ICU Management & Practice, Volume 18 - Issue 2, 2018
This article reviews the RRT system concept and provides an update on the current state of such systems, their challenges, their performance, the evidence supporting their usefulness and their evolution.
You might also like:Developing new approaches to patient safety
Concept of failure to rescue
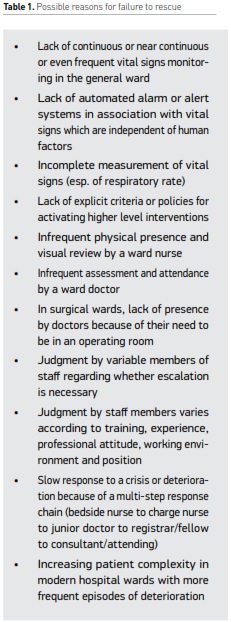
Epidemiology of serious adverse events
Warning signs
Responding to abnormal vital signs
Key principles
Rapid response team-based system
Triggering criteria
RRT composition

Interventions and outcomes
How to make a RRT work
Controversies
Conclusions
Conflict of interest
References:
Bagshaw SM, Mondor EE, Scouten C et al. (2010) A survey of nurses’ beliefs about the medical emergency team system in a Canadian tertiary hospital. Am J Crit Care, 19: 74-83.
Bell MB, Konrad D, Granath F et al. (2006) Prevalence and sensitivity of MET-criteria in a Scandinavian University Hospital. Resuscitation, 70: 66-73.
Bellomo R, Ackerman M, Bailey M et al. (2006) A controlled trial of electronic automated advisory vital signs monitoring in general hospital wards. Crit Care Med, 40: 2349-61.
Bellomo R, Goldsmith D, Uchino S et al. (2003) A prospective before-and-after trial of a medical emergency team. Med J Aust, 179: 283-7.
Brennan TA, Localio AR, Leape LL et al. (1990) Identification of adverse events occurring during hospitalization. A cross-sectional study of litigation, quality assurance, and medical records at two teaching hospitals. Ann Intern Med, 12: 221-6.
Brown C, Drosdowskyy A, Krishnasamy M (2017) An exploration of medical emergency team intervention at the end of life for people with advanced cancer. Eur J Oncol Nurs, 31: 77-83.
Buist M, Bellomo R (2004) MET: the emergency medical team or the medical education team? Crit Care Resus, 6: 88-91.
Buist M, Harrison J, Abaloz E et al. (2007) Six year audit of cardiac arrests and medical emergency team calls in an Australian outer metropolitan teaching hospital. BMJ, 335: 1210-2.
Buist, MD, Jarmolowski E, Burton PR et al. (1999) Recognising clinical instability in hospital patients before cardiac arrest or unplanned admission to intensive care. A pilot study in a tertiary-care hospital. Med J Aust, 171: 22-5.
Chan PS, Jain R, Brahmajee K et al. (2010) Rapid response teams: a systematic review and meta-analysis. Arch Intern Med, 170: 18-26.
Chen J, Bellomo R, Flabouris A et al; MERIT Study Investigators for the Simpson
Centre; ANZICS Clinical Trials Group (2009) The relationship between early emergency team calls and serious adverse events. Crit Care Med, 37: 148-53.
Chen J, Bellomo R, Flabouris A et al. (2015) Delayed emergency team calls and associated hospital mortality: a multicenter study. Crit Care Med, 43: 2059-65.
Chen J, Flabouris A, Bellomo R et al. (2008) The medical emergency team system and not-for resuscitation orders: results from the MERIT study. Resuscitation, 79: 391-7.
Chen J, Ou L, Hillman K (2014) The impact of implementing a rapid response system: A comparison of cardiopulmonary arrests and mortality among four teaching hospitals in Australia. Resuscitation, 85: 1275-81.
Cretikos M, Bellomo R, Hillman K et al. (2008) Respiratory rate: the neglected vital sign. MJA, 188: 657–9.
Cretikos M, Chen J, Hillman K et al.; MERIT study investigators. (2007) The objective medical emergency team activation criteria: a case-control study. Resuscitation, 73: 62-72.
Davis P, Lay-Yee R, Briant R et al. (2002) Adverse events in New Zealand public hospitals I: occurrence and impact. N Z Med J, 115: U271.
DeVita M (2005) Medical emergency teams: deciphering clues to crises in hospitals. Crit Care, 9: 325-6.
DeVita MA, Bellomo R, Hillman K et al. (2006) Findings of the first consensus conference on medical emergency teams. Crit Care Med, 34: 2463-78.
