ICU Management & Practice, ICU Volume 8 - Issue 3 - Autumn 2008
Authors
Introduction
After diagnosis of infection and prescription of antibiotics, the assessment of clinical response is based on either clinical or microbiological criteria, mostly the same used in diagnosis (Dennesen et al. 2001). However clinical parameters can be influenced by a number of non-infectious factors, such as the drugs used in critical care settings (Povoa et al. 2005a). Moreover microbiological criteria are of little help, because of the delay of culture results, the interference of antibiotics with bacterial growth in vitro and the difficulties in obtaining some microbiological samples (e.g. from central spine fluid or peritoneum). On the other hand, treatment failure may be wrongly presumed, in the presence of a slow improvement or the appearance of a superimposed problem, such as drug fever, inflammatory conditions or a new hospitalacquired infection (Niederman et al. 2001). In this context, the use of serum markers, namely C-reactive protein (CRP) and procalcitonin (PCT), has been shown to help in the assessment of response to therapy, and thereby contribute to clinical decision (Povoa, 2008).
Serum Biomarkers in the Evaluation of Infection Response to Antibiotics
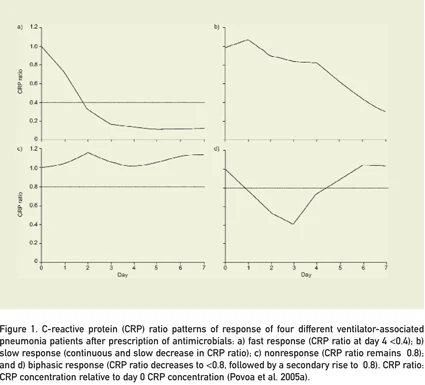
A marker of infection should: (i) be absent if the patient is not infected; (ii) appear concomitantly and ideally precede the clinical manifestations of the infection; (iii) disappear with successful therapy or (iv) remain elevated if infection is refractory to treatment (Marshall et al. 2003). To evaluate the clinical response, the marker should also exhibit large amplitude of variation and have a non ‘exhaustion’ or ‘fatigue’ behaviour in prolonged septic episodes. C-reactive protein has first order elimination kinetics. Therefore the withdrawal of the inflammatory stimulus is followed by a sharp decrease in its serum concentration. As a result, a decreasing concentration is related to clinical improvement whether rising levels are associated with a nonresolving inflammatory process.We were able to show that these relative variations of CRP concentration correlated with the patient outcome. Using CRP-ratio, that is its daily concentration divided by the one measured on the day of infection diagnosis, we were allowed to identify four different patterns of response to therapy (Povoa et al. 2005a, Povoa et al. 2005b). The first, a fast response pattern, consists on a rapid decline of the CRP-ratio to less than 0,4 by day 4. The second, slow response pattern, is a continuous decline of the CRP-ratio, being its value, by day 4, less than 0,8 (but more than 0,4). The third is a nonresponse pattern, which is defined by a CRP-ratio course persistently above 0,8 (and sometimes even increasing), and the last, a biphasic response pattern, characterized by an initial drop of the CRPratio bellow 0,8, followed by a secondary rise, to a value above that threshold (Figure 1).
In a study of our group, 47 documented ventilatorassociated pneumonia (VAP) patients (Povoa et al. 2005a) were classified according to these CRP patterns criteria. None of the 30 patients with one of the two first described CRP-ratio decreasing patterns died, whereas only 4 of the 17 patients with persistently elevated CRP levels survived. Besides this correlation with prognosis, we found that CRP kinetics also correlated with the adequacy of initial antibiotic therapy: those with an adequate empiric therapy showed a marked drop of CRP-ratio, whilst in patients with inadequate antibiotics CRP-ratio were always above 1. Using the same methodology, we obtained similar findings in 44 patients with bloodstream infections (Povoa et al. 2005b).
We also found in patients with severe community acquired pneumonia (CAP), a correlation between CRP-ratio and outcome (Coelho et al. 2007). Failure to decrease the CRP-ratio to less than 0,5 by day 3 of therapy was predictive of a bad outcome. Moreover in this study a decline of more than 31% of CRP levels in two consecutive days predicted recovery (of 29 patients with this finding, 27 survive).
