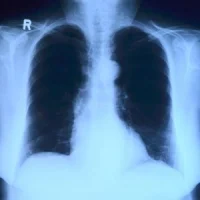
Intermountain Medical Center's (Utah, USA) new lung cancer screening programme that uses CT technology provides long-time smokers and past smokers a more accurate way of detecting whether or not they have the disease. The programme is based on the findings of the National Lung Screening Trial, a study that compared CT scans and standard chest X-rays in detecting lung cancer. The study showed that patients who received CT scans had 15 to 20 percent lower risks of dying compared to those who received a standard x-ray.
The lung cancer screening programme at Intermountain is a direct result of that effort, according to Denitza Blagev, MD, a pulmonologist at the medical centre. "Because of the best practices that came from this research, we were able to diagnose three people with early stage lung cancer in the programme's first 18 months." Dr. Blagev and her colleagues presented their findings at the American Thoracic Society International Conference in Denver.
Intermountain's new programme was tested between September 2014 and March 2015 and initially gave CT scans to 375 patients, 272 of whom were eligible to continue in the programme. Of the 272 patients who were eligible, 19 of their scans found evidence of malignant cancer. From those 19, 11 were confirmed malignant. Eight of the 11 were confirmed to have lung cancer, and three of the cases were considered early stage lung cancer.
Patients enrolled in the programme were considered high risk for lung cancer, Dr. Blagev said. They all had a smoking history of at least 30-pack years, which is the equivalent of smoking one pack of cigarettes every day for 30 years, and were current or former smokers without any signs or symptoms of lung disease.
"Our goal is to save every life that we can. Our results are comparable to the National Lung Screening Trial, in which it was deemed a success if they prevented just one cancer death for every 320 patients screened," Dr. Blagev pointed out. "The programme we created from this study detected three early stage lung cancers in the first 357 patients screened, which is incredible."
Results from the National Lung Screening Trial and from Intermountain's programme have shown that "screenings are very effective and will greatly benefit our patients going forward," she added.
Source: Intermountain Medical Center
Image credit: Pixabay
The lung cancer screening programme at Intermountain is a direct result of that effort, according to Denitza Blagev, MD, a pulmonologist at the medical centre. "Because of the best practices that came from this research, we were able to diagnose three people with early stage lung cancer in the programme's first 18 months." Dr. Blagev and her colleagues presented their findings at the American Thoracic Society International Conference in Denver.
Intermountain's new programme was tested between September 2014 and March 2015 and initially gave CT scans to 375 patients, 272 of whom were eligible to continue in the programme. Of the 272 patients who were eligible, 19 of their scans found evidence of malignant cancer. From those 19, 11 were confirmed malignant. Eight of the 11 were confirmed to have lung cancer, and three of the cases were considered early stage lung cancer.
Patients enrolled in the programme were considered high risk for lung cancer, Dr. Blagev said. They all had a smoking history of at least 30-pack years, which is the equivalent of smoking one pack of cigarettes every day for 30 years, and were current or former smokers without any signs or symptoms of lung disease.
"Our goal is to save every life that we can. Our results are comparable to the National Lung Screening Trial, in which it was deemed a success if they prevented just one cancer death for every 320 patients screened," Dr. Blagev pointed out. "The programme we created from this study detected three early stage lung cancers in the first 357 patients screened, which is incredible."
Results from the National Lung Screening Trial and from Intermountain's programme have shown that "screenings are very effective and will greatly benefit our patients going forward," she added.
Source: Intermountain Medical Center
Image credit: Pixabay
Latest Articles
healthmanagement, lung cancer, computed tomography, CT scans, chest x-rays, smoking
Intermountain Medical Center's (Utah, USA) new lung cancer screening programme that uses CT technology provides long-time smokers and past smokers a more accurate way of detecting whether or not they have the disease.