ICU Management & Practice, Volume 20 - Issue 4, 2020
An overview of the clinical application and future perspective of the Seraph® 100 haemoperfusion in critically ill patients with sepsis.
Background
Over the last few decades, sepsis has been spreading worldwide to such an extent that it has been declared global health priority by the World Health Organization (Reinhart et al. 2017). Sepsis is an old disease (Cutuli et al. 2017) and many efforts have been exerted by the scientific community in order to define this syndrome (Singer et al. 2016) and drive its management by evidence-based recommendations (Rhodes et al. 2017). Early identification and few, timely, interventions have been shown effective to improve sepsis-related clinical outcomes (Seymour et al. 2017). Rapid administration of appropriate antimicrobials and control of source infection are necessary to treat sepsis (Rhodes et al. 2017). However, the heterogeneous characteristics of this syndrome (mostly associated with patients’ inflammatory response to infection and comorbidities), the emergence of multi-drug resistant pathogens (Vincent et al. 2020), and the paucity of new discoveries in this setting (Laffey and Kavanagh 2018), have been challenging critical care clinicians. In the light of this view, extracorporeal blood purification by Seraph® 100 Microbind Affinity Blood Filter (Seraph-100, Exthera Medical Corporation, Martinez, CA), offers the possibility to remove bacteria, viruses and fungi from the systemic circulation (Seffer et al. 2020). Moreover, this newly designed membrane provides immune modulation by clearing the blood from inflammatory mediators, thus controlling both pathogen load and host response. Accordingly, Seraph® 100 Microbind Affinity Blood Filter represents a new frontier in the management of sepsis, prefiguring itself as a bridge to setup appropriate antimicrobial therapy as well as provide definitive control of source infection.
In the present paper we will report structural features, laboratory and clinical evidences supporting the application of Seraph® 100 Microbind Affinity Blood Filter in critically ill patients with sepsis. Finally, we will point out future perspectives in this field.
Structural Features
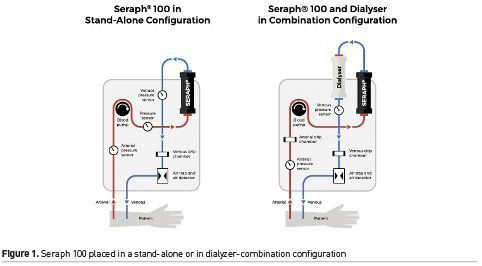
The Seraph® 100 Microbind Affinity Blood Filter is a single use, sterile, disposable column, made by polyethylene beads (diameter of 0.3 mm) which surface has been modified to contain end-point attached heparin (Seffer et al. 2020). The amount of heparin released to the systemic circulation during extracorporeal blood purification is negligible. Heparin mimics negatively charged heparan sulfates of cell surface. This feature fosters electrostatic interactions with many micro-organisms, inflammatory mediators and drugs, thus allowing their removal from the blood (Seffer et al. 2020). The Seraph® 100 Microbind Affinity Blood Filter is an extracorporeal haemoperfusion device, which is intended for use with standard, commercially available bloodlines compatible with the pump system used. When concomitant renal replacement therapy is performed, Seraph® 100 Microbind Affinity Blood Filter should be placed prior to the haemofilter (Figure 1). A large bore (12-13 French), double-lumen, venous catheter is required in order to deliver up to 400 mL/min blood flow rate.
Laboratory Evidence
Pathogens
Preclinical investigations showed that the Seraph® 100 Microbind Affinity Blood Filter clears the bloodstream from pathogens, inflammatory mediators and drugs. Specifically, pathogens are restrained into the column through electrostatic interactions with the negatively charged heparin graft of the Seraph® 100 Microbind Affinity Blood Filter. Among bacteria, Staphylococcus Aureus was demonstrated to adhere to heparinised beads (Mattsby-Baltzer et al. 2011). This pathogen represents one of the most common causes of infections among critically ill patients and recent epidemiological reports described an increasing incidence of methicillin-resistant strains (Vincent et al. 2020), which implies the risk of prescribing inappropriate empiric antibiotic therapy and worsen patients’ related clinical outcome. Accordingly, early removal of Methicillin-resistant Staphylococcus Aureus by Seraph® 100 Microbind Affinity Blood Filter may represent an effective complementary treatment to control the development of infection, especially when empiric antibiotic therapy is not appropriate and the spectrum of sensitivity to antibiotics is unknown.
Moreover, recent laboratory investigations showed that Seraph® 100 Microbind Affinity Blood Filter reduces bloodstream viral load of Zika virus (87%), Cytomegalovirus (79%), and Adenovirus (62%). Furthermore, previous evidences observed that the surface protein “Spike 1” of Sars-CoV-2 virus binds heparin and unpublished researches demonstrated that such pathogen is removed by the Seraph® 100 Microbind Affinity Blood Filter (Seffer et al. 2020).
Inflammatory mediators
Cytokines may be theoretically adsorbed by the Seraph® 100 Microbind Affinity Blood Filter. In an in vitro research, a significant reduction of tumour necrosis factor alpha concentration (which approximates 59%) was observed when blood from donors was exposed to heparin beads (Seffer et al. 2020).
