ICU Management & Practice, ICU Volume 13 - Issue 4 - Winter 2013/2014
Authors
David Pearson,MB BChir, MA (Cantab), FCICM
Staff Specialist, Intensive Care Unit, Gold Coast University Hospital, Southport, Queensland, Australia
Yoshiro Hayashi, MD, PhD
Director of Intensive Care Medicine, Kameda Medical Center, Kamogawa, Japan Honorary Associate Professor,
UQ Centre for Clinical Research, The University of Queensland Brisbane, Queensland, Australia
Brent Richards, FRACP FCICM
Director, Intensive Care, Gold Coast University Hospital, Southport, Queensland, Australia Honorary Associate Professor,
Bond University, Robina, Queensland, Australia
Jeffrey Lipman,MBBCh, MD
Professor and Head, Anesthesiology and Critical Care
The University of Queensland Brisbane, Queensland, Australia Director, Intensive Care, Royal Brisbane and Women’s Hospital
Herston, Queensland, Australia Director, Burns, Trauma and Critical Care Research Centre The University of Queensland,
, Queensland, Australia
Background
The incidence of ventilator-associated pneumonias (VAPs) has long been considered the reference benchmark for guiding continuous quality improvement in mechanically ventilated patients. In recent times, however, its validity as a tool for such surveillance has been called into question. This article will discuss the evolution of ventilator-associated pneumonia into the ventilator-associated event (VAE) and the importance of now validating this tool for use in both internal and external quality assurance processes.
Ventilator-Associated Pneumonias
A VAP is diagnosed when a mechanically ventilated patient satisfies certain systemic, clinical and pulmonary criteria (Horan et al 2008). Within these diagnostic criteria scope for subjectivity exists, resulting in an algorithm favouring sensitivity over specificity for VAP diagnosis. As the treatment of VAPs is antibiotics, the knock-on implications for this lack of specificity, in terms of antimicrobial stewardship, are clear. There remains no gold standard for VAP diagnosis in vivo, and, as a consequence, VAP prevalence is very difficult to quantify accurately. Evidence from prevention-targeted randomised controlled trials would suggest that this figure lies somewhere between 16% and 21% (Lorente et al 2012; Rello et al 2002; Rello et al 2013; Barbier et al. 2013; Melsen et al. 2013). Compounding this baseline variation is a lack of objectivity within some of the diagnostic criteria, reflected both in a widespread inter-observer variability in VAP diagnosis (Klompas 2008), and in post-mortem studies revealing that as many as half of all cases are misdiagnosed (Tejerina et al. 2010). Mathematical modelling has also been able to demonstrate that the prevalence of other pulmonary conditions will affect the rate of VAP diagnosis, despite a constant, fixed VAP incidence (Klompas et al. 2008).
Several bodies have attempted to standardise and increase the specificity of diagnostic criteria (Lorente et al. 2012; Torres et al. 2009; Guidelines 2009), of which the Centers for Disease Control and Prevention (CDC)’s version is the most widely utilised. Studies performed at the CDC’s prevention epicentres by Klompas and colleagues (Klompas et al, 2011) advanced this by replacing subjective criteria with objective, quantifiable data for pulmonary deterioration where possible. They then applied this modified VAP definition to retrospective data, and were able to demonstrate improved capacity to predict ‘hard’ outcomes of duration of mechanical ventilation, ICU length of stay and mortality when compared with their traditional VAP definition. Although clearly demonstrating an association between objective data and clinically relevant outcomes, this revised tool did not increase specificity for VAP diagnosis.
