ICU Management & Practice, ICU Volume 8 - Issue 3 - Autumn 2008
Author
Biomarkers, Namely CRP and PCT, can Help Clinicians at the Bedside Since when Used in Combination with the Clinical Examination they Add Useful Information in the Diagnosis of Infection.
Introduction
In clinical practice a biomarker can serve several objectives. In a quick review of the published literature, it is easily perceived that a large number of studies assess the value of biomarkers in the prognosis of infection and sepsis. Though, one could wonder, what the point is at the bedside, of utilising a prognostic biomarker. Ascertaining if a particular patient has a high risk of dying, is pointless especially when the attending physician can do nothing to change that prognosis! However, biomarkers can provide additional information to the clinical evaluation namely in the diagnosis of infection, risk stratification, assessment of the response to antibiotic therapy and finally in antibiotic stewardship (Povoa et al. 2002; Povoa et al. 2008). In this review we are going to discuss the role of biomarkers in the diagnosis of infection.Biomarkers in the Diagnosis of Infection
Physicians are often faced with the following questions; whether a patient is infected or not; what is the primary source of infection and what is/are the infecting organisms as well as their sensitivity patterns. However, non-infectious clinical situations can be associated with a sepsis-like syndrome that makes sepsis diagnosis even more difficult. Consequently, antibiotics are frequently prescribed without a definite diagnosis of sepsis because of the fear of missing an infection.The correct diagnosis represents our ability to identify in a target population those with the disease. A diagnostic test is something that indicates that a particular clinical condition will occur. The quality of the information provided by a diagnostic test, and consequently its usefulness, depends of the accuracy with which it identifies the target disorder. Since the ideal biomarker has not yet been identified, clinicians should know their limitations and strengths, namely sensitivity, specificity, areas under the receiver operating characteristics (AUC) curve and finally, the likelihood ratios (LR) (Table 1) obtained from high-quality studies.

Table1+LR – positive likelihood ratio; −LR – negative likelihood ratio; AUC – area under the ROC curve
From a very large number of investigations on biomarkers I will select three, C-reactive protein (CRP) and procalcitonin (PCT), probably the more studied biomarkers, and finally, soluble triggering receptor expressed on myeloid cells-1 (sTREM–1), recently proposed.Plasma CRP rises whenever an inflammatory process is present and, characteristically, its serum concentration depends only on the intensity of the stimulus and on the rate of synthesis (Povoa et al. 2002). Conflicting results have been reported from studies assessing the value of CRP as a diagnostic marker of infection, which are a consequence from differences in inclusion criteria (Simon et al. 2004; Tang et al. 2007). However, using more strict inclusion criteria CRP has been shown to be a valuable marker of infection. Recently our group showed that CRP of infected patients (N=76) was significantly higher than that of controls (N=36) (Povoa et al. 2005). A CRP concentration > 8.7 mg/dl was associated with infection with sensitivity of 93.4% and specificity of 86.1% (AUC 0.93; positive LR 6.71 and negative LR 0.08). Interestingly, Sierra et al., using similar inclusion criteria (infected N=70; non-infected N=80), came to almost identical findings (cut-off 8.0 mg/dl; AUC 0.94; positive LR 7.41 and negative LR 0.065) (Sierra et al. 2004). A recent study showed that the appropriateness of antibiotic therapy was associated with a reduction of tracheal bacterial load in monomicrobial ventilator associated pneumonia (VAP) (N=168) (Lisboa et al. 2008). Even though, diagnosis of infection was not the primary aim of this study, it nicely demonstrated a good correlation between CRP values and tracheal bacterial load indicating again that CRP could be used as a surrogate marker of infection.
