When transcatheter aortic valve implantation (TAVI) was introduced in 2002, it was proposed as a therapy for inoperable or very high risk patients. Recently, the procedure has also been performed on younger lower risk persons with less comorbidities. The evolution in the indication for TAVI might be regarded as a model for the uptake of a new therapy, first meeting an unmet clinical need and then broadening its target population based on strong clinical trial evidence, according to an Editorial appearing in the journal Heart, Lung and Circulation
See Also: Is PCI Safe for Patients Undergoing TAVR?
International guidelines initially recommended TAVI in inoperable or high risk patients whose life expectancy is more than one year and the patient’s quality of life is likely to improve with implantation, based on clinical trials which have demonstrated the safety and even superiority of this procedure over medical therapy or surgical aortic valve replacement (SAVR). TAVI is approved in over 50 countries for high risk patients.
High risk has been defined using a logistic EuroSCORE of over 20%, EuroSCORE II of over 10% or STS Score over 8%. Patient frailty is also utilised but is difficult to define, but ability to ambulate seems particularly important. Weight loss in the past year, low activity, exhaustion, low gait speed and reduced grip strength are predictive of surgical outcome.
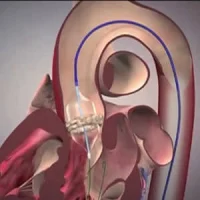
TAVI is now being performed with safety in Europe in lower risk patients. "Along with this shift in target population, there has been a remarkable evolution in the procedure itself, including a reduction in delivery sheaths from 21F to 14F, the provision of sealing cuffs, improvements in deliverability, the ability to reposition and retrieve, and the move to a minimalist approach under local anaesthesia," notes the author, Con Aroney, MBBS, MD, Cardiology Services, Holy Spirit Northside Hospital, Brisbane, Qld, Australia.
In addition, safety has been facilitated by developments in imaging and its analysis particularly using CT scanning, which has led to more accurate determination of device sizing and demonstrated potentially troublesome calcium nodules (which may interfere with device apposition or increase the risk of annular rupture).
In the meta-analysis reported in the same issue of the journal, compared to SAVR, short-term follow-up of TAVI patients showed a trend to lower mortality and stroke, with significantly lower risk of bleeding and acute kidney injury, but an increased risk of vascular injury, paravalvular regurgitation and pacemaker implantation. In the only randomised control trial included in the meta-analysis, the mean STS score was only 3.0%, and the TAVI patients also had a larger improvement in effective orifice area and a higher NYHA class at one year compared to patients undergoing SAVR.
"The awaited results of three large trials which have included low risk patients (STS score <4%), PARTNER 3, SURTAVI and UK TAVI will be pivotal in confirming the broadened indication," says Dr. Aroney.
Although there is increasing evidence for the short-term clinical safety of TAVI in lower risk patients, the author says extension of the procedure to these patients also requires consideration of key issues such as cost effectiveness, long-term valve durability, paravalvular regurgitation and permanent pacemaker requirement. "Despite higher procedural costs with TAVI than SAVR, overall costs with TAVI are offset by reduced ICU and hospital stays and post-discharge residential care," Dr. Aroney points out. Further improvements in TAVI technology, higher volumes and increased experience, lower complications, improved care, and reduced costs are all likely to improve cost-effectiveness in the future, the doctor adds.
The author also cites some major national and political influences regarding the appropriate use of TAVI. "In Europe and North America, where TAVI is approved and remunerated, the question now asked, and examined in this issue of HLC, is should TAVI be extended to low risk patients? In Australia, however, where TAVI does not currently enjoy a surgical item number or remuneration, the question is when will this life-saving therapy be made available at all?"
Source: Heart, Lung and Circulation
Image Credit: Lee Health
References:
Aroney, Con. (2017) TAVI or Not TAVI—in Low Risk Patients? That Is the Question. Heart, Lung and Circulation. doi.org/10.1016/j.hlc.2017.02.002
Latest Articles
TAVI, SAVR, Hospital Stays, Low Risk Patients, procedural costs
The evolution in the indication for TAVI might be regarded as a model for the uptake of a new therapy, first meeting an unmet clinical need and then broadening its target population based on strong clinical trial evidence, according to an Editorial.