The idea of liver assist is extremely attractive for patients with liver failure, as the liver possesses huge capacity to regenerate. The hypothesis behind the use of extracorporeal liver assist is to enhance the regenerative environment by removing or replacing toxic molecules while the liver can regenerate. Extracorporeal liver assist devices are either based on the principles of blood purification or attempt to provide some synthetic function. The devices based on the principles of blood purification include albumin dialysis, plasma separation and filtration or therapeutic plasma exchange. The alternative is the bioartificial liver, which incorporates hepatocytes in the extracorporeal circuit. Attempts to develop effective extracorporeal liver assist devices continue. With better device design, understanding of the pathophysiological basis of liver failure and emergence of tools to stratify patients, it is likely that an effective liver assist device will emerge.
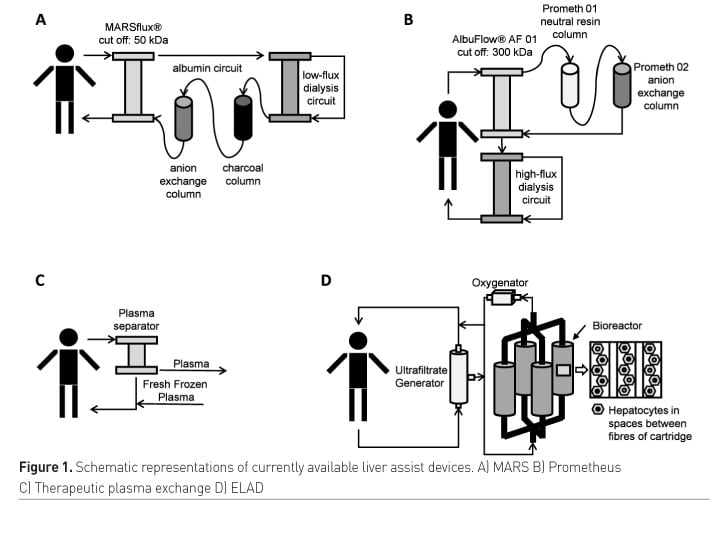
The liver has a huge capacity to regenerate. The hypothesis behind the use of extracorporeal liver assist is to enhance the regenerative environment by removing or replacing toxic molecules while the liver can regenerate. The two principal indications for the use of these devices are in patients with acute liver failure (ALF) and those with acute-on-chronic liver failure (ACLF). The predominant difference between these syndromes is that in ALF the native liver is normal whereas in ACLF the liver is cirrhotic. Extracorporeal liver assist devices are either based on the principles of blood purification or attempt to provide some synthetic function. The devices based on the principles of blood purification include albumin dialysis, plasma separation and filtration or therapeutic plasma exchange (TPE). The alternative is the bioartificial liver, which incorporates hepatocytes in the extracorporeal circuit. The available devices and emerging technologies are described below (Figure 1).
You might also like:Liver intensive care
Albumin dialysis
Two main types are currently available.
Molecular Adsorbents Recirculating System (MARS®, Gambro AB)
(Stange et al. 1993a; 1993b)
Albumin dialysis is achieved by dialysis of blood across an albumin-impermeable membrane (membrane pore size has a cut-off of approximately 50-60 kDa) against 20% human serum albumin (HSA). The HSA solution is continuously stripped of protein-bound and water-soluble substances by passage through a secondary circuit containing a charcoal column, an anion exchange resin column and a low-flux dialyser.