Procalcitonin is classified as hormokine since it has simultaneously hormone and cytokine properties (Christ-Crain et al. 2007). In a classic paper, in a paediatric patient population (N=79), PCT was much higher in infected children and severe bacterial infections elicit much higher PCT concentrations than viral or localized infections (Assicot et al. 1993). However, the cut-off of PCT for infection diagnosis has not yet been definitively established (de Werra et al. 1997; Ugarte et al. 1999) due, at least in part, to the diverse inclusion criteria (Cohen et al. 2001; Povoa et al. 2008). Using strict criteria, namely the presence of documented infection (N=111) versus no infection and no antibiotic therapy (N=79), Ugarte et al. found that the AUC of PCT for diagnosis of infection was 0.66 (positive LR 1.74; negative LR 0.53) (Ugarte et al. 1999). In patients undergoing cardiac surgery, Aouifi et al. found that, after cardiopulmonary bypass (N=36), PCT increase to levels above 1 ng/ml irrespective of the type of surgery and that a PCT > 5 ng/ml was suggestive of complications (N=10) either of infectious or non-infectious origin (N=7) (Aouifi et al. 1999). In another study (Aouifi et al. 2000), the same group found that patients with postoperative infectious complications showed PCT values significantly elevated (positive LR 17; negative LR 0.16). However, patients with mediastinis (N=9) presented similar PCT values to the non-infected group, 0.8 ± 0.58 ng/ml and 0.41 ± 0.36 ng/ml, respectively. Similarly, Luyt et al. in a study to assess the value of PCT kinetics in the evaluation of documented VAP course, found a good correlation with clinical severity. However, some patients presented undetectable PCT levels (Luyt et al. 2005).
The sTREM-1 is a member of the immunoglobulin family. In the original study, Gibot et al. measured sTREM-1 in bronchoalveolar-lavage fluid finding that it could be a very accurate marker in the diagnosis of pneumonia (Gibot et al. 2004). These results were reproduced by Determann et al. in 28 critically ill patients (AUC=0.829) (Determann et al. 2005). However, plasma sTREM-1 levels remained almost unchanged both in VAP as well as in controls. Phua et al. found that serum sTREM-1 was significantly elevated in 150 patients with community-acquired pneumonia, chronic obstructive pulmonary disease and asthma exacerbation (Phua et al. 2006). However, the AUC for serum sTREM-1, as a surrogate marker for the need of antibiotics in lower respiratory tract infections, was marginal, 0.77.
To overcome the limitations of a single biomarker some studies evaluated the relative accuracy of panels of biomarkers. Gaini et al. evaluated the accuracy of four biomarkers in 194 patients, in particular CRP and PCT, to identify patients with infection (Gaini et al. 2006). They found that CRP was the biomarker with the highest diagnostic performance, whereas PCT presented the lowest (AUC = 0.83 vs. 0.77, respectively). Similarly, Kofoed et al. evaluated six biomarkers, in particular CRP, PCT and sTREM-1, in the diagnosis of communityacquired sepsis (N=151, 96 with documented infection) (Kofoed et al. 2007). Again, CRP was found to have the best diagnostic performance for bacterial infection (AUC=0.81), well above the performance found for PCT (AUC=0.72) and sTREM-1 (AUC=0.61).
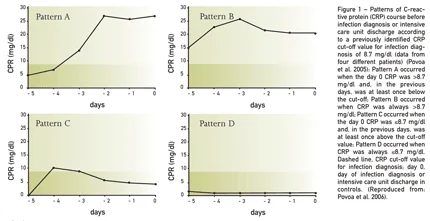
Biomarkers are not static but dynamic, presenting marked changes in response to different inflammatory stimulus, specifically, bacterial infections. Consequently serial measurements could be more informative than a single one. Our group showed that daily CRP monitoring could be useful in the early prediction of intensive care unit-acquired infections during the five days before infection diagnosis (Povoa et al. 2006). We identified four patterns of CRP course with different correlations with infection (Figure 1). A steady increase in and a persistently elevated CRP concentration (patterns A and B) were associated with high risk of infection. In contrast, a decrease in and continuously low CRP value (patterns C and D) were associated with low risk of infection. Furthermore, a maximum daily CRP variation >4.1 mg/dl was a good marker of infection prediction (sensitivity 92.1%, specificity 71.4%; positive LR 3.22 and negative LR 0.11), and, in combination with an absolute CRP concentration >8.7 mg/dl, its discriminative power increased even further (sensitivity 92.1%, specificity 82.1%). In a retrospective study (N=105), Vandijck et al. showed that both Gramnegative and Gram-positive bloodstream infections showed significant CRP increases in the days before diagnosis and that these changes were significantly higher with Gram-negatives (CRP d-2 to d+1 GPB vs GNB 3.1 vs 6.2 mg/dl, p=0.025) (Vandijck et al. 2007). In patients with suspected VAP, Luyt et al. evaluated changes in PCT in the five days before diagnosis (Luyt et al. 2007). The authors were unable to find any significant change both in concentration as well as delta PCT (positive LR 2.73; negative LR 0.69).