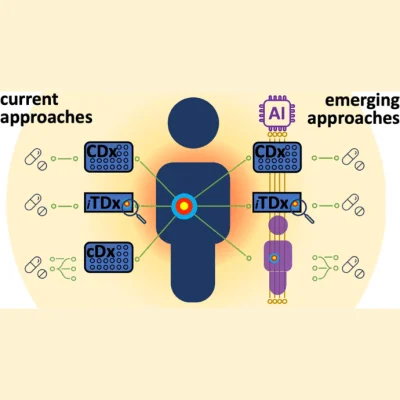
Precision cancer therapies represent a promising frontier in the fight against cancer, offering tailored treatments based on individual patient characteristics. However, a bottleneck in regulatory approval processes is hindering the translation of these advances into patient care. This delay has raised concerns among patients who perceive regulators as unnecessarily impeding access to life-saving therapies.
Regulatory frameworks need to evolve on pace with technological advancements
The current regulatory frameworks, designed primarily for traditional therapies, are struggling to adapt to the unique challenges posed by emerging precision cancer treatments. In particular, therapies based on advanced therapy medicinal products (ATMPs), which utilise genes, tissues, or cells, face lengthy approval timelines in both the US and the EU. This situation is compounded by the rapid technological advancements driving the development and use of cell and drug-based therapies.
AI-guided precision medicine needs specific regulatory frameworks
Researchers are exploring the integration of artificial intelligence (AI) into treatment personalisation, encompassing diagnosis, treatment planning, and dose adaptation. AI-driven tools show promise in enhancing diagnosis accuracy and treatment effectiveness, particularly in areas such as radiological image analysis and patient-reported outcomes monitoring.
Regulatory bodies are grappling with how to effectively evaluate and approve the potential of AI-guided precision medicine. In both the US and the EU, discussions are underway to develop regulatory strategies that can keep pace with technological advancements while ensuring patient safety and efficacy. Key areas of focus include clinical decision support systems for healthcare providers, precision diagnostics (companion and complementary diagnostics), drug companion apps, and personalised ATMPs. However, each of these areas presents specific regulatory challenges, from classification issues to concerns regarding transparency and explainability of AI-driven algorithms.
Streamlined and agile regulatory bodies are key to activate transformational changes
Moreover, the emergence of digital twins as a concept in diagnosis and therapy adds another layer of complexity to regulatory considerations. These real-time representations of patient data have the potential to revolutionise personalised care but require flexible regulatory frameworks to accommodate their dynamic nature. Addressing these regulatory gaps necessitates transformative changes in current approaches, including enhanced collaboration between regulatory bodies, streamlined approval processes, and proactive adoption of AI-specific regulations. Ethical implications also loom large, particularly concerning informed consent, physician-patient relationships, and the potential for AI-driven biases. Regulatory frameworks must ensure that patient-centric care remains paramount, with transparency and shared decision-making guiding the integration of AI into clinical practice.
However, achieving such changes requires overcoming institutional barriers and navigating divergent international regulatory landscapes. While precision cancer therapies hold immense promise for improving patient outcomes, regulatory bottlenecks threaten to delay their widespread adoption. Addressing these challenges requires a concerted effort to develop agile regulatory frameworks that can accommodate the rapid pace of technological innovation while upholding patient safety and ethical standards.
Source & Image Credit: CHEST