A critical care pharmacist’s perspective and advice on medication safety
around sedative and analgesic therapy in the ICU.
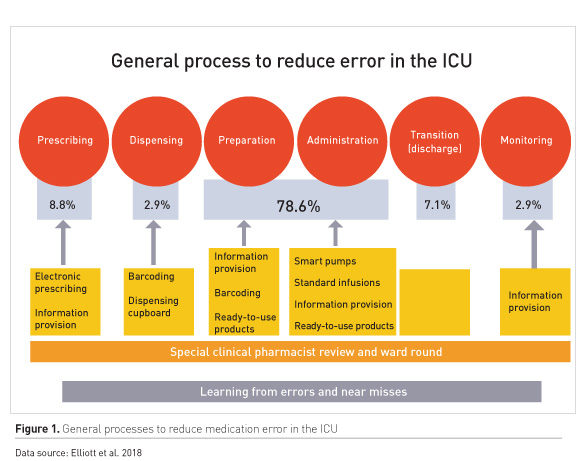
Medication errors occur at every stage of the drug therapy process. A recent report on medicines processes in English hospitals identified notably high error rates in prescribing (8.8%) and preparation and administration (78.6%) (Elliott et al. 2018).
Medication errors in ICU
ICU patients are at particular risk of errors around preparation and administration of intravenous therapies due to the high number of infused drugs. Furthermore, the nature of the ICU environment with the nurses often being interrupted at the bedside and the need for drug concentration calculations contributes to this risk. A multinational observational study found an error rate in parenteral drug administration of around 7% (Valentin et al. 2009). A similar ratio was highlighted in a single-centre observational study of ICU nurses, who knew they were being observed, and the author found an administration error rate of 6.6% (Tissot et al. 1999). A study in which nurses did not know they were being observed had an error rate of administration of 33%, excluding errors of wrong time (van den Bemt et al. 2002).
The 2009 24-hour observational study included 113 ICUs in 27 countries (Valentin et al. 2009). From 1328 patients and 12,000 medicines administrations 861 errors affecting 441 patients were reported. One-third of patients received one or more medication error, of which 19% had one error, and 14% had one or more error. Although most errors did not affect patient status, in 28% of cases medication errors led to temporary change in the patient status. Also, seven patients experienced permanent harm and five died. Looking at the involved drug categories, it appeared that 9% of administrations of sedatives and analgesics were associated with errors (181/2136); resulting in one death and one incident of permanent harm (Valentin et al. 2009). Another study, called the PROTECTED-UK study, analysed data from 21 ICUs from a 2-week period where pharmacists identified all their contributions to care, including noting errors, optimisations and consultations. Out of 20,517 prescriptions, 1 in 6 had such a contribution, and 1 in 15 prescriptions had an error (Shulman et al. 2015; Rudall et al. 2017). The data showed that 5.5% of all errors identified were around sedation and analgesia. Of contributions to care relating to sedation and analgesia (384/3294) 50% were errors, 45% optimisations and 5% consults (Shulman R, pers. comm.).
What can go wrong with sedation and analgesia in the ICU?
Potential errors include selecting the wrong drug, wrong dose, incorrect preparation, contamination in preparation of the product, administration at the wrong rate and compatibility issues. There may be inadequate monitoring of sedation and/or delirium, over- or under-sedation, and errors related to unlicensed use of medications. Commonly used drugs that are not licensed include clonidine (sedation) and haloperidol (delirium) and lidocaine for analgesia.
How to prevent medication errors in the ICU?
Several interventions at the different phases of drug therapy can help to mitigate errors (Figure 1).
Prescribing
Electronic prescribing is widely used in critical care. A 2005 study that analysed errors before and after introduction of electronic prescribing found that error rates went down, but that the types of errors were potentially more harmful than with handwritten prescribing (Shulman et al. 2005). Nevertheless, both electronic prescribing and information provision at the point of prescription can reduce errors. Electronic systems can include preset standards of infusions, such as the UK Intensive Care Society’s standard concentrations for infusions used in critical care (Intensive Care Society 2017).
Dispensing
Solutions to reduce selection errors include robotic dispensing, barcode readers and dispensing cupboards that are barcoded.
