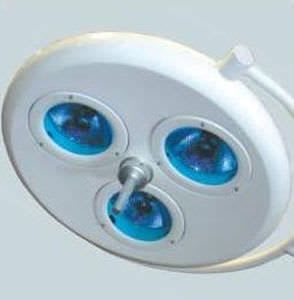
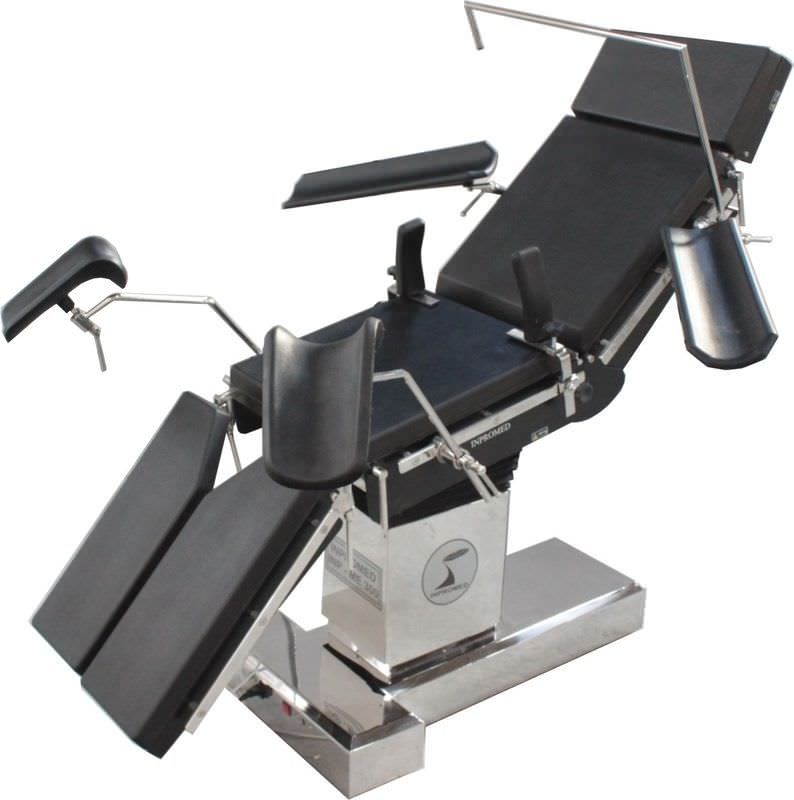
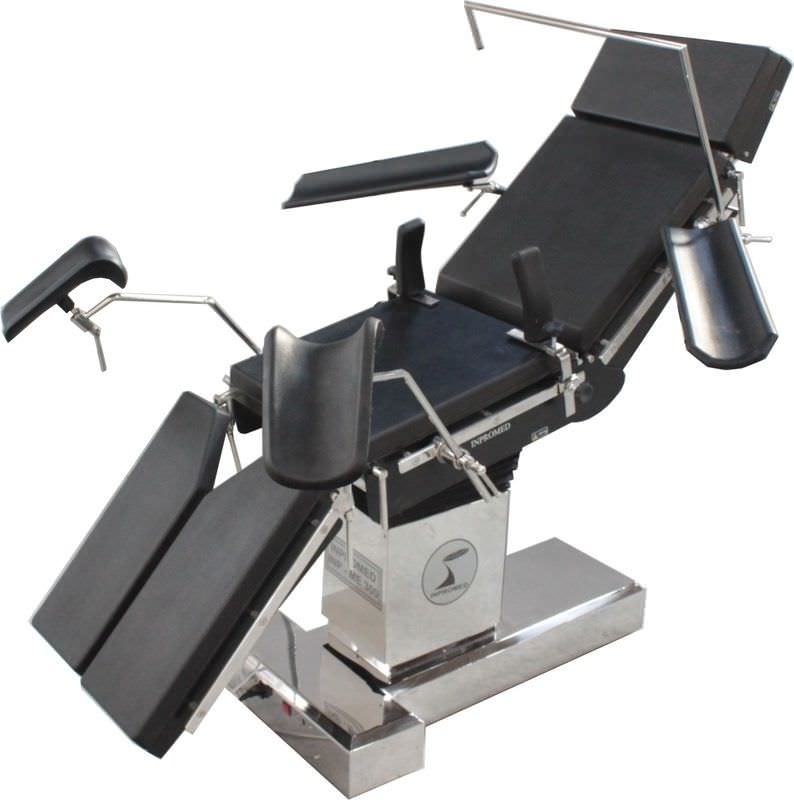
The INPROMED OF BRAZIL began in 1999 with the concept of sustainable growth, technological innovation and responsibility with all stakeholders, employees, customers, suppliers and society.
The constant improvement of its processes and the concern for their public Inpromed motivated in Brazil to consolidate the certifications: ISO 9001; ISO 13485; Good Manufacturing Practices - ANVISA and the European Marking - CE MARK, incontestable testimonies quality in national and international markets.
Brazil Inpromed of the aims to meet all the expectations of the market national and international consumer, providing medical products with the latest technology. This thanks to its team of highly qualified employees, who are able to develop, produce and perform after sales of equipment. Ensuring total customer satisfaction and Business Partners.