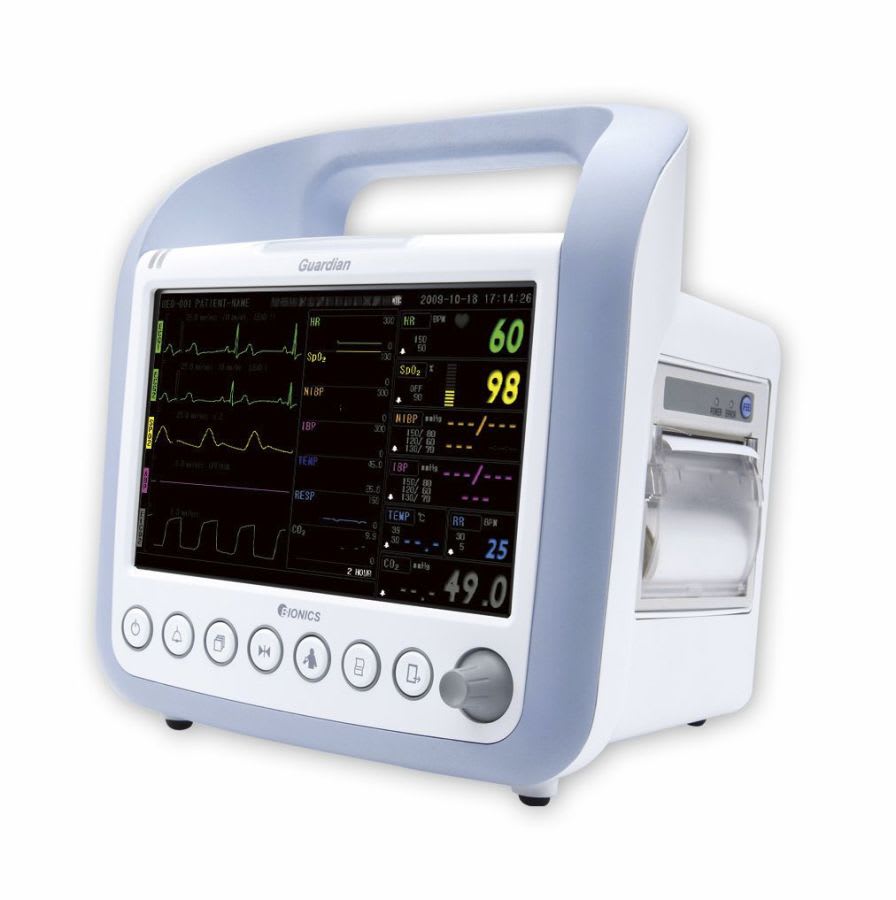
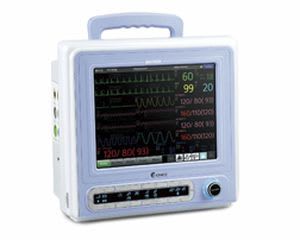
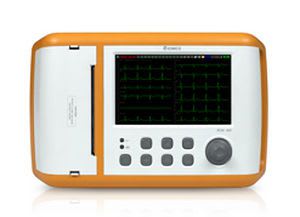
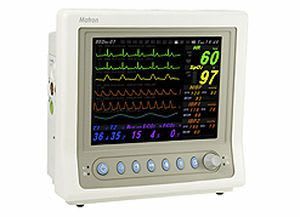
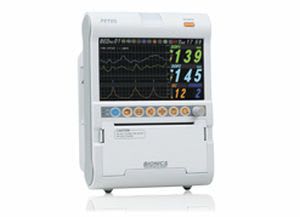
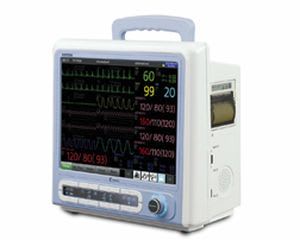
Bionics was established in Feb. 27, 2004 based on know-how of patient and fetal monitors. With the confidence and high quality of producing, we already have manufactured over 10 kinds of medical devices. We have achieved all sorts of certificates and standards. Our main export regions are Europe, Asia and Middle East.
In present, as International Quality System, we have achieved MDD and ISO13485. We have also achieved CE mark for Europe markets and will apply for FDA for North America and SFDA for China.