The earliest randomised trial of MARS in ACLF was performed 20 years ago in 13 patients (n=8 MARS vs 5 controls) with type 1 hepatorenal syndrome and demonstrated a survival advantage with MARS, with no survivors in the control group (standard medical therapy [SMT]) versus 37.5% in the MARS group at day 7 (Mitzner et al. 2000). Subsequent randomised controlled trials (RCTs) of MARS versus SMT have yielded mixed results, with some studies showing a survival benefit (Heemann et al. 2002) and others no benefit (Sen et al. 2004; Banares et al. 2013). The largest study of MARS in ACLF (RELIEF study) (Banares et al. 2013) (Table 1) involved 189 patients (n=95 MARS vs 94 SMT) with there being no difference observed in 28-day survival on an intention-to-treat analysis (60.7% [MARS] versus 58.9% [SMT]). However, MARS treatment was associated with a significant reduction in serum bilirubin (26.4% vs 8.9%, p < 0.001), serum creatinine (20.0% vs 6.4%, p=0.022) and some improvement in the degree of hepatic encephalopathy (HE) (from grade II-IV to grade 0-I) (OR 0.37; 95% CI 0.12-1.09; p = 0.07). A meta-analysis of MARS in liver failure, which included 6 RCTs totalling 453 ACLF patients, showed no improvement in survival with MARS (OR 0.88; 95% CI 0.74-1.06; p = 0.16) (He et al. 2015). Another meta-analysis included 10 studies (7 ACLF and 3 ALF), of which 9 were RCTs and 1 non-RCT. No beneficial effect of MARS on mortality was observed (OR 0.91; 95% CI 0.64-1.31; p = 0.62) (Vaid et al. 2012). However, a significant reduction in serum bilirubin (net change -7.0mg/dl; 95% CI -10.4, -3.7; p <0.001) and an improvement in the grade of HE (OR 3.0; 95% CI 1.9 - 5.0; p<0.001) were noted with MARS (Vaid et al. 2012). The failure to observe a beneficial effect with MARS in ACLF may be due to heterogeneity of studies, both due to varying patient populations and a variance in defining ACLF; all the studies pre-date the seminal definition of ACLF as per the European Foundation for the Study of Chronic Liver Failure (EF-CLIF) (Moreau et al. 2013). Gerth et al. (2017) performed a retrospective analysis using the EF-CLIF ACLF definition. Of 101 ACLF patients (n=47 MARS vs 54 SMT) included at a single centre, higher survival was seen at 14 days with MARS (91.5% vs 50% SMT), which only remained significant when extended to 28 days in those with ACLF grade 2 or 3 (p = 0.022) (Gerth et al. 2017). In the re-analysis of the RELIEF cohort those with ACLF grade 2 or above benefitted most with MARS but only in the short term (14-day survival, 77.4% vs 66.1% SMT), whereas those with ACLF grade 1 treated with MARS had an increased mortality (14-day survival 20% vs 10% SMT) (Gerth et al. 2017). Overall in ACLF patients current evidence supports a limited role for MARS, in carefully selected patients with higher grades of ACLF or in those with advanced HE.
The largest RCT of MARS in ALF included 102 patients (n=53 MARS vs 49 SMT) from 16 French transplant centres and showed no survival benefit of MARS at 6 months (84.9% vs 74.4% SMT, p = 0.28) (Saliba et al. 2013). A significant criticism of this study was the short time from randomisation to liver transplantation (median 16.2 hours), which may have limited any demonstrable effect from albumin dialysis.
Fractionated Plasma Separation, Adsorption and Dialysis device (Prometheus®, Fresenius Medical Care AG & Co. KGaA)
(Falkenhagen et al. 2013; Rifai et al. 2003)
Plasma separation from blood is achieved by passage of blood across an approximately 300 kDa cut-off membrane. Plasma is cleansed by direct passage through two cartridges containing different adsorbents. Plasma is then returned to the blood circuit for clearance of water-soluble substances by a high-flux dialyser. A modification of this device, which excludes the high flux dialyser, known as double plasma molecular absorption system (DPMAS, Fresenius Medical Care) is also under investigation, as a plasma-sparing alternative to TPE (Wan et al. 2017).
There are few controlled studies of Prometheus® in liver failure (Rifai et al. 2003; Laleman et al. 2006). The largest RCT included 145 ACLF patients (n=77 vs 68 SMT) and demonstrated no survival advantage at 28 days (66% vs 63% in the SMT group, p = 0.70) (Kribben et al. 2012). A retrospective case series of 23 ALF patients supports a potential role for Prometheus® as a bridge to transplantation or recovery (Senturk et al. 2010), but further data are needed for validation before any recommendation.
