The
number of confirmed cases of the novel coronavirus (2019-nCoV) continues to
grow. WHO and other institutions and organisations around the world are trying to implement measures to contain the outbreak and find treatments.
You may also like: WHO Declares Public Health Emergency
WHO is convening a global research and innovation forum, to be held 11–12 February in Geneva, Switzerland. The forum, organised in collaboration with the Global Research Collaboration for Infectious Disease Preparedness, will bring together leading scientists, public health agencies, ministries of health and research funders to discuss several areas of research, including identifying the source of the virus as well as sharing of biological samples and genetic sequences, and to produce a global research agenda for the new coronavirus.
WHO is also strengthening its support to African countries, which due to their direct links or high volume of travel to China need to be particularly vigilant for the novel coronavirus. Africa has 1.2 billion people and only six laboratories that can test for coronavirus. The organisation would help in the detection and management of suspect cases, should a first case be detected. WHO is dispatching specialised staff and protective equipment for health workers, as well as thermometers and other essential supplies for screening and handling suspect cases at airports and other points of entry.
Additional coverage of African nations’
responses to the novel coronavirus is available from The
Guardian and Reuters.
Financial Support
Simultaneously, the organisation is facilitating the launch a €614 million ($675 million) preparedness and response plan by the international community, for February – April 2020. The plan lays out activities and resources needed by international health organisations globally to implement priority public health measures. It aims to:
- Limit human-to-human transmission of the virus, particularly in the most vulnerable countries.
- Identify, isolate and care for patients early.
- Communicate critical risk and event information.
- Minimise social and economic impact.
- Reduce virus spread from animal sources.
- Address crucial unknowns.
In the meantime, the Bill and Melinda Gates
Foundation is spending €91 million ($100 million) to fight the coronavirus
outbreak (including the previously pledged $10 million). ‘The funding will help
strengthen detection, isolation, and treatment efforts; protect at-risk
populations; and develop vaccines, treatments, and diagnostics,’ the foundation
said in a press
release.
Latest Figures
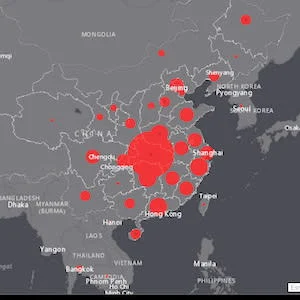
According to the situation report from WHO as of 6 February, there were 28,276 confirmed cases globally, of which 28,060 in China, where 564 people died. Outside of China there were 216 confirmed cases in 24 countries, with one death. However, according to the China National Health Commission (7 February), the virus has killed 638 people and infected 31,161 in mainland China. It has spread overseas with confirmed infections in at least 25 nations and two deaths, one in Hong Kong and one in the Philippines.
You may also like:Wuhan City Coronavirus: Health Tech To The Rescue
WHO assesses that the outbreak poses a very high risk in China, and high risk regionally and globally. The risk assessment was based on factors including the likelihood of further spread, the potential impact on human health, and the varying levels of effectiveness in national preparedness and response measures. The latest WHO situation reports can be found here.
The authors of a modelling research, published
in The Lancet, caution
that the true size of the epidemic and its pandemic potential remains unclear.
They estimate that up to 75,800 individuals in the Chinese city of Wuhan may
have been infected with 2019-nCoV as of January 25, 2020.
Infectious disease researchers at The
University of Texas at Austin and other institutions have concluded
there is a high probability that the deadly Wuhan coronavirus spread beyond
Wuhan and other quarantined cities before Chinese officials were able to put a
quarantine in place. The team estimated new cases of the virus doubled roughly
every week, and on average, that every infected person transmitted the disease
to approximately two other people.
Laboratory testing by three institutions in Germany
has revealed
that the coronavirus can be isolated from nose and throat swabs even where
these have been obtained from patients with mild symptoms. The research groups conclude
that even persons with mild symptoms are capable of transmitting the virus.
Current Research
According to WHO, currently there are 15 laboratories identified to provide reference testing support for 2019-nCoV.
A number of drug manufacturers are working on
vaccines or antivirals and other treatments for 2019-nCoV. Investment costs could
reach €727 million ($800 million) and the process, even if accelerated, could take
at least 12
to 18 months until approval.
None of the coronaviruses known to infect
humans has an approved treatment, according to the
U.S. Centers for Disease Control and Prevention (CDC). However, a handful
of repurposed drugs, from drugs targeting Ebola to HIV, have already shown
promise.
On 6 February, China began
enrolling patients in a clinical trial of remdesivir, an antiviral medicine
made by Gilead, the American pharmaceutical company. The drug is experimental
and not yet approved for any use, but the first coronavirus patient in the U.S.
was treated with it, and his symptoms improved the next day, according to a report in The
New England Journal of Medicine.
Another report by scientists in China showed that remdesivir blocked the
new coronavirus from infecting cells grown in the lab. Another reported finding
was that chloroquine, a cheap drug used for decades to treat malaria, could
also fight the new coronavirus.
During similar coronavirus (SARS) outbreak in
2003, another class of drugs (protease inhibitors) approved to treat HIV proved
also be effective in some cases. Two of the drugs are now being tested for
their ability to treat 2019-nCov in a clinical trial in China, as reported
in JAMA. The JAMA Network Coronavirus Resource Center
has also streamed a
discussion of latest developments in the 2019-nCoV outbreak between immunologist
Anthony Fauci (the National Institute of Allergy and Infectious Diseases) and JAMA Editor Howard Bauchner.
Steroids, often used by doctors to reduce
inflammation, should be avoided in the treatment of the current novel
coronavirus, experts advise
in a commentary article published in The Lancet.
The U.S. Department of Health and
Human Services and Regeneron Pharmaceuticals are collaborating
to identify antibodies that will stop the coronavirus from entering cells,
while researchers at the U.S. National Institutes of Health are developing
a vaccine with a clinical trial to be launched within the next three months.
In a special report in Radiology, researchers describe CT imaging features that aid in the early detection and diagnosis of Wuhan coronavirus.
A team of Lawrence Livermore National
Laboratory (LLNL) researchers has
developed a preliminary set of predictive three-dimensional protein
structures of the virus to aid research efforts to combat the disease. The most
current 3D models can be obtained by contacting the LLNL Biosecurity
Center.
Regenstrief
Institute is providing
help to track cases of the illness. The team is creating a series of codes to
identify the lab tests used to screen patients for the virus.
Insilico Medicine announced that it would publish
structures of small molecules targeting the key protein 2019-nCoV 3C-like
protease. The generated molecules are published here and will be continuously
updated.
Sources: LiveScience,
Reuters,
New
York Times, EurekAlert, BBC
Image credit: Johns Hopkins Center for Systems Science and Engineering