ICU Management & Practice, ICU Volume 14 - Issue 2 - Summer 2014
Authors
Olav Rooyackers, PhD
Professor of Anaesthesia and Intensive Care Medicine
Division of Anaesthesia at CLINTEC, Karolinska Institutet
Stockholm, Sweden

Jan Wernerman, MD PhD
Professor of Anaesthesiology and Intensive Care Medicine
Division of Anaesthesia at CLINTEC, Karolinska Institutet
Stockholm, Sweden
Senior Consultant
Department of Anaesthesia and Intensive Care Medicine,
Karolinska University Hospital
Huddinge Stockholm, Sweden
Background
Nutritional interventions in ICU patients are intended to influence outcome. However, there are often difficulties in interpreting outcome data, as selection of patients may not allow the results to be generally applied. There are no known mechanisms linking nutrition directly to outcome, and therefore designing outcome studies without completely understanding the possible relation between intervention and outcome is not easy. Feeding critically ill patients has repeatedly been questioned, primarily in the acute and often unstable phase, sometimes called the ebb phase. Hypothetically, preservation of body proteins, or perhaps better attenuation of the decrease in body proteins, may be the immediate target of a nutritional intervention. Consequently, assessment of body proteins may be used as a substitute marker for outcome. Here we discuss possibilities for this type of assessment with different techniques.
Nutrition Studies
Several recently published clinical trials evaluating the effect of nutrition in critical illness are subject to conflicting interpretations (Casaer et al. 2011; Doig et al. 2013; Heidegger et al. 2013; National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network et al. 2012). To some extent the difficulty of defining patients at nutritional risk is the problem. Should all critically ill patients be given nutrition support? And at what time point? As mentioned there are a number of unknown steps from a nutritional intervention to mortality outcomes. Not surprisingly there are no prospective randomised studies in unselected ICU patients demonstrating survival advantages in relation to nutrition.
What should then be the target for nutritional intervention in ICU
patients? Obviously long-term starvation leads to death, but what is optimal nutrition
in critical illness? Will optimal nutrition be constant over time or may there
also be a temporal component in optimal nutrition? At least one aspect of the
efficacy of nutrition support is to save body proteins, and the key question
becomes how to estimate body protein mass. Which techniques are applicable in
everyday clinical practice with sufficient accuracy? Imaging may give
information regarding tissue volume, while biochemical analyses may give
information about possible mechanisms. Several different techniques together
with measures of function need to be considered. When preservation of body
proteins in the critically ill can be documented, prospective outcome studies
should be performed to evaluate whether or not preservation of body proteins is
a suitable proxy for core outcomes such as mortality and morbidity.
Muscle as an Index of Nutritional Status
The degree of muscle depletion relates to mortality and to post ICU quality of life. The loss of muscle proteins can be quantified (see Figure 1), but muscle biopsies are not possible in everyday clinical practice (Gamrin et al. 1997; Gamrin et al. 1996; Larsson et al. 2000). Therefore imaging is very attractive, in particular if imaging devises can be used bedside.
Ultrasound may be used to monitor sarcopenia in critically ill patients
over time (Reid et al. 2004). It has also been possible to link the extent of
the shrinkage of muscle cross section area to the severity of illness (Puthucheary
et al. 2013). The technique is cheap and readily available, and it reflects the
muscle loss objectively. A strict protocol for these measurements is needed to achieve
reproducible results. The risk of large investigator-related variability is
obvious. A direct link between changes in biochemistry and in ultrasound
imaging in individual subjects has not yet been presented, but such study
protocols should be encouraged.
When abdominal CT scans have been performed for other purposes, several investigators have utilised the images to diagnose sarcopenia and its progress over time (Baracos et al. 2010; Mourtzakis et al. 2008; Braunschweig et al. 2013; Moisey et al. 2013). Such imaging may also have a prognostic value (Weijs et al. 2014; Casaer et al. 2013). For the time being CT scanners are located outside the ICU, which limits accessibility and possibly imposes risks. However, the high resolution of modern CT scanners makes it a very promising tool. The literature contains reports of both leg muscle and abdominal muscle. A different time course of depletion may be at hand (Casaer et al. 2013). The investigator neutrality is attractive. More reports are warmly welcome, to elucidate the full potential of this technique.
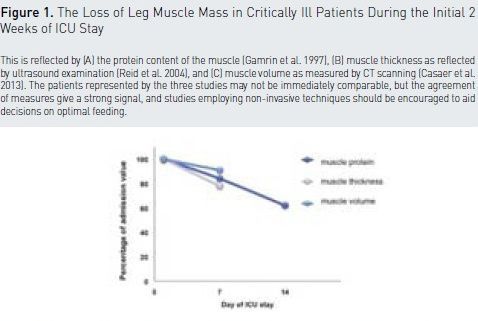
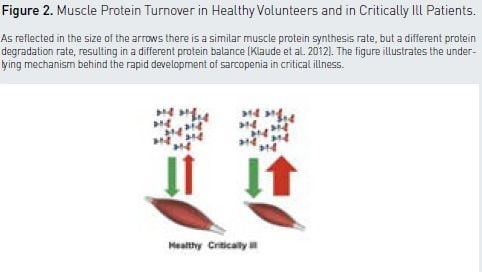
The full value of imaging will become obvious when the image is directly linked to the results of biochemical alterations in the tissue, such as protein turnover, gene expression, and tissue signalling (Fredricksson et al. 2008; Klaude et al. 2012). In critically ill patients sarcopenia is driven by skeletal muscle degradation (Klaude et al. 2012; Klaude et al. 2005; Klaude et al. 2007), while protein synthesis rate in skeletal muscle is unaltered in most cases during critical illness and the development of sarcopenia (see Figure 2). This example also nicely illustrates the need to understand underlying mechanisms to direct intervention studies correctly. In this case, finding nutritional interventions to inhibit muscle breakdown will most likely have a better effect.
The development of sarcopenia and the temporal development of muscle depletion is attributable to many other factors beside nutrition, and the extrapolation of muscle mass to assess outcome risk and possibly be an indicator of a favourable response to nutrition must be viewed as hypothetical. The link between metabolic care and nutrition of the critically ill patients is challenging, and to monitor this by muscle mass seems very attractive.
Muscle Mass and Muscle Function
Muscle volume and muscle function are not closely correlated, and post
ICU muscle function studies are sometimes difficult to interpret. Usually
patient recruitment is quite selective and limited to patients able and
motivated to participate in training activities (Poulsen et al. 2011).
Nevertheless muscle mass may be a proxy for more than locomotor function in
evaluations of post ICU quality of life, which usually rely upon questionnaires
(Herridge et al. 2011; Griffiths et al. 2013). The possibility of an objective
measurement is always attractive, but the relevance in post ICU care is still
totally open. Sarcopenia during ICU stay is possibly a reflection of
malnutrition, and a relation to mortality outcome has been reported (Weijs et
al. 2014).
Conclusion
For full references, please email [email protected], visit the website at www.icu-management.orgor use the QR code at the top of the article.