Therapeutic plasma exchange
Therapeutic plasma exchange (TPE) requires use of an extracorporeal blood cell separator for removal of plasma from blood. Patient plasma is then discarded, whilst blood cells are mixed with a replacement fluid and returned to the patient. Fresh frozen plasma (FFP) is the typical replacement fluid, but HSA has also been reported. TPE aims to achieve removal of toxins plus harmful inflammatory mediators and replacement of beneficial plasma proteins normally synthesised by the liver.
There have been no RCTs of TPE in ACLF, but evidence from prospective studies supports evidence for an improvement in Model for End-Stage Liver Disease (MELD) score and survival (Yue-Men et al. 2016; Ling et al. 2012; Zhou et al. 2015). Early case series of TPE improving outcomes in ALF (Nakamura et al. 2000; Kondrup et al. 1992; Larsen et al. 1995) have been supported by a multicentre RCT of high-volume plasma exchange (HVP) (15% of body weight [8-12L FFP]), which included 182 patients (92 HVP vs 90 SMT), and showed an improved hospital survival; 58.7% versus 47.8% (SMT) hazard ratio (HR) 0.56; 95% CI 0.36-0.86; p = 0.0083) (Larsen et al. 2016). In those patients that did not receive emergency liver transplantation, HVP improved survival compared to SMT (p=0.03), but in those who received a transplant prior use of HVP did not improve outcome compared to SMT (Larsen et al. 2016).
Bioartificial liver support devices
The rationale of bioartificial devices is to augment any residual liver function and support the failing liver by incorporating functional hepatocytes in combination with blood detoxification or additional mechanisms to attenuate liver injury.
Extracorporeal Liver Assist Device (ELAD™, Vital Therapies Inc.)
The key component of ELAD™ is a quartet of hollow fibre dialysis cartridges containing HepG2/C3A cells, a human hepatoblastoma cell line, within the extra-fibre spaces. ELAD has been trialled in a small number of ALF patients with limited efficacy (Millis et al. 2002; Sussman et al. 1992; Ellis et al. 1996), but the larger phase 2 VTI-212 trial of ELAD in ALF was terminated. An RCT of ELAD therapy in severe alcoholic hepatitis (VTI-208), which included 203 patients (96 ELAD and 107 SMT), did not demonstrate any survival benefit of ELAD therapy at 91 days (51.0% vs 49.5% SMT), thus failing its primary endpoint (Thompson et al. 2018). Although not pre-specified, survival in patients with a combination of both MELD <28 and age <46.9 years (n=59) was significantly better in the ELAD group compared to SMT (100% vs 73%, p = 0.006) at 91 days (Thompson et al. 2018), which is the basis for the current VTI-308 trial of ELAD in alcoholic hepatitis (
clinicaltrials.gov/ct2/show/NCT02612428).
Emerging technologies in extracorporeal artificial liver support devices
ADVanced Organ Support (ADVOS previously known as Hepa Wash, Hepa Wash GmbH)
ADVOS® detoxifies blood by albumin dialysis against a 2% albumin dialysate (Al-Chalabi et al. 2013). The albumin dialysate is recirculated via the ADVOS circuit, which contains two parallel conventional haemofilters, in which albumin-bound toxins are released through exposure to an alkaline or acid environment and subsequently removed by filtration. This design aims to maintain clearances of protein bound toxins through the treatment period (Al-Chalabi et al. 2013). In a pig liver failure model ADVOS® resulted in improvement in survival, cerebral perfusion pressure, haemodynamic status and kidney function (Al-Chalabi et al. 2013; 2017). A retrospective report of the first fourteen patients treated with ADVOS (mean treatment session 575 minutes) showed that there was significant reduction in serum bilirubin and creatinine (Huber et al. 2017).
