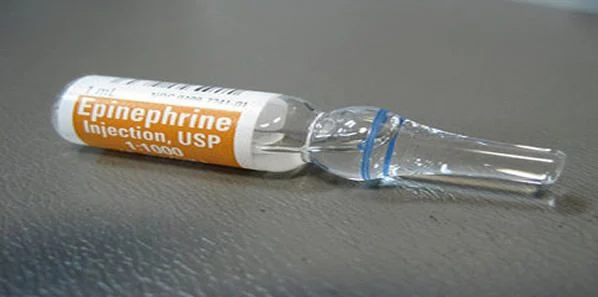
Over the past decade several scientists have suggested that adrenaline, used for more than fifty years as a key part of resuscitation, may actually do more harm than good. This is based on the evidence that adrenaline may damage the brain by reducing blood supply to the head and may in fact diminish the patient's chances of survival. This is a definite cause for concern since more than 50,000 people suffer from cardiac arrests outside of hospitals each year in the UK. It is believed that most people do not survive a cardiac arrest and only 8 percent of people actually leave the hospital alive.
Warwick University has received approval for a study that will investigate the effect of placebo in cardiac arrest patients versus adrenaline, to kick-start their heart. The study will be conducted with 8,000 people in London, Wales, the West Midlands and the South Coast and north-east of England.
Half of the study patients will be given the dummy medicine while the other half will receive adrenaline. Since the patients will be those who are suffering from a cardiac arrest, there will be no consent gathered before the drugs are administered. Paramedics will also not be obtaining any consent from family members or passers-by. Relatives will also not be informed whether their family member received the placebo or adrenaline. However, local residents will be provided information about the study and will also be given details as to how they can opt out.
While there is a definite need to investigate the effects of adrenaline, there is some concern that a study regarding a life-and-death intervention could be conducted without informed consent. It is being questioned whether this research is beneficial enough to forego this element. The idea of informed consent has always been at the heart of clinical trials. However, since this particular study will be conducted in patients who will be unconscious, there is a possibility that the aspect of consent could be let go. The trial could potentially save many lives but will definitely involve patients that will not be part of the decision making process.
According to Professor Peter Weissberg of the British Heart Foundation, "It is important to remember we don't actually know whether this routine use of adrenaline is a safe and effective practice." The researchers are aware of the difficulty under which this trial will be conducted but they believe that it is necessary and that there are still well established ethical guidelines that will be followed when undertaking this study. They are of the opinion that it is unacceptable to continue giving patients a treatment that could possibly cause them more harm than good. The study is set to begin this autumn.
Source: BBC News
Image Credit: Flickr.com
Warwick University has received approval for a study that will investigate the effect of placebo in cardiac arrest patients versus adrenaline, to kick-start their heart. The study will be conducted with 8,000 people in London, Wales, the West Midlands and the South Coast and north-east of England.
Half of the study patients will be given the dummy medicine while the other half will receive adrenaline. Since the patients will be those who are suffering from a cardiac arrest, there will be no consent gathered before the drugs are administered. Paramedics will also not be obtaining any consent from family members or passers-by. Relatives will also not be informed whether their family member received the placebo or adrenaline. However, local residents will be provided information about the study and will also be given details as to how they can opt out.
While there is a definite need to investigate the effects of adrenaline, there is some concern that a study regarding a life-and-death intervention could be conducted without informed consent. It is being questioned whether this research is beneficial enough to forego this element. The idea of informed consent has always been at the heart of clinical trials. However, since this particular study will be conducted in patients who will be unconscious, there is a possibility that the aspect of consent could be let go. The trial could potentially save many lives but will definitely involve patients that will not be part of the decision making process.
According to Professor Peter Weissberg of the British Heart Foundation, "It is important to remember we don't actually know whether this routine use of adrenaline is a safe and effective practice." The researchers are aware of the difficulty under which this trial will be conducted but they believe that it is necessary and that there are still well established ethical guidelines that will be followed when undertaking this study. They are of the opinion that it is unacceptable to continue giving patients a treatment that could possibly cause them more harm than good. The study is set to begin this autumn.
Source: BBC News
Image Credit: Flickr.com
Latest Articles
resuscitation, cardiac arrest, clinical trials, adrenaline, informed consent
Over the past decade several scientists have suggested that adrenaline, used for more than fifty years as a key part of resuscitation, may actually do more...