Preparation
Most ICUs prepare intravenous (IV) infusions at the bedside. Ready-to-use products reduce handling and preparation complexity. The case for pre-filled syringes is strong. It is generally accepted that 10% too high or too low dose is acceptable (Wheeler et al. (2008). Ferner et al. (2001) studied concentrations in discarded bags of N-acetylcysteine (NAC) administered to 66 patients. Of these 63% were outside of 10% of the intended dose, 39% outside of 20% and 9% outside of 50%. Parshuram et al. (2003) randomly sampled 232 opioid infusions in a Paediatric Intensive Care Unit and found that 65% were outside of 10% of the intended dose and 6% had two-fold errors or greater. In 2008, the same author tested a scenario of 464 morphine calculations and preparation and found that 35% were outside of 10% of the intended dose, and 8% had two-fold errors or greater (Parshuram et al. 2008).
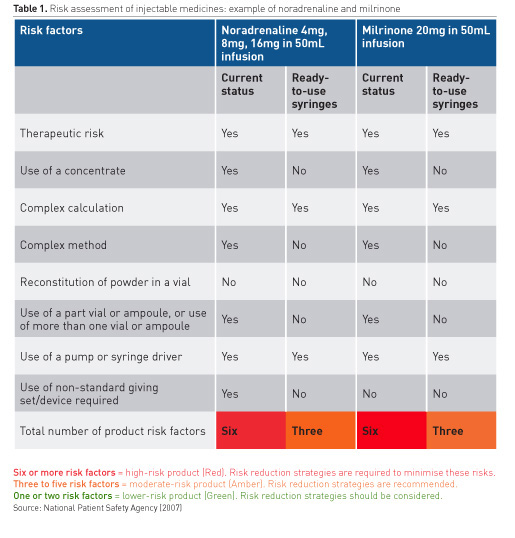
The UK National Patient Safety Agency produced a risk assessment tool for preparation and administration of injectable medicines in clinical areas (NPSA 2007). Table 1 shows risk assessment for noradrenaline and milrinone; using pre-filled syringes halved the number of risk factors.
Purchasing pre-filled syringes or ready-to-use infusion vials reduces the number of manipulations of the product and improves safety.
Contaminated propofol
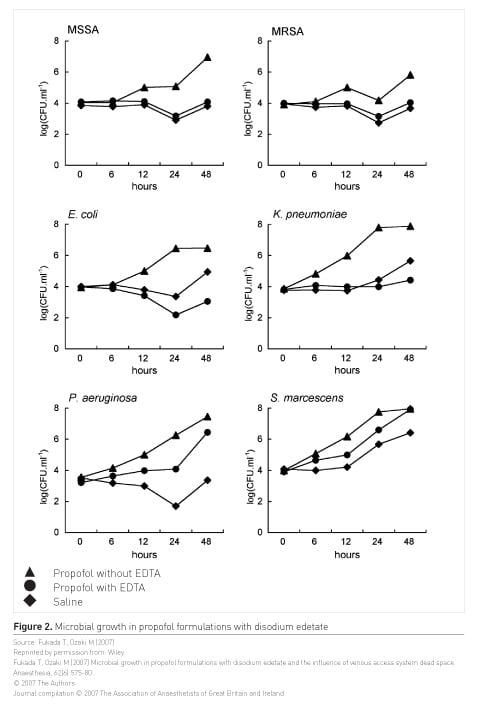
Propofol has been associated with healthcare-associated infections; a review of 58 studies identified 103/1405 (7.3%) incidents of contaminated propofol in theatres and 36/894 (4%) incidence of contaminated propofol in ICUs (Zorrilla-Vaca et al. 2016). Not all propofol formulations contain disodium edetate or EDTA, which reduces microbial growth; Fukada and Ozaki (2007) studied microbial growth in propofol preparations and found that propofol with disodium edetate suppressed bacterial growth more than propofol without. Taking a purchasing for safety approach to propofol, ICUs should consider using prefilled syringes and formulations that contain EDTA.
Figure 2 shows microbial growth in commercially available formulations; Fukada and Ozaki (2007) found that propofol with EDTA suppressed the growth of MSSA, MRSA, E. coli, and K. pneumoniae to a greater extent than propofol without EDTA.
Clinical pharmacist role
Having clinical pharmacists review and attend ward rounds has been shown to reduce errors. Meaningful reviews of critical incidents and near miss events, and disseminating solutions to colleagues can all help reduce errors (Shulman et al. 2015; Leape et al. 1999; MacLaren and Bond 2009).