Importance of VAP
For the very reasons outlined above, ascribing accurate attributable mortality to VAP is fraught with confounding issues. A recent meta-analysis concluded that the overall attributable mortality of VAP is 13% (Melsen et al 2013). Original patient data were taken from 24 randomised controlled trials assessing a broad range of VAP prevention techniques. VAP incidence was most commonly the primary outcome rather than mortality. Acknowledging that no gold standard exists for VAP diagnosis, the authors grouped included studies into categories, depending upon whether invasive specimens were required as part of the diagnostic key or not, thus allowing for regional variation in practice. Noteworthy here is an Australian-wide study showing little if any use of bronchoalveolar lavage in the diagnosis of VAP (Boots et al. 2005). As suspected, there was a variation between subgroups with higher rates for surgical patients and patients with mid-range severity as expressed by acute physiology and chronic health evaluation (APACHE) and simplified acute physiology score (SAPS) at admission (Melsen et al. 2013). The authors of the meta-analysis concluded that the predominant cause of this increased risk of dying was the prolonged exposure to intensive care therapies (Melsen et al. 2013).
Quality Assurance
Any quality assurance marker must be evidencebased, clinically relevant and have optimal sensitivity and specificity. In addition, when used as a surveillance tool, it must be sufficiently common and preventable to have a demonstrable impact upon morbidity and mortality. As a subdivision of nosocomial infection, VAPs fulfill some of these criteria, but what is not often reported is the non-modifiable contribution to their aetiology. As an example, it would be hard to believe that an elective post surgical patient has the same baseline risk profile as a complicated medical patient; so it would not be fair to draw conclusions on standards of underlying care based solely on VAP incidence. Further emphasising the unreliability of this data was a study by Klouwenberg and colleagues (2013), who were able to demonstrate concordance in diagnosis of just 35%, for the same patient cohort, between different personnel responsible for surveillance reporting. As a tool for internal audit, if confounders of personnel and diagnostic criteria can be controlled for, then VAP surveillance may be of use, but when the generated data is used for external inter-ICU comparison then the impact of confounding bias is too great. Many interventions and ‘bundles’ have targeted VAP prevention (Melsen et al. 2013; Bouadma et al. 2012), with subsequent falls in VAP rates. However, a failure to reliably improve upon ‘hard’ clinical outcomes such as length of stay and mortality would suggest that in addition to the intervention, surveillance artifact may be present. Although this discordance between VAP rate lowering and static mortality rates may just relate to a failure to power for these outcomes, a more sinister aetiology may be true: in an era where financial disincentives are applied to perceived poor clinical performance, some hospital administrators have been accused of ‘playing’ the system to avoid penalty (Magill and Fridkin 2012). In the UK, the NHS has placed the responsibility for data integrity firmly at the feet of clinicians, advocating for firm clinician engagement in developing quality assurance tools. In turn, this has led to some campaigning for the abandonment of VAP incidence as a quality assurance tool in favour of newer markers (Shorr and Zilberberg 2012).
In regards to ventilator-associated events, whilst VAPs may be the most frequently documented they are far from the only complication of mechanical ventilation. Barotrauma, atelectasis and pulmonary oedema could all be considered to be common, preventable complications independently associated with poor clinical outcomes. For this reason, their incidence should have a role to play in benchmarking and subsequent quality improvement.
Thus, criticism can be grouped into three broad categories: i) the poor specificity for VAP of commonly used diagnostic tools ii) a multi-factorial lack of concordance in diagnosis between surveillance personnel, and iii) the lack of importance placed upon other, highly morbid, complications of mechanical ventilation in quality control initiatives.
Ventilator-Associated Events and Ventilator-Associated Conditions
Klompas et al. explored the feasibility of purely objective diagnostic surveillance criteria for VAP (Klompas et al. 2012). Thirty-two different candidate definitions were created, composed of different combinations of the following signs: i) three thresholds for respiratory deterioration defined by sustained increases in daily minimum positive end-expiratory pressure or FiO2 after either 2 or 3 days of stable or decreasing ventilator settings, ii) abnormal temperature iii) white blood cell counts iv) purulent pulmonary secretions defined by neutrophils on Gram stain, and v) positive cultures for pathogenic organisms. They concluded that only definitions requiring objective evidence of respiratory deterioration, detected through documentation of alterations in respiratory support, were significantly associated with increased hospital mortality. Crucially, placing these alterations on the first level of a novel diagnostic key would enable tracking of not only VAPs but also clinically significant non- VAP complications of mechanical ventilation. Collectively, these complications would be termed Ventilator-Associated Events (VAEs). As a surveillance tool, previously privileged pathologies would now be trapped and available for quality control.