DeVita MA, Hillman K (2006) Potential sociological and political barriers to medical emergency team implementation. In: DeVita MA, Hillman K, Bellomo R, eds. Medical emergency teams. Implementation and outcome measurement. New York: Springer Science and Business Media, pp. 91-103.
England K, Bion JF (2008) Introduction of medical emergency teams in Australia and New Zealand: a multicentre study. Crit Care, 12: 151.
Foraida MI, DeVita MA, Braithwaite RS et al. (2003) Improving the utilization of medical crisis teams (Condition C) at an urban tertiary care hospital. J Crit Care, 18: 87-94.
Franklin C, Mathew J (1994) Developing strategies to prevent in hospital cardiac arrest: analyzing responses of physicians and nurses in the hours before the event. Crit Care Med, 22: 244-7.
Gao H, McDonnell A, Harrison DA et al. (2007) Systematic review and evaluation of physiological track and trigger warning systems for identifying at-risk patients on the ward. Intensive Care Med, 33: 667-9.
Hillman K, Chen J, Cretikos M et al; MERIT study investigators (2005) Introduction of the medical emergency team (MET) system: a cluster-randomised controlled trial. Lancet, 365: 2091-7.
Hodgetts TJ, Kenward G, Vlackonikolis I et al. (2002) Incidence, location and reasons for avoidable in-hospital cardiac arrest in a district general hospital. Resuscitation, 54: 115-23.
Jones D, Bates S, Warrillow S et al. (2005) Long term effect of a medical emergency team on cardiac arrests in a teaching hospital. Critical Care, 9: R808-15.
Jones D, Bellomo R, DeVita MA (2009) Effectiveness of the medical emergency team: the importance of dose. Crit Care, 13: 313.
Jones D, Duke G, Green J et al. (2006) Medical emergency team syndromes and an approach to their management. Critical Care, 10: R30.
Jones D, Opdam H, Egi M et al. (2007) Long-term effect of a medical emergency team on mortality in a teaching hospital. Resuscitation, 74: 235-41.
Jones DA, Baldwin I, McIntyre T et al. (2006) Nurses’ attitudes to a medical emergency team service in a teaching hospital. Qual Saf Health Care, 15: 427-32.
Leuvan CH, Mitchell I (2008) Missed opportunities? An observational study of vital sign measurements. Crit Care Resusc, 10: 111-5.
Loekito E, Bailey J, Bellomo R (2013) Common laboratory tests predict imminent death in ward patients. Resuscitation, 84: 280-5.
Ludikhuize J, Brunsveld-Reinders AH, Dijkgraaf MG et al. (2015) Outcomes associated with the nationwide introduction of rapid response systems in The Netherlands. Crit Care Med, 43(12): 2544-51.
Maharaj R, Raffaele I, Wendon J (2015) Rapid response systems: a systematic review and meta-analysis. Crit Care, 19: 254.
McGaughey J, Alderdice F, Fowler R et al. (2007) Outreach and early warning systems (EWS) for the prevention of intensive care admission and death of critically ill adult patients on general hospital wards. Cochrane Database Syst Rev, (3): CD005529.
McQuillan P, Pilkington S, Allan A et al. (1998) Confidential inquiry into quality of care before admission to intensive care. BMJ, 316: 1853-8.
Sebat F, Musthafa AA, Johnson D et al. (2007) Effect of a rapid response system for patients in shock on time to treatment and mortality during 5 years. Crit Care Med, 35: 2568-75.
Sharek PJ, Parast LM, Leong K et al. (2007) Effect of a rapid response team on hospital-wide mortality and code rates outside the ICU in a children's hospital. JAMA, 298(19): 2267-74.
Steel AC, Reynolds SF (2008) The growth of Rapid Response Systems. Jt Comm J Qual Patient Saf, 34: 489-95.
Subbe CP, Duller B, Bellomo R (2017) Effect of an automated notification system for deteriorating ward patients on clinical outcomes. Crit Care, 21: 52.
Tan LH, Delaney A (2014) Medical emergency teams and end of life care: a systematic review. Crit Care Resusc, 16: 62-8.
Thomas EJ, Studdert DM, Burstin HR et al. (2000) Incidence and types of adverse events and negligent care in Utah and Colorado. Med Care, 38: 261-71.
Wilson RM, Harrison BT, Gibberd RW et al. (1999) An analysis of the causes of adverse events from the Quality in Australian Health Care Study. Med J Aust, 170: 411-5.
Wilson RM, Runciman WB, Gibberd RW et al. (1995) The Quality in Australian Health Care Study. Med J Aust, 163: 458-71.
Winters BD, Pham J, Pronovost PJ (2006) Rapid response teams – walk don’t run. JAMA, 296: 1645-7.







