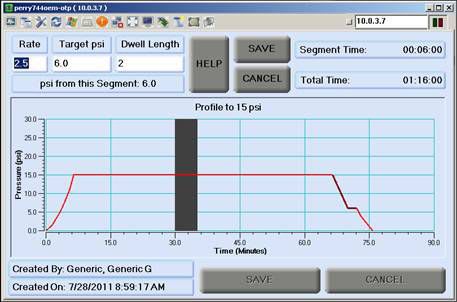
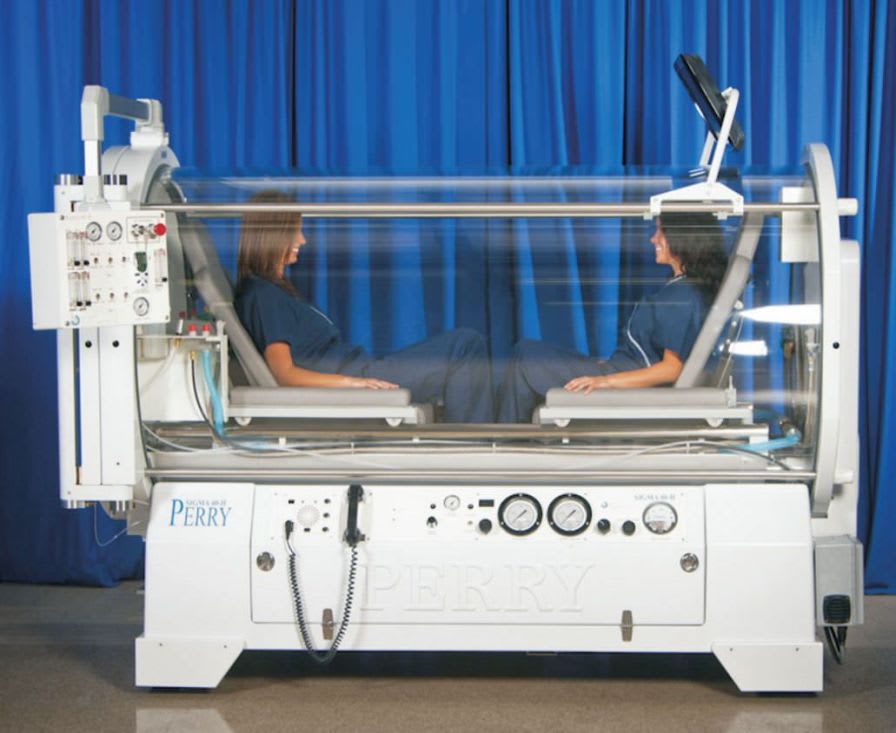
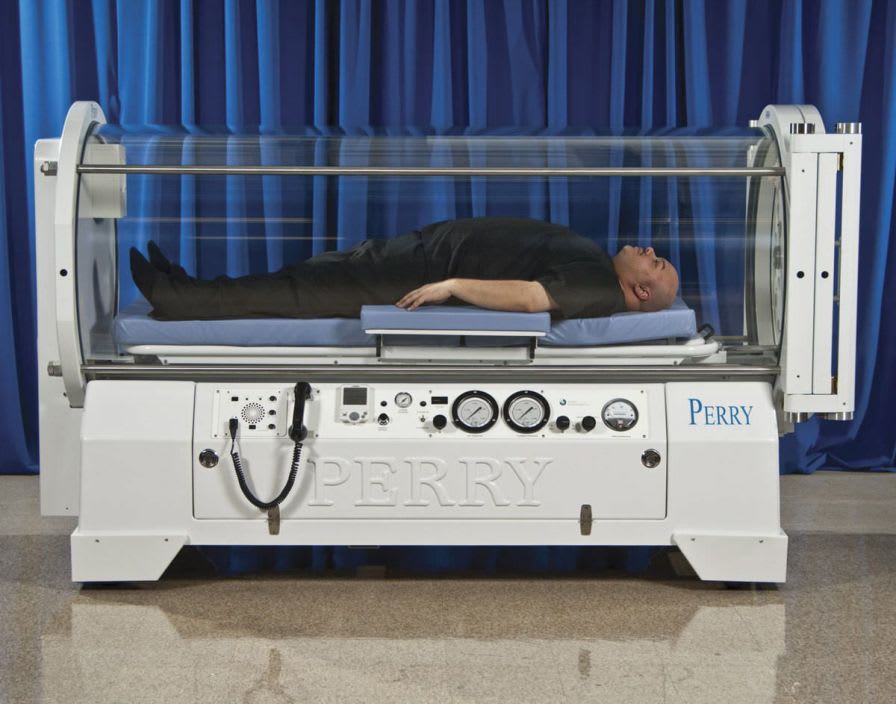
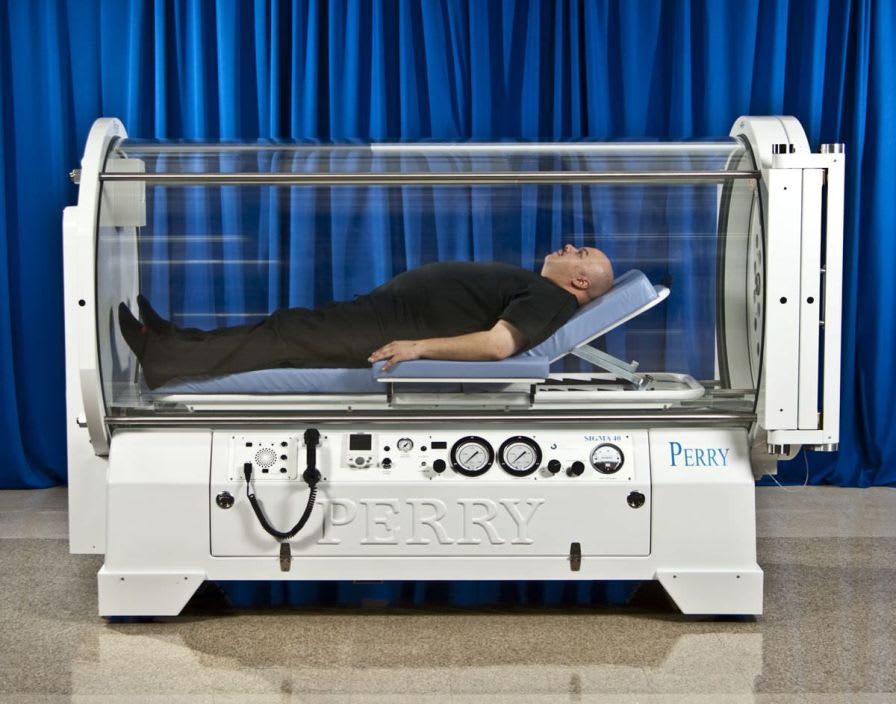
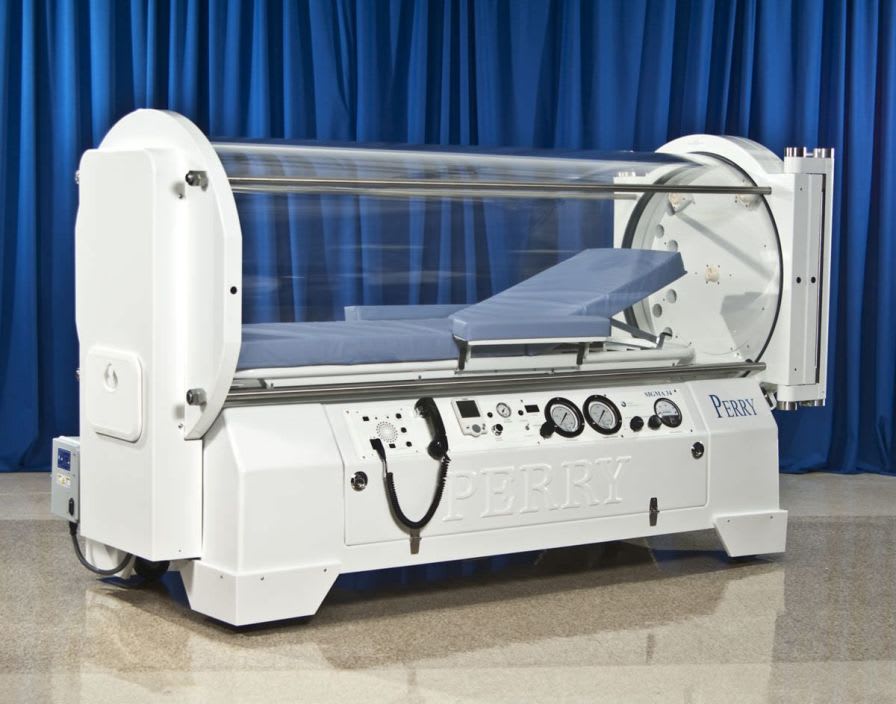
Perry Baromedical is a registered medical device manufacturer. Perry Baromedical is an ISO 13485:2003 certified world leader in the manufacture, installation and service ofhyperbaric oxygen therapy systems. Hyperbaric Oxygen Chambers are United States Food and Drug Administration (FDA) Class II devices and are controlled by federal law. All systems are designed, manufactured, tested and installed in accordance with the current regulations of the FDA, The American Society of Mechanical Engineers (ASME/PVHO-2) Codes, The Pressure Equipment Directive (CE Mark), Medical Device Directive (CE Mark) and the requirements of the National Fire Protection Agency (NFPA).
Products
3750 Prospect Ave,
Riviera Beach, FL
33404
× × × × × ×