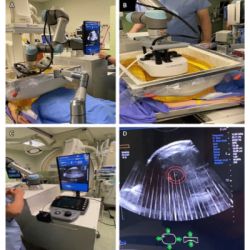
University College London Hospitals NHS Foundation Trust (UCLH), supported by the UCLH Charity, has placed the first UK order of the Biograph™ mMR system from Siemens Healthcare. The system, which is the same size as a typical 3T MRI scanner, is designed for the simultaneous acquisition of MR morphological and functional details in human tissue and PET cellular activity. It is the only system available on the market that combines the functionality of the two modalities into a single integrated solution.
The order by UCLH follows the global launch of the Biograph mMR at RSNA in December 2010. The system will be used for 60 percent routine clinical work and 40 per cent research projects. The overall aim is to increase the understanding, progress and diagnosis of diseases, plus unlock new patient treatment pathways.
When used for routine work, the system has the potential to reduce patient examination times as both MR and PET data can be obtained more quickly from sequential scans rather than separate examinations. With minimal motion artefacts, exact spatial recognition and low dose, high quality and accurate information can be gained from deep inside the human body.
The Biograph mMR will also benefit UCLH’s research programme. Plans are in preparation to explore the use of different biomarkers and therapeutic approaches. Professor Peter Ell, Senior Investigator of the NIHR comments, “PETMR will excel in defining the phenotype expression of the individual patient pathologies. Signal multiplexing with PET and MR will enhance our discriminatory power, expanding greatly the value of our clinical service to patients, whilst representing a major research opportunity in key neurodegeneration, oncology and cardiovascular entities.”
Lawrence Foulsham, Molecular Imaging Product Manager at Siemens Healthcare states, “The Biograph mMR is a pioneering, hybrid innovation that enables clinicians to gain detailed and quicker information from simultaneous scans. It is not a solution that incorporates two separate systems with image fusion afterwards, but an advanced integrated scanner that delivers fit-in-time images to ensure precise diagnosis, better therapy planning and more effective follow up treatment.”
Gunter Dombrowe, head of the Siemens Healthcare Sector for North West Europe, said “UCLH is a global pioneer in placing one of the very first orders for this highly innovative system. We are proud to be working with a first class clinical and research institution to expand the understanding of the cause and spread of disease, develop and validate new diagnostic approaches for better disease management and outcomes, as well as enhance departmental efficiency and productivity.”
The Biograph mMR has recently received a CE mark acknowledging that it meets EU safety, clinical benefit and environmental protection requirements. The system at UCLH is due to be operational in Spring 2012.