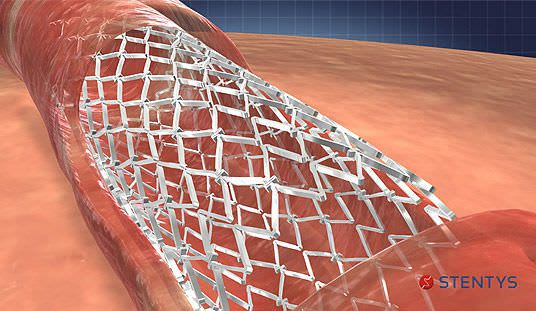
STENTYS® offers innovative solutions to the interventional cardiologist in the treatment of patients with acute myocardial infarction and other complex coronary disease. The STENTYS product portfolio consists of the STENTYS Self-Apposing® BMS and DES(P) stents and the STENTYS AC aspiration catheter. Only the STENTYS BMS is currently being studied in the U.S. in the APPOSITION V study to support marketing authorization by the FDA.
With offices in France (Paris) and in the U.S. (Princeton, NJ), STENTYS became listed on the Paris EuroNext stock exchange (STNT.PA) in October 2010. STENTYS® received CE-Marking for its BMS and DES(P) stents in early 2010 and these products have been marketed and sold in more than eight European countries.