Li-Artificial Liver Support (Li-ALS)
Li-ALS combines a low-volume TPE (exchange of approximately 2.5% body weight of plasma) circuit with a modified MARS secondary circuit, in which high-flux haemofiltration replaces low-flux haemodialysis (Zhou et al. 2015). This approach seeks to benefit from the more comprehensive detoxification achieved by TPE compared to MARS, without need for a supply of exogenous fresh frozen plasma, as patient plasma is returned post-detoxification to the patient. In a D-galactosamine pig model of ALF, Li-ALS resulted in an improvement in survival compared to treatment with low-volume TPE alone and to treatment with the modified MARS circuit alone (Zhou et al. 2015).
University College London-Liver Dialysis Device (DIALIVE)
In DIALIVE blood is filtered across two filters; one a high-cut off membrane (nominal cut-off of 60kDa) through which albumin passes, the second is a selective endotoxin adsorption membrane. Albumin lost during filtration is replaced by HSA infusion (Lee et al. 2015), thus this effective albumin exchange addresses the irreversible loss of detoxifying function of albumin in liver failure. Targeted removal of endotoxin aims to reduce innate immune response, which is a key pathophysiological component driving ACLF. In a pig model of paracetamol-induced ALF, DIALIVE improved survival and cardiovascular and respiratory function and reduced circulating dysfunctional albumin, endotoxaemia and immune system activation (Lee et al. 2015). DIALIVE is currently being evaluated in a multi-centre RCT to assess its performance and safety (
clinicaltrials.gov/ct2/show/NCT03065699).
Academic Medical Centre Bioartificial Liver (AMC-BAL)
The bioreactor features of AMC-BAL are a non-woven matrix for 3D hepatocyte cultures; spiralling of this 3D matrix around oxygen carrying capillaries; and direct exposure of hepatocytes to patient plasma (van de Kerkhove et al. 2005). The first trial of this device in 12 ALF patients used primary porcine hepatocytes; however, the risk of zoonoses has tempered further development as porcine DNA was found in patient plasma (van de Kerkhove et al. 2005).
Spheroid reservoir bioartificial liver (SRBAL)
The bioreactor of SRBAL contains primary porcine hepatocytes in suspension, which when exposed to an oscillation frequency of 0.25Hz cluster into spheroids with stable cell viability (Nyberg et al. 2005; McIntosh et al. 2009). Hepatocyte spheroids demonstrate good hepatocyte function in terms of: phase I and phase II drug metabolism; ammonia conversion to urea via the urea cycle; and albumin synthesis (Nyberg et al. 2005). A trial using a pig ALF model has shown improved survival, but further development is required (Glorioso et al. 2015).
Conclusions
The idea of liver assist is extremely attractive for patients with liver failure, as the liver possesses huge capacity to regenerate. If the patient can be kept alive long enough, it may be possible for the liver to regenerate and return the patient to the state of health they were in prior to developing liver failure. Although many of the devices available have been shown to improve the clinical condition, only plasma exchange was able to improve transplant-free survival of patients with ALF. Attempts to develop effective extracorporeal liver assist devices continue. With better device design, understanding of the pathophysiological basis of liver failure and emergence of tools to stratify patients, it is likely that an effective liver assist device will emerge.
Conflict of interest
Rajiv Jalan has research collaborations with Takeda, Ocera, and Yaqrit, and consults with Yaqrit. Rajiv Jalan is the founder of Yaqrit Limited, which is developing UCL inventions for treatment of patients with cirrhosis. Rajiv Jalan is an inventor of ornithine phenylacetate, which was licensed by UCL to Mallinkrodt Pharma. He is also the inventor of Yaq-001, DIALIVE and Yaq-005, the patents for which have been licensed by his university into a UCL spinout company, Yaqrit Ltd. No other authors declared conflicts of interest.
