Our products and systems are sold in over 120 countries worldwide. We sell our products through direct sales offices operations in most of our major markets, and a network of distributors that sell to hospitals, homecare providers and other manufacturers of medical devices.
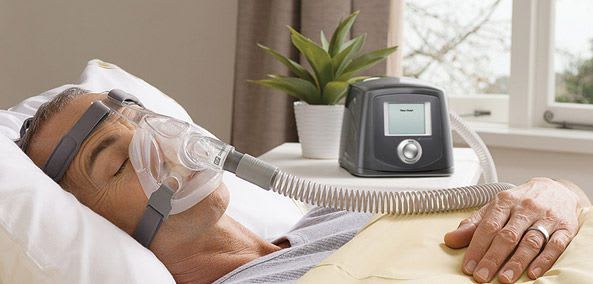
Fisher & Paykel entered the respiratory care market in 1971 with the development of a unique respiratory humidifier system for use in critical care. We now offer a broad range of products and systems for use in respiratory and acute care and in the treatment of obstructive sleep apnea (OSA).
Our fact sheet provides more detail on our company’s financial performance.
Strategy
Research & Development
International Reach
The growing number of employee(s) in these markets helps us to ensure that our product range is well supported and new products and applications for our products are quickly brought to the attention of health professionals and our customers. Approximately 99% of our sales revenue is derived from outside New Zealand.
Quality & Regulatory
With the healthcare device industry regulated worldwide, the ability to meet stringent standards is vital to ensuring market acceptance of our products. We assist our compliance with these standards by operating a quality management system certified to a range of international standards which apply to both our manufacturing facilities and our sales network.
Manufacturing & Operations
We manufacture, assemble and test our complete range of products, including many components, in our facilities in New Zealand and Mexico. Our facilities incorporate controlled working environments and our manufacturing and design processes are certified to meet the international medical device quality standard ISO13485.
Business Areas
We have two major product groups, Respiratory & Acute Care and Obstructive Sleep Apnea.
Respiratory & Acute Care
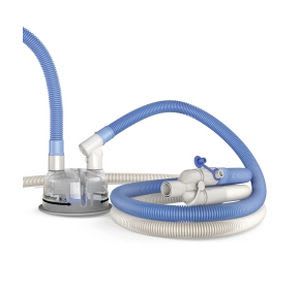
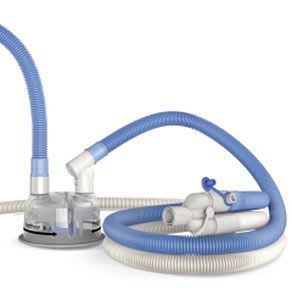
We offer a broad range of heated humidification products and systems for use during the treatment of respiratory conditions by ventilation or oxygen therapy, which form part of a comprehensive family of solutions for all therapies in the F&P Respiratory Care Continuum.
Incorporating patented and other proprietary technologies, our respiratory devices are designed to overcome many of the challenges of effectively creating, controlling and delivering gases to a patient's airway at close to physiologically normal levels of temperature and humidity.
Our therapy solutions incorporate technologies designed to benefit both the clinician and the patient, translating to efficient delivery of care and improved patient outcomes.
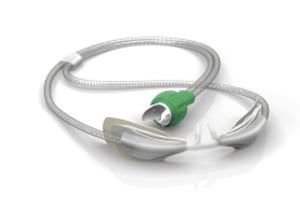
Our products include respiratory humidifiers, single-use and reusable chambers and breathing circuits, infant resuscitators, infant warmers and accessories. We also offer humidification systems for use in surgical procedures to condition dry carbon dioxide gas to normal physiological levels of temperature and humidity.