Accordingly, in 2013 the CDC National Healthcare Safety Network (NHSN) introduced VAE surveillance, a novel tool for monitoring mechanically ventilated patients. Designed to replace VAP surveillance, VAE monitoring would provide objective, reproducible data tracking all complications of mechanical ventilation leading to an alteration in ventilator settings and an increase in respiratory support. This restratification comprises a three tiered mode (see Figure 1). Entrance into the VAE surveillance tool is through detection of an increase in daily minimum PEEP or FiO2, objective data that can be reliably trapped by clinical information systems. In this way, all patients with clinically significant pulmonary complications are identified and their data recorded, irrespective of whether a VAP or non-VAP is causative. At this level they are referred to as Ventilator-Associated Conditions (VACs). The second tier, infection-related ventilator-associated complications (IVAC), is reached when VAC criteria are complemented by: i) traditional SIRS (systemic inflammatory response syndrome) signs of leucocytosis and abnormal temperature, and ii) the commencement of antimicrobials. Progression to the third tier occurs when evidence exists of a pulmonary source of infection, a ‘possible’ pneumonia being differentiated from ‘probable’ pneumonia on the basis of at least semi-quantitative pathogenic organism culture in the latter.
According to CDC NHSN, the uppermost two tiers, namely VAC and IVAC, are designed to be suitable for use in potential future public health reporting, inter-facility comparisons and payfor- performance programmes. Requiring only objective data confers adequate external validity for this purpose and will also enable patterns of antimicrobial prescribing to be compared more reliably. Possible and probable VAP diagnosis would be utilised within internal quality control systems to allow for the variation in practice in obtaining the necessary respiratory specimens to confirm diagnosis.
Validation
Hayashi and colleagues performed a retrospective evaluation of VAC (Hayashi et al. 2013). One of the aims of the study was to capture commonly recorded prospectively documented objective data (FiO2, PEEP). They were able to demonstrate that VAC events were associated with both increased length of stay in ICU and increased days of mechanical ventilation. In addition to these negative outcomes, Klompas et al. (2011) were able to demonstrate an increase in mortality associated with VAC diagnosis compared with traditional VAP criteria. Therefore, a prospective study with a larger sample size is required to evaluate the utility of VAC and IVAC surveillance before its implementation in Australian ICUs. The introduction of a common clinical information system platform in South East Queensland will help to facilitate more efficient multi-site data collection and collaboration for this purpose.
Summary and Conclusion
At the bedside there is no doubt that there is sporadically a need to treat respiratory tract infections in patients on a ventilator with antibiotics. Exactly when this is required and how this is defined is being questioned, as there is not a uniform, unequivocal diagnostic set of criteria for the diagnosis of VAP. This makes comparisons, incidences and outcomes difficult to compare and to track. Especially as regards a quality indicator, objective, commonly captured data for all patients on ventilators is a more logical way forward. VAC has been put forward to address this concern. All patients on a ventilator will have FiO2, PEEP and PaO2 regularly recorded, and with clinical information systems now permitting realtime tracking and recording of incremental changes in these parameters we believe VAC will replace VAP as a quality assurance tool. The incorporation of antimicrobial use into the IVAC tier will also permit more reliable comparisons of stewardship models, and by differentiating between probable and possible VAPs, the final tier of the VAE key will also allow for local variations in diagnostic specimen sampling.