Abbreviations
ADVOS ADVanced Organ Support
ALF acute liver failure
ACLF acute-on-chronic liver failure
CI confidence interval
DPMAS double plasma molecular absorption system
EF-CLIF European Foundation for the Study of Chronic Liver Failure
FFP fresh frozen plasma
HE hepatic encephalopathy
HSA human serum albumin
HR hazard ratio
HVP high-volume plasma exchange
kDA kilodalton
MELD Model for End-Stage Liver Disease
OR odds ratio
RCT randomised controlled trial
SRBAL Spheroid Reservoir Bioartificial Liver
SMT standard medical therapy
TPE therapeutic plasma exchange
Al-Chalabi A, Matevossian E, v Thaden AK et al. (2013) Evaluation of the Hepa Wash® treatment in pigs with acute liver failure. BMC Gastroenterol, 13: 83
Al-Chalabi A, Matevossian E, von Thaden A et al. (2017) Evaluation of an ADVanced Organ Support (ADVOS) system in a two-hit porcine model of liver failure plus endotoxemia. Intensive Care Med Exp, 5: 31.
Banares R, Nevens F, Larsen FS et al. (2013) Extracorporeal albumin dialysis with the molecular adsorbent recirculating system in acute-on-chronic liver failure: the RELIEF trial. Hepatology, 57: 1153-62.
Ellis AJ, Hughes RD, Wendon JA et al. (1996) Pilot-controlled trial of the extracorporeal liver assist device in acute liver failure. Hepatology, 24: 1446-51.
Falkenhagen D, Strobl W, Vogt G et al. (1999) Fractionated plasma separation and adsorption system: a novel system for blood purification to remove albumin bound substances. Artif Organs, 23: 81-6.
Gerth HU, Pohlen M, Thölking G et al. (2017) Molecular Adsorbent Recirculating System can reduce short-term mortality among patients with acute-on-chronic liver failure—a retrospective analysis. Crit Care Med, 45: 1616-24.
Glorioso JM, Mao SA, Rodysill B et al. (2015) Pivotal preclinical trial of the spheroid reservoir bioartificial liver. J Hepatol, 63: 388-98.
Hassanein TI, Tofteng F, Brown Jr RS et al. (2007) Randomized controlled study of extracorporeal albumin dialysis for hepatic encephalopathy in advanced cirrhosis. Hepatology, 46: 1853-62.
He GL, Feng L, Duan CY et al. (2015) Meta-analysis of survival with the molecular adsorbent recirculating system for liver failure. Int J Clin Exp Med, 8(10): 17046-54.
Heemann U, Treichel U, Loock J et al. (2002) Albumin dialysis in cirrhosis with superimposed acute liver injury: a prospective, controlled study. Hepatology, 36: 949-58.
Huber W, Henschel B, Schmid R et al. (2017) First clinical experience in 14 patients treated with ADVOS: a study on feasibility, safety and efficacy of a new type of albumin dialysis. BMC Gastroenterol, 17: 32.
Kondrup J, Almdal T, Vilstrup H et al. (1992) High Volume plasma exchange in fulminant hepatic failure. Int J Artif Organs, 15: 669-76.
Kribben A, Gerken G, Haag S et al. (2012) Effects of fractionated plasma separation and adsorption on survival in patients with acute-on-chronic liver failure. Gastroenterology;142:782-789.e783.
Laleman W, Wilmer A, Evenepoel P et al. (2006) Effect of the molecular adsorbent recirculating system and Prometheus devices on systemic haemodynamics and vasoactive agents in patients with acute-on-chronic alcoholic liver failure. Crit Care, 10: R108.
Larsen FS, Ejlersen E, Hansen BA et al. (1995) Systemic vascular resistance during high-volume plasmapheresis in patients with fulminant hepatic failure: relationship with oxygen consumption. Eur J Gastroenterol Hepatol, 7: 887-92.
Larsen FS, Schmidt LE, Bernsmeier C et al. (2016) High-volume plasma exchange in patients with acute liver failure: An open randomised controlled trial. J Hepatol, 64: 69-78.
Lee KC, Baker LA, Stanzani G et al. (2015) Extracorporeal liver assist device to exchange albumin and remove endotoxin in acute liver failure: results of a pivotal pre-clinical study. J Hepatol, 63: 634-42.
Ling Q, Xu X, Wei Q et al. (2012) Downgrading MELD improves the outcomes after liver transplantation in patients with acute-on-chronic hepatitis B liver failure. PLoS One, 7: e30322.
