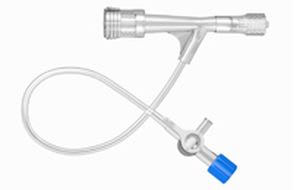
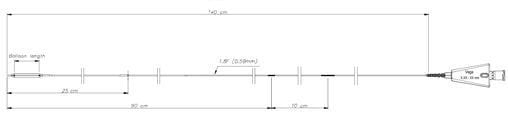
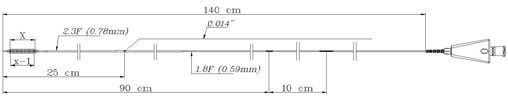
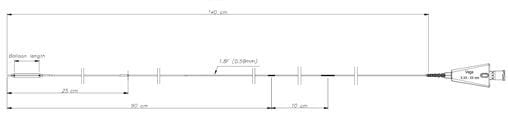
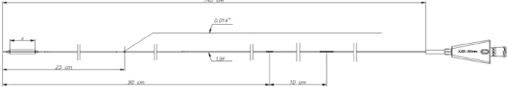
Founded in 1998 and based close to Paris, France, ARTHESYS has become the leader in OEM manufacturing for cardiovascular and endovascular catheter based state of the art products.
The company is ISO 13485 certified with LNE-Gmed, and is GMP compliant; its facilities are FDA registered. ARTHESYS portfolio covers catheters for plain dilatation, as well as for stent and/or drug delivery for all sorts of applications: cardiovascular, peripheral, renal, carotid, neurology.
Catheter platforms are either balloon based, or self-expandable. ARTHESYS has developped a unique CE-marked stent (available for OEM/OBL) with unique crimping technologies (ActivGrip Process) and expertise. Various accessories have been uniquely designed for easy access for intervention. Extrusion, packaging and sterilization are also part of the company’s portfolio of services.