The world's first leadless pacemaker has
demonstrated promising results as per a study conducted by Vivek Reddy, MD,
Director of Arrhythmia Services for the Mount Sinai Hospital. Twelve-month follow-up data with 32 bradycardia
patients showed that the performance of the leadless pacemaker device was
equivalent to those in traditional pacemakers and the patients did not
experience any infections or any failure to pace, sense or communicate with the
pacemaker.
The results of the study were presented by Dr. Reddy at Heart Rhythm 2014, the 35th Annual Scientific Session of the Heart Rhythm Society. According to Dr. Reddy, "our results show the leadless pacemaker is comparable to traditional pacemakers. Our latest findings further support the promising performance and safety of this minimally-invasive non-surgical pacing device."
Dr. Reddy is the first to implant this leadless pacemaker in the United States. Mount Sinai will be launching LEADLESS II, a clinical trial that will further test the effectiveness of the leadless pacemaker, with a specific focus on safety and efficacy. The trial will include 670 patients at 50 centers across Canada and the United States.
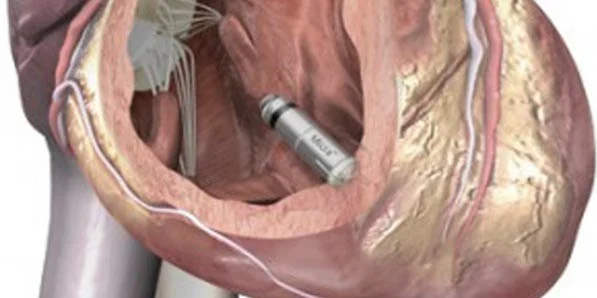
The leadless pacemaker is miniature-sized and resembles a tiny, metal silver tube that is only a few centimeters in length. The size of this pacemaker is less than 10 percent the size of a traditional pacemaker, and placed directly inside the patient's heart. The promising results from this study indicate the possible improvements that will occur in the years to come for the 4 million patients around the globe in need of a pacemaker.
The biggest advantage of this new-age device is the fact that it eliminates the need for surgery and that there is no visible device under the patient's skin. There is also no incision scar, no connector or leads and no limitations on a patient's daily activities. Overall, patients can greatly benefit from less discomfort, infections, complications and dysfunction.
Image source: Google Images