McIntosh MB, Corner SM, Amiot BP et al. (2009) Engineering analysis and development of the spheroid reservoir bioartificial liver. Conf Proc IEEE Eng Med Biol Soc, 2009: 5985-8.
Millis JM, Cronin DC, Johnson R et al. (2002) Initial experience with the modified extracorporeal liver-assist device for patients with fulminant hepatic failure: system modifications and clinical impact. Transplantation, 74: 1735-46.
Mitzner SR, Stange J, Klammt S et al. (2000) Improvement of hepatorenal syndrome with extracorporeal albumin dialysis MARS: results of a prospective, randomized, controlled clinical trial. Liver Transpl, 6: 277-86.
Moreau R, Jalan R, Gines P et al. (2013) Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology, 144: 1426-37.
Nakamura T, Ushiyama C, Suzuki S et al. (2000) Effect of plasma exchange on serum tissue inhibitor of metalloproteinase 1 and cytokine concentrations in patients with fulminant hepatitis. Blood Purif, 18: 50-4.
Nyberg SL, Hardin J, Amiot B et al. (2005) Rapid, large-scale formation of porcine hepatocyte spheroids in a novel spheroid reservoir bioartificial liver. Liver Transpl, 11: 901-10.
Rifai K, Ernst T, Kretschmer U et al. (2003) Prometheus--a new extracorporeal system for the treatment of liver failure. J Hepatol, 39: 984-90.
Saliba F, Camus C, Durand F et al. (2013) Albumin dialysis with a noncell artificial liver support device in patients with acute liver failure: a randomized, controlled trial. Ann Intern Med, 159: 522-31.
Sen S, Davies NA, Mookerjee RP et al. (2004) Pathophysiological effects of albumin dialysis in acute‐on‐chronic liver failure: a randomized controlled study. Liver Transpl, 10: 1109-19.
Senturk E, Esen F, Ozcan PE et al. (2010) The treatment of acute liver failure with fractionated plasma separation and adsorption system: experience in 85 applications. J Clin Apher, 25: 195-201.
Stange J, Mitzner S, Ramlow W et al. (1993) A new procedure for the removal of protein bound drugs and toxins. ASAIO J, 39(3): M621-5.
Stange J, Ramlow W, Mitzner S et al. (1993) Dialysis against a recycled albumin solution enables the removal of albumin-bound toxins. Artif Organs, 17: 809-13.
Sussman NL, Chong MG, Koussayer T et al. (1992) Reversal of fulminant hepatic failure using an extracorporeal liver assist device. Hepatology, 16: 60-5.
Thompson J, Jones N, Al-Khafaji A et al. (1996) Extracorporeal cellular therapy (ELAD) in severe alcoholic hepatitis: A multinational, prospective, controlled, randomized trial. Liver Transpl, 24: 380-93.
Vaid A, Chweich H, Balk EM et al. (2012) Molecular adsorbent recirculating system as artificial support therapy for liver failure: a meta-analysis. ASAIO J, 58: 51-9.
van de Kerkhove MP, Poyck PP, Deurholt T et al. (2005) Liver support therapy: an overview of the AMC-bioartificial liver research. Dig Surg, 22: 254-64.
Wan YM, Li YH, Xu ZY et al. (2017) Therapeutic plasma exchange versus double plasma molecular absorption system in hepatitis B virus-infected acute-on-chronic liver failure treated by entercavir: a prospective study. J Clin Apher, 32: 453-61.
Yue-Meng W, Yang L-H, Yang J-H et al. (2016) The effect of plasma exchange on entecavir-treated chronic hepatitis B patients with hepatic de-compensation and acute-on-chronic liver failure. Hepatol Int, 10: 462-9.
Zhou N, Li J, Zhang Y et al. (2015) Efficacy of coupled low-volume plasma exchange with plasma filtration adsorption in treating pigs with acute liver failure: a randomised study. J Hepatol, 63: 378-87.
Zhou PQ, Zheng SP, Yu M et al. (2015) Prognosis of acute-on-chronic liver failure patients treated with artificial liver support system. World J Gastroenterol, 21: 9614-22.










