ICU Management & Practice, Volume 18 - Issue 4, 2018
Secondary complications
Maintenance of sufficient cerebral oxygen supply to meet metabolic demand is a key goal in managing patients with acute brain injury and in perioperative settings. A discrepancy between oxygen supply and demand can lead to cerebral hypoxia/ischaemia and deleterious outcome, with time-critical windows to prevent or minimise permanent ischaemic neurological injury.
As clinical manifestations of cerebral hypoxia/ischaemia may remain hidden in unconscious or sedated patients, brain monitoring is required to detect impaired cerebral oxygenation in such circumstances (Kirkman and Smith 2016).
Cerebral oxygenation monitoring methods
- Jugular venous oxygen saturation monitoring: this was the first bedside monitor of cerebral oxygenation; however, other monitoring tools are superseding its use.
- Near-infrared spectroscopy: these are commercial devices that measure regional cerebral oxygen saturation and enable simultaneous measurement of multiple regions of interest, although its inclusion in routine clinical practice is not widespread (Ghosh et al. 2012).
- Brain tissue oxygen pressure monitoring: In recent years, there has been an increasing trend towards direct measurement of partial brain tissue oxygen pressure (PtiO2); furthermore, this offers the most solid evidence of all cerebral oxygen monitors (Bouzat et al. 2013).
Technical aspects
PtiO2 catheters, which are inserted into subcortical white matter through single or multi-lumen bolts via a burr hole or during a craniotomy, are similar in size to intra-parenchymal ICP monitors.
PtiO2 is a localised measurement; the region of interest monitored by a PtiO2 probe is approximately 17mm. Precise insertion can be technically challenging or impossible, in addition to the risk of inadvertent intra-lesional placement, which does not provide useful information. A major caveat for PtiO2 monitoring is that the heterogeneity of brain oxygenation, even in undamaged areas, is well recognised (Van den Brink et al. 2000).
Study purpose, materials and methods
This study analysed complications arising from insertion of the multimodal monitoring sensor using a dual lumen introducer kit—Integra® Licox® Brain Tissue Oxygen Monitoring (Integra LifeSciences).
All patients in León Hospital ICU diagnosed with subarachnoid haemorrhage (SAH), head injury or intracranial haemorrhage (ICH), and who had been neuro-monitored either solely with ICP or with ICP+ PtiO2 were studied retrospectively for one year (from March 2017 to March 2018). We analysed the presence of haemorrhage associated with the catheter, infection and bone fragments in the brain.
Results
During the study period 126 patients were admitted with a diagnosis of head injury, subarachnoid or intraventricular haemorrhage. Twenty-one of these patients were monitored (16.6%); 20 underwent multimodal monitoring, and only one was fitted with just an ICP sensor.
Fourteen patients (70%), monitored with a ICP+ PtiO2 sensor, were found to have a bone fragment in the brain on undergoing a control CT after removal of the sensors. In another five patients, they either did not present with this complication or the control CT was not performed because they were discharged. Only one patient presented with a haematoma in the catheter trajectory. The only patient monitored with an ICP sensor had no bone fragments.
No infections whatsoever were associated with the sensor under any circumstance.
In 9 out of 20 patients monitored with ICP+ PtiO2, figures were normal for both ICP and PtiO2.
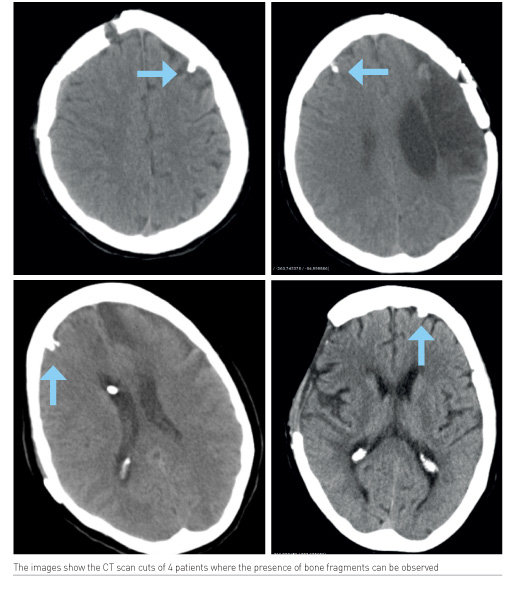
A neurosurgical team followed up these patients, and no infectious complications in the bone fragment area or untreatable seizures were observed. Nevertheless, it was impossible to follow up all those patients long-term.
On reviewing patients monitored solely with an ICP sensor, bone fragments have never been observed in the same.
Discussion
We did not find any publication describing the presence of bone fragments as a complication, so we were unable to make a comparison.
Technical considerations
The drill used for inserting a PtiO2 +ICP Integra® Licox® Brain Tissue Oxygen Monitoring (Integra LifeSciences) has a diameter of 6.3 mm; sheet geometry and cutting grooves mean that on drilling the bone, fragments enter the brain. It would seem a controlled delicate dissection is not possible.
According to Dings et al. (1997), this technique is considered relatively safe with a low haematoma incidence (<2% usually small and clinically insignificant). The most common technical complication described was dislocation or drift whose frequency may reach 13.6%. No infections have been described.
Advances in PtiO2 technology should lead to improved techniques, since the addition of a further lesion (bone fragment) in an already damaged brain should not be acceptable.
Conclusions
Insertion of ICP+PtiO2 sensors using the dual lumen insertion technique Integra® Licox® Brain Tissue Oxygen Monitoring (Integra LifeSciences) is associated with a high rate of intra-cerebral bone fragments. Long-term complications of these lesions have yet to be determined.
Co-Author
References:
Kirkman MA, Smith M (2016) Brain oxygenation monitoring. Anesthesiol Clin, 34(3):537-56.
Ghosh A, Elwell C, Smith M (2012) Review article: cerebral near-infrared spectroscopy in adults: a work in progress. Anesth Analg, 115(6):1373-83.
Bouzat P, Sala N, Payen J-F et al. (2013) Beyond intracranial pressure: optimization of cerebral blood flow, oxygen, and substrate delivery after traumatic brain injury. Ann Intensive Care, 3(1):23.
van den Brink WA, van Santbrink H, Steyerberg EW et al. (2000) Brain oxygen tension in severe head injury. Neurosurgery, 46(4):868-76.
Dings J, Meixensberger J, Roosen K (1997) Brain tissue pO2-monitoring: catheter stability and complications. Neurol Res, 19(3):241-5.







