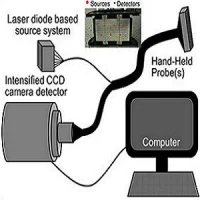
MAMMOCARE's new breast biopsy system is guided by 3D real-time PEM (positron emission mammography) imaging, offering real-time guidance and correction of the needle trajectory for higher accuracy and efficiency in tumour sampling. It is the first breast biopsy system in the market offering real-time monitoring and guidance, according to researchers involved in the project.
As José Luis Peris, a researcher from Biomechanics Institute of Valencia (IBV), Spain, explained: “Realtime monitoring and control of biopsy procedure, together with the high precision mechanics of the biopsy positioning module assure an accurate sampling of the target lesion as well as shortening the procedure with regard to current techniques”.
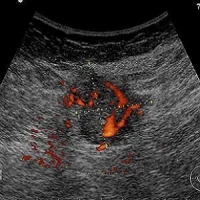
Early diagnosis of breast cancer is essential to reduce the risk of mortality. First diagnostic tool is clinical imaging (mainly mammography and ultrasound), but suspicious findings usually require a biopsy to confirm the diagnosis. Data show that 2 percent of women who undergo a screening mammogram will require some type of breast biopsy — 1.3 million women each year in Europe.
Apart from ultrasound-guided biopsy, which is a manual procedure with limited indications, all current solutions are based on discrete scans of static images taken at different steps during the procedure. This guidance method makes current biopsy techniques to be long procedures with limited accuracy. To improve these procedures, MAMMOCARE has developed the PEM-guided system that automatically calculates the best needle position to perform the biopsy, and it places and inserts accordingly for the physician to extract the selected sample.
MAMMOCARE is a European project funded by the Research Executive Agency, through the research for the benefit of SMEs programme, under the 7th Framework Programme of the European Union.
Clinical feasibility of the new breast biopsy system has been assessed through a clinical validation study at the facilities of Stichting Het Nederlands Kanker Instituut-Antoni Van Leeuwenhoek Ziekenhuis (Netherlands). The European NHS could save up to 7.6 million euros per year using the innovative biopsy system.
The MAMMOCARE project is led by Oncovision (Coordinator, Spain), Statice (France) and Virtual Angle (Netherlands), three companies with extensive experience in PEM imaging technology, mechatronics applied to health, and development of software products. The research and development activities have mainly been developed by the Institute of Instrumentation for Molecular Image (I3M), the Higher Council for Scientific Research (CSIC) Agency, the IBV (Spain), and IK Health & Environment Research Institute (UK).
Source and image credit: Asociación RUVID
As José Luis Peris, a researcher from Biomechanics Institute of Valencia (IBV), Spain, explained: “Realtime monitoring and control of biopsy procedure, together with the high precision mechanics of the biopsy positioning module assure an accurate sampling of the target lesion as well as shortening the procedure with regard to current techniques”.
Early diagnosis of breast cancer is essential to reduce the risk of mortality. First diagnostic tool is clinical imaging (mainly mammography and ultrasound), but suspicious findings usually require a biopsy to confirm the diagnosis. Data show that 2 percent of women who undergo a screening mammogram will require some type of breast biopsy — 1.3 million women each year in Europe.
Apart from ultrasound-guided biopsy, which is a manual procedure with limited indications, all current solutions are based on discrete scans of static images taken at different steps during the procedure. This guidance method makes current biopsy techniques to be long procedures with limited accuracy. To improve these procedures, MAMMOCARE has developed the PEM-guided system that automatically calculates the best needle position to perform the biopsy, and it places and inserts accordingly for the physician to extract the selected sample.
MAMMOCARE is a European project funded by the Research Executive Agency, through the research for the benefit of SMEs programme, under the 7th Framework Programme of the European Union.
Clinical feasibility of the new breast biopsy system has been assessed through a clinical validation study at the facilities of Stichting Het Nederlands Kanker Instituut-Antoni Van Leeuwenhoek Ziekenhuis (Netherlands). The European NHS could save up to 7.6 million euros per year using the innovative biopsy system.
The MAMMOCARE project is led by Oncovision (Coordinator, Spain), Statice (France) and Virtual Angle (Netherlands), three companies with extensive experience in PEM imaging technology, mechatronics applied to health, and development of software products. The research and development activities have mainly been developed by the Institute of Instrumentation for Molecular Image (I3M), the Higher Council for Scientific Research (CSIC) Agency, the IBV (Spain), and IK Health & Environment Research Institute (UK).
Source and image credit: Asociación RUVID
Latest Articles
healthmanagement, breast cancer, ultrasound, breast biopsy, positron emission mammography, European Union
MAMMOCARE's new breast biopsy system is guided by 3D real-time PEM (positron emission mammography) imaging, offering real-time guidance and correction of the needle trajectory for higher accuracy and efficiency in tumour sampling. It is the first breast