Q&A with Dr. Ellen T. Roche
A customisable “soft robot” has been developed by Harvard University and Boston’s Children Hospital that could open new treatment methods for failing hearts. Unlike currently available devices such as ventricular assist devices (VADs), the soft robot has no direct contact with blood. The sleeve wraps around the heart and is filled with pressurised air which then twists and compresses in synch with the beating heart.
According to Ellen T. Roche, PhD, former PhD student at the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS) and currently postdoctoral researcher with Professor Peter McHugh in biomedical engineering at NUI Galway, who is the first author of the paper published in Science Translational Medicine, (Roche et al. 2017), this groundbreaking soft robot can “potentially reduce the burden of heart disease and improve the quality of life for patients”. This interesting development in devices can potentially decrease the risk of clotting and the use of dangerous blood thinners, which will overall improve cardiac rehabilitation and recovery.
The device has so far been tested on animal models and requires more research before being implanted in humans. Nevertheless, the research is a first step towards improving the augmentation of organ function and it shows a strong promise to patients with failing hearts.
Dr. Ellen T. Roche spoke to HealthManagement.org to provide more insight into this breakthrough in heart failure treatment options.
How is your technology different than other external cardiac assist devices?
This device is an entirely new design and approach based on McKibben pneumatic actuators, integrated into a soft robotics design. Composed of soft materials that have similar properties to the native heart muscle, the device is non blood-contacting and its motion can be fine-tuned for optimal therapeutic effect. One novel and important characteristic of this device is that the system is able to augment systolic and diastolic function of the failing myocardium.
How could this device help alleviate the burden on the healthcare system?
Current devices are dependent on direct exposure to the blood, which could potentially lead to complications such as bleeding and stroke. Because no direct blood/device interface exists, it is hoped that related complications could be reduced. In addition, this technology offers an alternative type of circulatory support to heart failure patients, and does not require them to simultaneously take blood thinners.
Why is it important that the device does not come into direct contact with blood?
Neurologic complications resulting from blood clots and/or anticoagulation are a feared complication of current devices. This technology reduces the risk of clotting and associated adverse complications (for example stroke), and enables patients to avoid the use of blood thinner medication, which can require constant monitoring or cause bleeding problems.
What follow up work is needed to get this device into humans?
These findings represent an exciting proof-of-concept result, demonstrating significant improvements in cardiac function. Further work will be required to refine the technology and make it suitable for longer-term implantation in the body. Some technical hurdles remain, including developing the optimal means by which the device is attached to the heart. In addition, long term animal studies are needed to assess device durability and chronic complications.
For what other conditions, besides cardiac, might the device be used for in the near future?
Soft robotic devices are ideally suited for interacting with soft tissue. As such, there are many other applications for devices inside and outside the body where small to moderate levels of assistance can potentially help with healing and recovery.

Models shows different types of actuation in the soft robotic sleeve and how much volume they can pump.
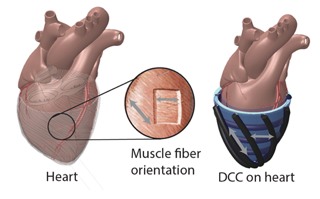
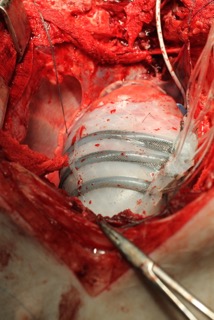
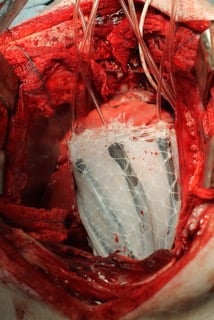
Image credits: Ellen Roche/ Harvard University
References:
Roche ET, Horvath MA, Wamala I, Alazmani A, Song SE, Whyte W, Machaidze Z, Payne CJ, Weaver JC, Fishbein G, Kuebler J, Vasilyev NV, Mooney DJ, Pigula FA, Walsh CJ (2017) Soft robotic sleeve supports heart function. Sci Transl Med, 9(373). pii: eaaf3925. doi: 10.1126/scitranslmed.aaf3925.






