Conclusion
There are many initiatives which ICUs can take to mitigate against error at every stage of the drug therapy process, and ensure patient safety. These include minimising interruptions during preparation, including a specialist clinical pharmacist in the multidisciplinary team, using electronic prescribing systems with guidelines and pre-prepared products.
Key Points
- Medication errors are likely to occur at the preparation and administration stages particularly in Intensive Care Units
- Consider pre-filled syringes to decrease the risks related to drug preparation
- Prefer formulations of propofol that include a microbial growth retardant (e.g. EDTA)
- A clinical pharmacist in the ICU can improve medication safety
- Barcoding solutions with ready-to-use products can improve patient safety
Elliott R, Camacho E, Campbell F et al. (2018) Prevalence and economic burden of medication errors in the NHS in England. Rapid evidence synthesis and economic analysis of the prevalence and burden of medication error in the UK. Policy Research Unit in Economic Evaluation of Health and Care Interventions. Universities of Sheffield and York. Available from eepru.org.uk/prevalence-and-economic-burden-of-medication-errors-in-the-nhs-in-england-2
Ferner RE, Langford NJ, Anton C et al. (2001) Random and systematic medication errors in routine clinical practice: a multicentre study of infusions, using acetylcysteine as an example. Br J Clin Pharmacol, 52(5):573-7.
Fukada T, Ozaki M (2007) Microbial growth in propofol formulations with disodium edetate and the influence of venous access system dead space. Anaesthesia, 62(6):575-80.
Intensive Care Society; Faculty of Intensive Care Medicine (2017) Medication concentrations in adult critical care (v 2.2). Available from https://ics152.files.wordpress.com/2017/02/ics-standard-medication-concentrations-2016.pdf
Leape LL, Cullen DJ, Clapp MD et al. (1999) Pharmacist participation on physician rounds and adverse drug events in the intensive care unit. JAMA, 282(3): 267-70.
MacLaren R, Bond CA (2009) Effects of pharmacist participation in intensive care units on clinical and economic outcomes of critically ill patients with thromboembolic or infarction-related events. Pharmacotherapy, 29(7): 761-8.
National Patient Safety Agency (2007) 0434H: Promoting safer use of injectable medicines - Risk assessment tool - 2007 - V1. Available from webarchive.nationalarchives.gov.uk/20171030132159/http://www.nrls.npsa.nhs.uk/resources/patient-safety-topics/medication-safety/?entryid45=59812&p=2
Parshuram CS, Ng GY, Ho TK et al. (2003) Discrepancies between ordered and delivered concentrations of opiate infusions in critical care. Crit Care Med, 31(10):2483-7.
Parshuram CS, To T, Seto W et al. Systematic evaluation of errors occurring during the preparation of intravenous medication. CMAJ, 178(1):42-8.
Rudall N, McKenzie C, Landa J et al. (2017) PROTECTED-UK - Clinical pharmacist interventions in the UK critical care unit: exploration of relationship between intervention, service characteristics and experience level. J Pharm Pract, 25(4):311-9.
Shulman R, McKenzie CA, Landa J et al.; PROTECTED-UK group. Pharmacist's review and outcomes: Treatment-enhancing contributions tallied, evaluated, and documented (PROTECTED-UK). J Crit Care, 30(4):808-13.
Shulman R, Singer M, Goldstone J et al. (2005) Medication errors: a prospective cohort study of hand-written and computerised physician order entry in the intensive care unit. Crit Care, 9(5):R516-21.
Tissot E, Cornette C, Demoly P et al. (1999) Medication errors at the administration stage in an intensive care unit. Intensive Care Med, 25(4):353-9.
van den Bemt PM, Fijn R, van der Voort PH et al. (2002) Frequency and determinants of drug administration errors in the intensive care unit. Crit Care Med, 30(4):846-50.
Wheeler DW, Degnan BA, Sehmi JS et al. (2008) Variability in the concentrations of intravenous drug infusions prepared in a critical care unit. Intensive Care Med, 34(8):1441-7.
Zorrilla-Vaca A, Arevalo JJ, Escandón-Vargas K et al. (2016) Infectious disease risk associated with contaminated propofol anesthesia, 1989-2014(1). Emerg Infect Dis, 22(6):981-92.