complex cases and valve-in-valve procedures. Furthermore, a sharp move toward a simplified transfemoral strategy was observed, including conscious sedation, pure percutaneous transfemoral access with preclosing, no periprocedural transesophageal echocardiography guidance, and short hospitalisation stay.
As the editorial notes, "Impressive growth of TAVR can be predicted in the next decade with advanced technologies and procedural simplification. A further and broad expansion of TAVR in low-risk or younger patients may come from the expected results of three ongoing major randomised trials in this population."
Source: Journal of the American College of Cardiology
Image Credit: Lee Health
References:
Cribier, Alain et al. (2017) TAVR, 15 Years Down : Shooting for the Moon, Reaching the Stars. Journal of the American College of Cardiology. doi.org/10.1016/j.jacc.2017.05.008
Latest Articles
TAVR, cardiovascular medicine, transcatheter aortic valve replacement, TAVR devices
TAVR has altered the landscape of cardiovascular medicine and can be considered as among the major clinical breakthroughs of the past decade, says an Editorial in the Journal of the American College of Cardiology.