Bioresorbable splint used for first time, successfully stopped
life-threatening tracheobronchomalacia, case featured in New England
Journal of Medicine.
Every day, their baby stopped breathing, his collapsed bronchus blocking the crucial flow of air to his lungs. April and Bryan Gionfriddo watched helplessly, just praying that somehow the dire predictions weren’t true. “Quite a few doctors said he had a good chance of not leaving the hospital alive,” says April Gionfriddo, about her now 20-month-old son, Kaiba. “At that point, we were desperate. Anything that would work, we would take it and run with it.” They found hope at the University of Michigan (U-M), where a new, bioresorbable device that could help Kaiba was under development. Kaiba’s doctors contacted Glenn Green, M.D., associate professor of pediatric otolaryngology at the University of Michigan.
Green and his colleague, Scott Hollister, Ph.D., professor of
biomedical engineering and mechanical engineering and associate
professor of surgery at U-M, went right into action, obtaining emergency
clearance from the Food and Drug Administration to create and implant a
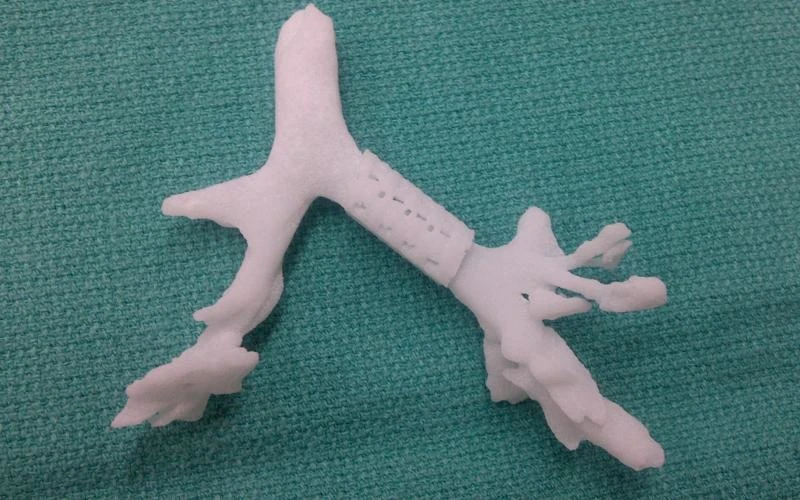
tracheal splint for Kaiba made from a biopolymer called
polycaprolactone. On February 9, 2012, the specially-designed splint was
placed in Kaiba at C.S. Mott Children’s Hospital. The splint was sewn
around Kaiba’s airway to expand the bronchus and give it a skeleton to
aid proper growth. Over about three years, the splint will be reabsorbed
by the body. The case is featured in the New England Journal of
Medicine. “It was amazing. As soon as the splint was put in, the lungs
started going up and down for the first time and we knew he was going to
be OK,” says Green.
Green and Hollister were able to make the
custom-designed, custom-fabricated device using high-resolution imaging
and computer-aided design. The device was created directly from a CT
scan of Kaiba's trachea/bronchus, integrating an image-based computer
model with laser-based 3D printing to produce the splint. “Our vision at
the University of Michigan Health System is to create the future of
health care through discovery. This collaboration between faculty in our
Medical School and College of Engineering is an incredible
demonstration of how we achieve that vision, translating research into
treatments for our patients,” says Ora Hirsch Pescovitz, M.D., U-M
executive vice president for medical affairs and CEO of the U-M Health
System. “Groundbreaking discoveries that save lives of individuals
across the nation and world are happening right here in Ann Arbor. I
continue to be inspired and proud of the extraordinary people and the
amazing work happening across the Health System.” Kaiba was off
ventilator support 21 days after the procedure, and has not had
breathing trouble since then. “The material we used is a nice choice for
this. It takes about two to three years for the trachea to remodel and
grow into a healthy state, and that’s about how long this material will
take to dissolve into the body,” says Hollister. “Kaiba’s case is
definitely the highlight of my career so far. To actually build
something that a surgeon can use to save a person’s life? It’s a
tremendous feeling.” The image-based design and 3D biomaterial printing
process can be adapted to build and reconstruct a number of tissue
structures. Green and Hollister have already utilized the process to
build and test patient specific ear and nose structures in pre-clinical
models. In addition, the method has been used by Hollister with
collaborators to rebuild bone structures (spine, craniofacial and long
bone) in pre-clinical models. Severe tracheobronchomalacia is rare.
About 1 in 2,200 babies are born with tracheomalacia and most children
grow out of it by age 2 or 3, although it often is misdiagnosed as
asthma that doesn’t respond to treatment. Severe cases, like Kaiba’s,
are about 10 percent of that number. And they are frightening, says
Green. A normal cold can cause a baby to stop breathing. In Kaiba’s
case, the family was out at a restaurant when he was six weeks old and
he turned blue. “Severe tracheobronchomalacia has been a condition that
has bothered me for years,” says Green. “I’ve seen children die from it.
To see this device work, it’s a major accomplishment and offers hope
for these children.” Before the device was placed, Kaiba continued to
stop breathing on a regular basis and required resuscitation daily.
“Even with the best treatments available, he continued to have these
episodes. He was imminently going to die. The physician treating him in
Ohio knew there was no other option, other than our device in
development here,” Green says. Kaiba is doing well and he and his
family, including an older brother and sister, live in Ohio. “He has not
had another episode of turning blue,” says April. “We are so thankful
that something could be done for him. It means the world to us.”
The
University of Michigan is becoming a leader in pediatric device
manufacturing, as is the case with the manufacturing process device used
to save this baby's life. The FDA-funded University of Michigan
Pediatric Device Consortium (M-PED) offers product development guidance and strategic support for advancing devices to market. Journal reference: DOI: 10.1056/1 NEJMc1206319
Additional authors: Of the University of Michigan: David A. Zopf, M.D., Richard G. Ohye, M.D. Of Akron Children’s Hospital: Marc E. Nelson, M.D.