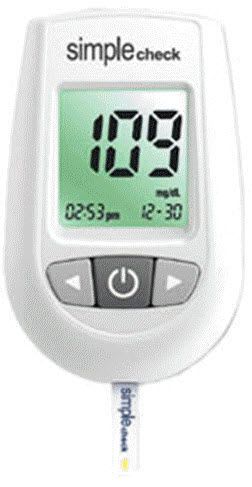
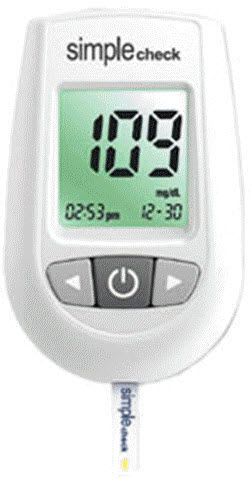
Chungdo Pharm Co., Ltd. We have established our standards with the active assistance of our associates in U.S.A. and the Korea Institute of Science and Technology. Our commitment is to provide the very best quality product available. We go to great lengths to ensure that our products will perform exactly as indicated. Using the best available controls on our state of the art equipment, we rigorously test every lot to ensure that it performs to the highest standards.
We train all our staff throughout the entire manufacturing process to guard our quality standards carefully, so that our quality is manufactured into the product, not inspected in. Our new facility outside of Pusan, Korea is a state of the art manufacturing plant. In developing this we sought out the most respected engineers in the field of dry reagent technoloty available world wide.
We practice Good Manufacturing Practices(GMP), our standards are designed to comply with all United States FDA regulations and our Leucocytes is patented in U.S.A. Our research and development facilities are also state of the art.
Our programs are in the process of being coordinated with our U.S. associates to ensure the best cooperative efforts of both laboratories. We are sure that in ensuring years, our technology will be ranked among the highest in the industry. However, we shall never lose sight of our objective of providing this quality at the lowest possible price.