AVAflex Vertebral Balloon System Enables Targeted Balloon Placement
CareFusion, a leading global medical technology company, has announced it recently received 510(k) clearance from the US Food and Drug Administration (FDA) for its new AVAflex Vertebral Balloon System.
The new system is the latest innovation in CareFusion’s AVAmax Advanced Vertebral Augmentation portfolio. The focus of these products is to provide a minimally invasive solution to treat vertebral compression fractures, while also promoting safety for the patient, physician and staff.
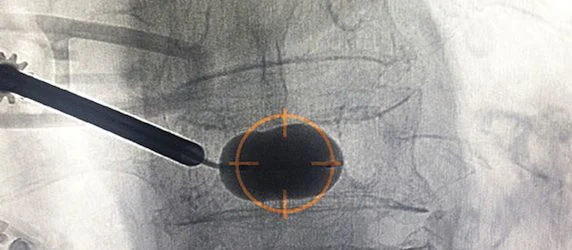
The AVAflex Vertebral Balloon System combines the unique features of the AVAflex Curved Vertebral Augmentation Needle and the AVAmax Vertebral Balloon to enable targeted balloon placement across the midline of the vertebral body, followed by targeted cement placement for optimal fill through a single pedicle.
The AVAflex Vertebral Balloon System is in limited commercial release in multiple hospitals across the US.
“The ability to steer a balloon through a unipedicular approach cannot be overstated in its simplicity and efficiency,” said Dr. Michael Verdolin of Verdolin Pain Specialists in Chula Vista, California, after using the product. “The procedure was faster, with less patient discomfort.”
Dr. Allan Brook, an interventional radiologist with Montefiore Medical Center in Bronx, N.Y. and the first physician to use the new device on the East Coast, said “The unipedicular approach offers a less risky approach to the midline of a vertebral body. Having another tool that places the cavity in the desired location can improve the effectiveness of vertebral augmentation in less time and with less radiation.”
Another physician using the device in limited commercial release, Dr. Langham Gleason, a neurosurgeon with the South Texas Brain and Spine Center in Corpus Christi, Texas, said “The AVAflex Vertebral Balloon System works extremely well to allow bilateral fracture reduction using a unilateral approach. I suspect that placement of bilateral vertebral cannulas will largely fade away in the future."
CareFusion expects a full commercial launch of the AVAflex Vertebral Balloon System in Spring 2014.
Source: CareFusion
3 February 2014
Latest Articles
CareFusion, Fractures, Surgery, Spine, Neurosurgery, vertebral balloon system, vertebral body
AVAflex Vertebral Balloon System Enables Targeted Balloon Placement CareFusion, a leading global medical technology company, has announced it recently r...