Endologix, Inc. develops and manufactures minimally invasive treatments for aortic disorders. The Company's focus is endovascular stent grafts for the treatment of abdominal aortic aneurysms (AAA). AAA is a weakening of the wall of the aorta, the largest artery in the body, resulting in a balloon-like enlargement. Once AAA develops, it continues to enlarge and, if left untreated, becomes increasingly susceptible to rupture. The overall patient mortality rate for ruptured AAA is approximately 75%, making it a leading cause of death in the U.S. Approximately 1.7 million Americans have AAA, and the incidence of the condition increases with age.
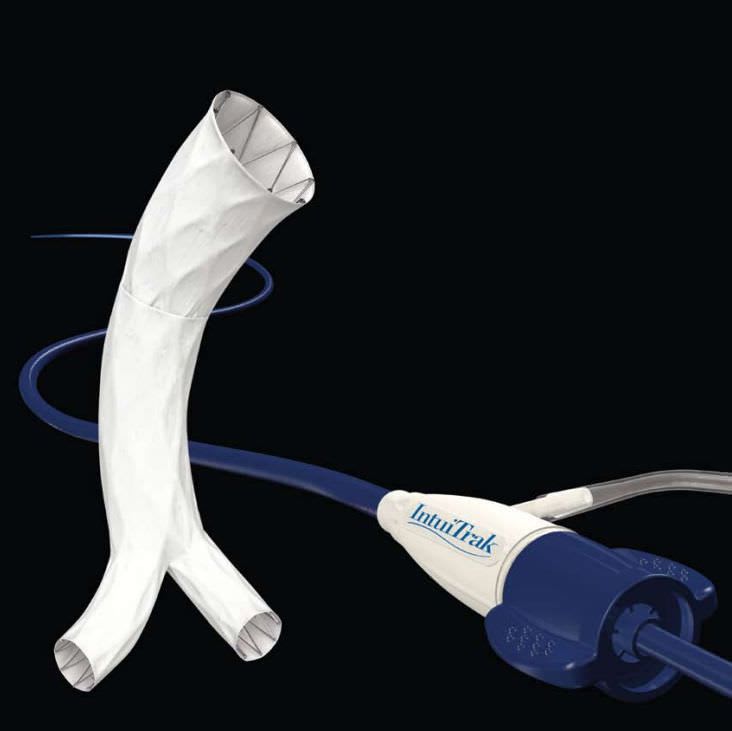
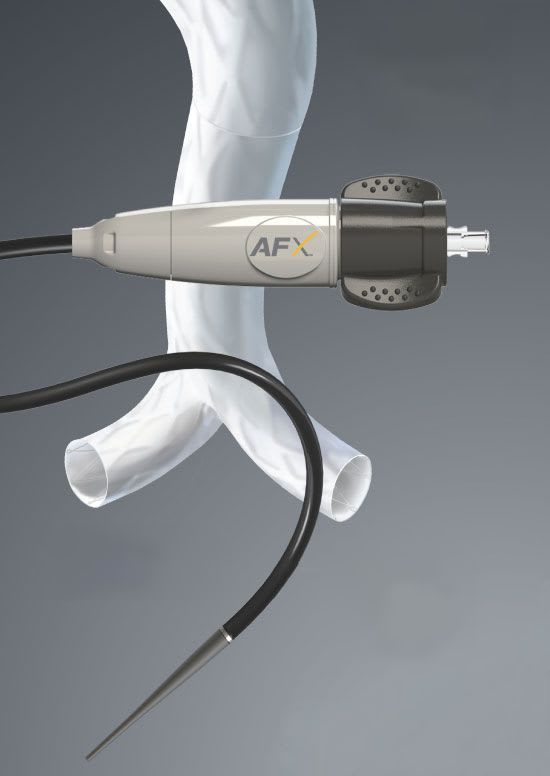
Endologix is currently marketing the AFX® Endovascular AAA System is available in the U.S., Europe, Latin America and other regions. The Nellix® EndoVascular Aneurysm Sealing System, a novel aneurysm sealing system, is approved for sale in Europe and other markets and for investigational use in the U.S. The Powerlink with IntuiTrak® Delivery System is available in Japan.