Yentis et al. found CRP to be elevated in patients with microbiological proven infection, with no differences in peak concentration between single or multiple septic episodes (Yentis et al. 1995). Additionally, the relative variation of CRP concentration, that is a decrease in its level by 25% in one day, was highly predictive of sepsis resolution (sensitivity of 97%, specificity of 95%).
Similarly, in a 28 CAP inpatient population a sharply decrease in CRP-ratio (to less than 0,32) after 96 hrs of therapy was noted (Smith et al. 1995). All the four patients considered to be antibiotic failures had persistently elevated CRPratios.
Lisboa et al. evaluated in 68 VAP patients not only CRP-ratio but also its correlation with microbiological burden (measured by quantitative tracheal aspirates) (Lisboa et al. 2008). In patients with adequate antibiotic therapy, the CRP-ratio at day 4 felt to 0,58 (±0,32) while, with inadequate antimicrobial therapy, CRP-ratio eventually rose (1,36±1,11), as we have already noted (Povoa et al. 2005a). These results correlate with bacterial load that also stayed high in patients with inadequate antibiotic therapy.
No study was accomplished to evaluate the performance of CRP-ratio within the first 2 days of therapy. However, its serial measurements may provide some degree of confidence that antibiotics are adequate (or not), well before the culture results are made available. Also, even in patients with microbiological identification, a lack of a decreasing CRP-ratio, may provide information about an ongoing complication (like endocarditis or an abcess), or another hidden infection (Eisenhut, 2008).
Therefore, in high risk patients failure to reduce CRP-ratio as soon as by day 2 or 3 of antibiotic therapy, should prompt an aggressive diagnostic and therapeutic approach, with source control efforts (e.g. removing central lines, debriding necrotic tissues) and performance of a full diagnostic approach (repeating microbiological cultures, performing ultrasound or CT scan) (Coelho and Povoa 2008). Additionally enlargement of antibiotic spectrum should be considered, as these patients have a very high risk of death (Kollef et al. 1999).
The value of PCT kinetics was also assessed in a population with documented VAP. A good correlation with clinical severity was found (Luyt et al. 2005). However some patients with documented VAP presented undetectable PCT levels at the day of diagnosis and, as a result, in these patients PCT relative variations could not be used to monitor response.
In another study, PCT was proposed to diagnose and guide the duration of antibiotic therapy in CAP. Patients in the PCT-guided group reduced their antibiotic therapy to 5 days, compared with 12 days in patients treated according with the guidelines (Christ-Crain et al. 2004). However an almost undetectable level of PCT on the day of diagnosis was also found in 29% of these patients. Consequently, in those patients, it was virtually impossible to evaluate the rate of PCT decline.
In 75 VAP patients the decrease of both PCT and CRP at day 4 of therapy was predictive of survival, with odds-ratio (OR) of 4.4 and 7.4 respectively (Seligman et al. 2006). In that study the CRP-ratio at day 4 of therapy was 0,67 for survivors and 0,88 for nonsurvivors.
To overcome the limitations of a single biomarker some studies evaluated the relative accuracy of panels of biomarkers. Gaini et al. evaluated the accuracy of four biomarkers in 194 patients, namely CRP and PCT, to identify patients with infection (Gaini et al. 2006). They found that CRP was the biomarker with the highest diagnostic performance, whereas PCT presented the lowest (AUC = 0,83 vs. 0,77, respectively).
Another unsolved issue is the optimal duration of antibiotic therapy. Eventually it should vary with the severity of the infection as well as with the clinical course. Serum markers may help to identify patients who can benefit from a short antibiotic course. In a 425 neonatal paediatric population, CRP was used to define length of therapy: its peak levels were used to define the duration of treat ment and its normalization were used to stop antibiotics (Philip and Mills, 2000). Only 19 patients received antibiotics for more than 5 days and there were no readmissions within a month.
Conclusion
Considering CRP kinetics, serial determinations of this marker can be very useful for monitoring inflammatory activity, the onset of infections and patient responses to antimicrobials and sepsis therapies. On the other hand, PCT seems to be a better indicator of illness severity and prognostic marker. Both these markers are dynamic, so their concentration should be measured on a daily basis, since changes in concentration over time, particularly in their ratios, are more informative than a single value.However, the use of laboratory markers should not preclude a correct and thorough clinical evaluation.