ABOUT EXTHERA
ExThera Medical Corporation is developing revolutionary medical devices that address unmet clinical needs for the treatment of bloodstream infections and pathogen-reduction.
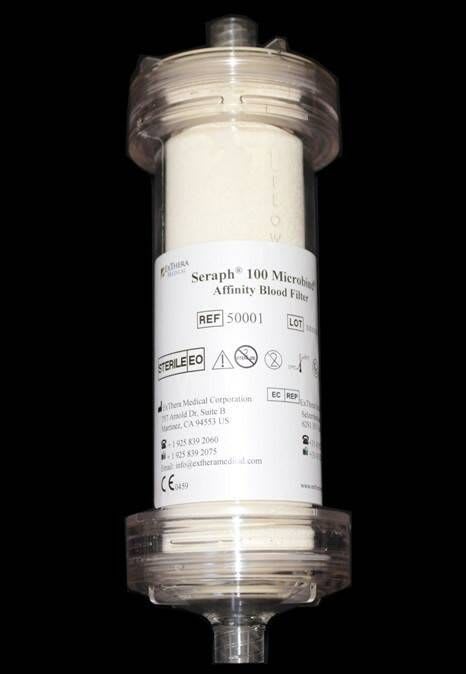
Based in Martinez, California with an office in the Netherlands, ExThera Medical Corporation is a privately held medical device company developing innovative, single-use blood filters capable of capturing a broad range of bacteria, viruses, parasites, toxins and other sepsis mediators.
ExThera was formed in 2007 through a joint venture between the Emergence Venture Partners Life Science Incubator and ExThera AB, at the Karolinska Institute of Stockholm. The company’s first product, the Seraph® 100 Microbind® Affinity Blood Filter, is CE Marked and commercially available in Europe.
The company is focused is on therapeutic applications to treat drug-resistant and drug-susceptible bloodstream infections in high-risk patient populations, with field deployable versions for biodefense and military use. ExThera also intends to develop, oncology, cardiovascular and diagnostic applications of its technology.
WHAT WE’RE ADDRESSING
Bloodstream infections are a global health issue and are increasing in incidence despite significant efforts to find effective treatments. If not identified and treated early, these infections can cause serious illness by progressing to sepsis and septic shock with organ damage or organ failure and death.