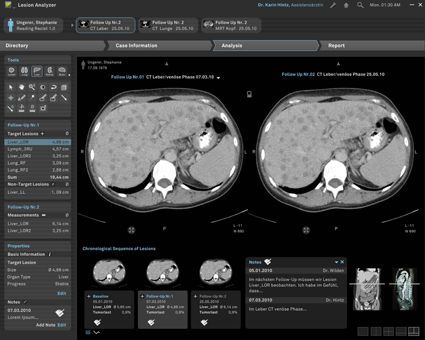
Comprehensible treatment assessment in Oncology and Radiology mint Lesion™ supports radiologists and oncologists in assessing the effectiveness of a cancer therapy. On basis of medical image interpretation, it provides immediate feedback about individual patient cases or clinical trial patient cohorts in terms of whether and how patients are responding to a particular treatment. mint Lesion™ is an approved medical product in the European Union (CE-labeled), the United States (FDA 510k-cleared), and further countries. Fields of Application What can mint Lesion™ be used for? - mint Lesion™ as Tumor Response Evaluation Solution: Streamline your daily readings and benefit from instant access to in-depth knowledge about any lesion from various imaging modalities. - mint Lesion™ as a Clinical Trial Imaging Management System (CTIMS) in clinical pharmaceutical research: Optimize efficiency and quality of clinical trials and benefit from a 100 % compliance to your particular clinical trial protocol. - mint Lesion™ in Quantitative Imaging Research: Gain substantial contributions to research by linking your quantitative reading results and benefit from our unique flexibility in modeling individual criteria and procedures with mint Lesion™.
- Function:viewing , diagnostic
- Application domain:medical imaging, medical