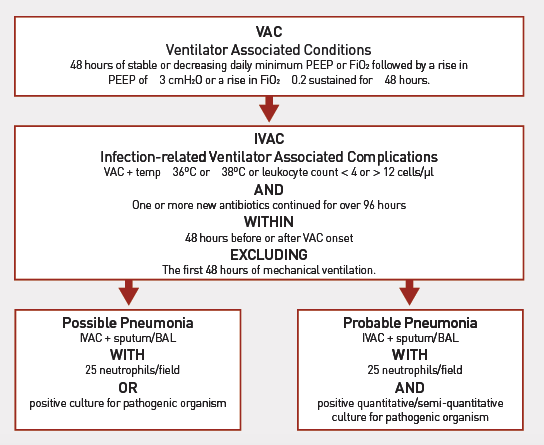
Figure 1: Diagnostic key for Ventilator-Associated Events
References:
Boots RJ, Lipman J, Bellomo R et al. (2005). The spectrum of practice in the diagnosis and management of pneumonia in patients requiring mechanical ventilation. Australian and New Zealand practice in intensive care (ANZPIC II). Anaesth Intensive Care, 33(1): 87-100.
Bouadma L, Wolff M, Lucet JC (2012). Ventilator-associated pneumonia and its prevention. Curr Opin Infect Dis, 25(4): 395-404.
American Thoracic Society and Infectious Diseases Society of America (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med, 171(4): 388-416.
Hayashi Y, Morisawa K, Klompas M et al. (2013). Toward improved surveillance: the impact of ventilator-associated complications on length of stay and antibiotic use in patients in intensive care units. Clin Infect Dis, 56(4):471-7.
Horan TC, Andrus M, Dudeck MA. (2008). CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting Am J Infect Control, 36(5): 309-32.
Klompas M, Kulldorff M, Platt R. (2008). Risk of misleading ventilator-associated pneumonia rates with use of standard clinical and microbiological criteria. Clin Infect Dis, 46(9):1443-6.
Klompas M. (2010). Interobserver variability in ventilator-associated pneumonia surveillance. Am J Infect Control, 38(3):237-9.
Klompas M, Khan Y, Kleinman K et al.(2011). Multicenter evaluation of a novel surveillance paradigm for complications of mechanical ventilation. PLoS ONE, 6(3):e18062.
Klompas M, Magill S, Robicsek A et al.(2012). Objective surveillance definitions for ventilator-associated pneumonia. Crit Care Med, 40(12):3154-61.
Klouwenberg et al. (2013). Interobserver agreement of Centers for Disease Control and Prevention criteria for classifying infections in critically ill patients. Crit Care Med, 41(10): 2373-8.
Lorente L, Lecuona M, Jiménez A et al. (2012). Ventilator-associated pneumonia with or without toothbrushing: a randomized controlled trial. Eur J Clin Microbiol Infect Dise, 31(10): 2621-9.
Magill SS and Fridkin SK. (2012). Improving surveillance definitions for ventilator-associated pneumonia in an era of public reporting and performance measurement. Clin Infect Dis, 54(3): 378-80.
Melsen WG, Rovers MM, Groenwold RH et al. (2013). Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis, 13(8): 665-71.
Rello J, Ollendorf DA, Oster G et al. (2002). Epidemiology and outcomes of ventilator-associated pneumonia in a large US database. Chest, 122(6): 2115-21.
Rello J, Afonso E, Lisboa T et al.(2013). A care bundle approach for prevention of ventilator-associated pneumonia. Clin Microbiol Infect, 19(4): 363-9.
Shorr AF and Zilberberg MD. (2012). Nature (and the ICU) abhors a VACuum. Chest, 142(6):1365-6.
Tejerina E et al. (2010). Accuracy of clinical definitions of ventilator-associated pneumonia: comparison with autopsy findings. J Crit Care, 25(1): 62-8.
Torres A, Ewig S, Lode H et al. (2009). Defining, treating and preventing hospital acquired pneumonia: European perspective. Intensive Care Med, 35(1):9-29.





