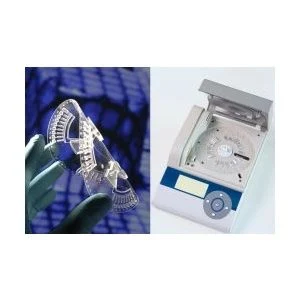
A new ‘lab-on-a-disc’ technology developed by an EU project research team can diagnose malaria and other febrile infectious diseases simultaneously in just an hour – allowing faster point-of-care treatment and precise drugs administration that could save thousands of lives.
A major problem with current testing for infectious diseases in Africa is that it focuses on individual diseases and cannot reliably discriminate between them. Since most infectious diseases have the same feverish symptoms, diagnosis is often inaccurate, resulting in thousands of deaths and increased resistance to antimicrobial drugs.
Clinical surveys show that up to 30 percent of patients are treated for malaria without even being infected by it. Considering the approximately 200 million malaria cases worldwide, it is vital that accurate diagnostic tools are developed to distinguish between infectious diseases such as malaria, typhoid, dengue fever and pneumonia, so the right therapies are applied.
The EU’s EUR 2.9 million DISCOGNOSIS project has come up with a new diagnostic tool: an easily-portable lab-on-a-disc, which can test for several tropical diseases at the same time, discriminate between them and guide healthcare personnel to proper patient treatment.
“It is a very simple and cheap system that can be used in regions with low medical infrastructure,” explained the project’s coordinator Dr. Konstantinos Mitsakakis, of the Department of Microsystems Engineering (IMTEK) at Freiburg University in Germany. “Results can be obtained from a finger prick of blood in just one hour, whereas lab culture currently takes up to three days.”
Identifying the Disease at the Molecular Level
The doctor or nurse injects the patient’s blood sample onto a plastic disc, the ‘LabDisk’, which is roughly the size of a CD, and then places the disc in the ‘disc player’. The device weighs just 2 kg, making it perfect for transportation to remote villages. The disc has pre-stored biochemical components, which allow fully automated analysis.
The blood sample is processed on disc and centrifugally distributed into microfluidic chambers where the disease pathogens can be identified from their DNA/RNA – whether it be from parasites (malaria), bacteria (typhoid or pneumonia) or viruses (dengue).
This generic point-of-care platform can be applied to many other infectious diseases for example Ebola, only by changing its bio-components. Early diagnosis can help limit the effects of an extended epidemic.
The researchers will validate the diagnostic device in the field, before the three-year project ends in October 2015, and have chosen two locations: the Pasteur Institute in Dakar, Senegal (with ‘bio-banked’ samples); and the Medical Center in Bunia, Democratic Republic of Congo (with recruitment and testing of around 100 patients).
Commercialising the Technology Worldwide
The economics of the LabDisk are very promising. Costs are currently estimated to be up to $10 per disc per patient, assuming some millions discs are manufactured, which is cheaper than a complete set of multiple infectious disease testing procedures currently in use in Africa.
The DISCOGNOSIS team is now seeking to increase the number of patients that can be tested simultaneously. Not only will this be more cost-effective, but it would prove a vital help in handling future epidemics.
Other follow-up activities include performing clinical trials and developing remote connection of the LabDisk player to a central database. “This could mean very important progress, not just for patient management, but also for epidemiological mapping of regions and countries, as we will be able to monitor the frequency and distribution of various infectious diseases,” Dr. Mitsakakis points out.
Source: European Commission, CORDIS
Image Credit: DiscoGnosis