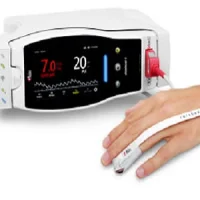
Masimo Rad-97™ Pulse CO-Oximeter® (center), Rad-97 with Integrated Noninvasive Blood Pressure (left), and Rad-97 with Integrated NomoLine™ Capnography (right)
Masimo has announced FDA 510(k) clearance and full market release of Rad-97™ Pulse CO-Oximeter®,including configurations with integrated NomoLine™ capnography and noninvasive blood pressure (NIBP) measurement from SunTech Medical. Rad-97 offers Masimo noninvasive and continuous monitoring, through Measure-through Motion and Low Perfusion™ SET® pulse oximetry and upgradeable rainbow®technologies, in a compact, standalone monitor that incorporates advanced customizability, connectivity, and device integration capabilities.
“We’re excited to bring the easy-to-use, compact Rad-97 to the US,” said Joe Kiani, Founder and CEO of Masimo. “Rad-97 brings together our core SET® and rainbow® technologies with advanced, workflow-enhancing connectivity solutions. We believe it will be an indispensable addition to many healthcare environments, including lower-acuity settings in hospitals, alternate care, and telehealth.”
Rad-97 combines its portable, compact form factor with a high-resolution, multi-touch color display that allows clinicians to easily customize the device for each monitoring use case – bringing rainbow SET™ measurements to care areas where a small footprint or high portability is desired. Users can also rapidly configure the device to accommodate different patient populations using customizable profiles. A rechargeable battery lasting approximately four hours allows Rad-97 to be used in situations where extended operation without access to AC power is needed. An optional roll stand allows for tetherless device transport, offering additional flexibility in situations where space is limited.
Rad-97 features built-in enterprise WiFi capability, allowing it to connect wirelessly to supplemental patient monitoring systems including Masimo Patient SafetyNet™*, facilitating automatic data transfer to hospital electronic medical record (EMR) systems. The easy-to-use, intuitive interface helps to simplify charting workflows for vital sign monitoring and patient data capture. Rad-97 is also compatible with existing nurse call systems. Data from extended monitoring sessions, such as sleep studies, can be rapidly downloaded via USB, Ethernet, or WiFi.
Rad-97 also supports point-to-point Bluetooth® wireless connections with compatible devices, such as thermometers, glucometers, and weight scales, allowing patient data to be seamlessly transferred to Rad-97 and connected upstream systems. Rad-97 will also be available with an optional camera that will provide a high-resolution video feed, as well as audio, to the Patient SafetyNet view-station. The camera-equipped Rad-97 will allow patients and clinicians to communicate remotely with compatible Patient SafetyNet software, making it well-suited as a point-of-care device for telehealth.
Rad-97 with capnography features an integrated ISA™ CO2 module with NomoLine sampling lines for sidestream capnography, with an adapter for intubated patients – meeting continuous monitoring and capnography needs in a single device. Rad-97 with capnography displays continuous end-tidal carbon dioxide (EtCO2) monitoring with numeric, trend, and waveform viewing options, as well as fractional concentration of inspired carbon dioxide (FiCO2) and respiration rate. NomoLine capnography reduces delays in respiration rate measurement in both low and high ranges, accurately measures respiration rate, and reduces the incidence of filter line occlusions, as a result of NomoLine moisture wicking technology.
With Rad-97 NIBP, oscillometric blood pressure is available in three NIBP measurement modes: spot-check, automatic interval (which measures blood pressure routinely, at a desired interval), and stat interval (which continually measures blood pressure for a desired duration). An integrated port allows clinicians to connect a blood pressure cuff inflation hose directly to Rad-97; the port is compatible with both disposable and reusable cuffs, for adult, pediatric, and neonatal patients.
*The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
Source: Masimo