Tiered FDA medical device security guidance concerns industry
- IT
- 26/03/2019
The U.S. Food and Drug Administration (FDA) recently released its draft guidance on managing cybersecurity in medical device premarket submissions....
READ MORE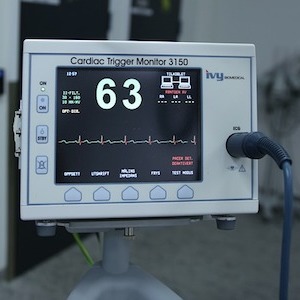
The U.S. Food and Drug Administration (FDA) recently released its draft guidance on managing cybersecurity in medical device premarket submissions....
READ MORE
Geneva Annual Blockchain Congress 2020
READ MORE

The Australian Telehealth Conference
READ MORE
With the launch of Samitivej "Virtual Hospital" app, Thai patients will have a convenient way to consult with specialist doctors – anywhere they...
READ MORE
Physician-scientist, EricTopol , has published his fourth book, honing in on how artificial intelligence (AI) has the potential to re-humanise...
READ MORE
As more medical equipment and mHealth devices get interconnected with healthcare IT systems , providers and other organisations are increasingly...
READ MOREThe importance of getting access management right cannot be overstated. You need to make sure that anybody accessing your systems – and the data within – is exactly who they say they are. This is especially important when dealing with healthcare professionals who have access to vast amounts of sensitive medical data. Most compliance...
READ MORE

Amazon Web Services (AWS) is supporting machine learning in patient care at Beth Israel Deaconess Medical Center (BIDMC) with an academ
READ MORE