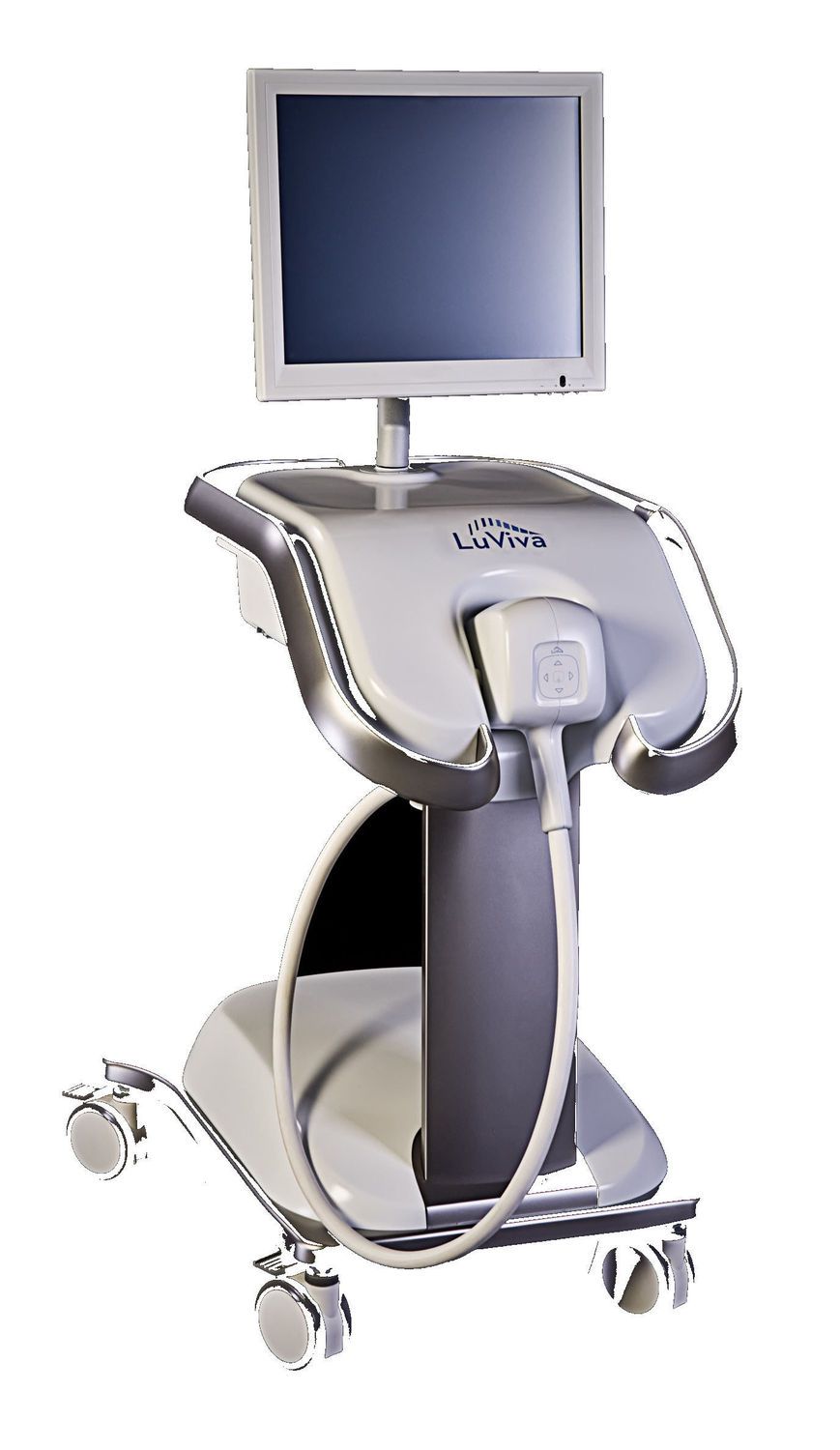
LuViva™ Advanced Cervical Scan, under development by Guided Therapeutics, is designed as a new non-invasive test that has the potential to significantly improve the early detection of cervical precancers. LuViva™ is designed as a fast, painless test that - unlike Pap smears and HPV testing - does not require a tissue sample or the delay of laboratory analysis. Early detection of cancer and precancers greatly improves survival, and LuViva™ is designed to help preserve the reproductive health of women. LuViva™ uses proprietary technology to identify cancers and precancers quickly by analyzing light reflected from the cervix. LuViva™ creates an image of the cervix for the doctor that highlights the location and severity of disease. The technology distinguishes between normal and diseased tissue by detecting biochemical and morphological changes at the cellular level. More than 1,600 women at risk for cervical disease were tested with the LuViva™ in a multi-center pivotal clinical trial. Results of the trial showed that: LuViva™ detected cervical disease up to two years earlier than Pap test, HPV test, colposcopy and biopsy. LuViva™ detected 86.3% cervical disease cases that had been missed by Pap, HPV (human papillomavirus) tests and biopsy. LuViva™ would have reduced the number of unnecessary biopsies by about 40 percent.
- Technology:biophotonic
- Cancer type:cervical