Drugs
Schmidt et al. (2020) investigated the in vitro adsorptive properties of the Seraph® 100 Microbind Affinity Blood Filter on 18 anti-infective drugs (acyclovir, amphothericin B, ceftazidime, cefazolin, clindamycin, daptomycin, fluconazole, fosfomycin, gentamicin, levofloxacin, linezolid, meropenem, moxifloxacin, piperacillin, rifampicin, tazobactam, tobramycin and vancomycin) added to human donor plasma. The authors demonstrated that extracorporeal haemoperfusion with this device reduced the concentration of gentamycin and tobramycin within the first five minutes of treatment. Although plasma level concentrations of the remaining drugs were also reduced within the same time-point, the variation was smaller compared to aminoglycosides. Moreover, no additional drug plasma level changes were observed at 60 minutes from the commencement of haemoperfusion for any of the investigated antimicrobials.
Clinical Evidence
The safety of Seraph® 100 Microbind Affinity Blood Filter was demonstrated in 15 patients undergoing renal replacement therapy. In this study, a reduction of bacteria load (assessed by colony forming units/mL or time to positivity of blood cultures) in the bloodstream was observed within the first 4 hours of treatment. More recently, Olson et al. (2020) reported their preliminary experience with the Seraph® 100 Microbind Affinity Blood Filter in two critically ill patients with Sars-CoV-2 infection requiring mechanical ventilation and vasopressor support. The authors observed haemodynamic parameters improvement, vasopressor load reduction and body temperature modulation associated with this treatment (Olson et al. 2020).
Future Perspectives
The Seraph® 100 Microbind Affinity Blood Filter use has been widely increasing and many studies have started with the aim to collect further evidences on the application of this device in clinical practice. Currently, the research agenda includes four trials on this topic registered in ClinicalTrials.gov (Identifier: NCT04361500, NCT04413955, NCT04547257, NCT04260789). A prospective, multicentre, open-label, randomised, controlled clinical investigation has been recently commenced, with the aim to evaluate the safety and performance of Seraph® 100 Microbind Affinity Blood Filter in the reduction of pathogen load from the blood in septic patients with suspected, life-threatening bloodstream infection (primary endpoint). This trial will collect evidence on 232 adult critically ill patients, evaluating also 90 days all-cause mortality, persistence/recurrence of bacteraemia, sepsis, organ dysfunction-free days, reduction of intensive care unit complications, ventilator-free days, length ICU and hospital stay within 7 days from the enrollment in the trial.
Conclusion
The Seraph® 100 Microbind Affinity Blood Filter is a newly designed device for extracorporeal blood purification, which offers promising clinical perspectives in the management of sepsis. This device may become of paramount importance in the era of multidrug resistant pathogens, giving time to the clinician in order to prescribe effective antimicrobial therapy, identify and resolve the source of infection and modulate inflammatory-mediated organ dysfunction, thus controlling the development of sepsis and improving patients’ related outcome.
References:
Cutuli S, Pascale GD, Antonelli M (2017) 'σήψις' yesterday, sepsis nowadays: what's changing? J Thorac Dis, 9(2):E166-E167.
Laffey J, Kavanagh B (2018) Negative trials in critical care: why most research is probably wrong. Lancet Respir Med, 6(9):659-660.
Mattsby-Baltzer I, Bergstrom T, McCrea K et al. (2011) Affinity apheresis for treatment of bacteremia caused by Staphylococcus aureus and/or methicillin-resistant S. aureus (MRSA). J Microbiol Biotechnol, 21(6):659-664.
Olson S, Oliver J, Collen J et al.(2020) Treatment for Severe Coronavirus Disease 2019 With the Seraph-100 Microbind Affinity Blood Filter. Crit Care Explor, 2(8):e0180.
Reinhart K, Daniels R, Kissoon N et al. (2017) Recognizing Sepsis as a Global Health Priority - A WHO Resolution. N Engl J Med, 377(5):414-417.
Rhodes A, Evans L, Alhazzani W et al. (2017) Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med, 43(3):304-377.
Schmidt J, Eden G, Seffer M et al. (2020) In vitro elimination of anti-infective drugs by the Seraph ® 100 Microbind ® affinity blood filter. Clin Kidney J, 13(3):421-424.
Seffer M, Cottam D, Forni L et al. (2020) Heparin 2.0: A New Approach to the Infection Crisis. Blood Purif. Online ahead of print:1-7.
Seymour C, Gesten F, Prescott H et al. (2017) Time to Treatment and Mortality during Mandated Emergency Care for Sepsis. N Engl J Med, 376.(23):2235-2244.
Singer M, Deutschman C, Seymour C et al. (2016) The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3).JAMA, 315(8):801–810.
Vincent J, Sakr Y, Singer M et al. (2020) Prevalence and Outcomes of Infection Among Patients in Intensive Care Units in 2017. JAMA, 323(15):1478-1487.









