Moderna has begun testing its COVID-19 vaccine in children ages 6 months to less than 12 years. This is the first such study with paediatric participants.
You might also like: Moderna and IBM will work together on implementation of technologies such as artificial intelligence, blockchain and hybrid cloud, to improve COVID-19 vaccine management in the U.S. Learn more
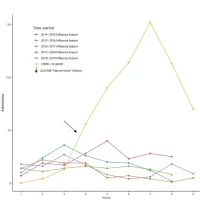
The Phase 2/3 study called KidCOVE comprises two parts.
1. Part 1: open label, dose-escalation, age de-escalation study.- 2 yo – up to 12 yo: each participant may receive either 50 μg or 100 μg dose of the vaccine.
- 6 mo – up to 2 yo: each participant may receive either 25 μg, 50 μg, or 100 μg dose.
The participants will receive two doses of the vaccine 28 days apart. To evaluate the medicine’s safety, tolerability, reactogenicity and effectiveness, the company will observe the participants for 12 months after the second jab. It plans to enrol about 6,750 paediatric participants within the stated age range, in the U.S. and Canada.
According to Stéphane Bancel, Moderna’s Chief Executive Officer, the company is “encouraged by the primary analysis of the Phase 3 COVE study of mRNA-1273 in adults ages 18 and above” and now is looking to “assess the potential safety and immunogenicity of our COVID-19 vaccine candidate in this important younger age population”.
The ClinicalTrials.gov identifier for this study is NCT04796896.
This is the first COVID-19 vaccine trial in paediatric population. In December 2020, Moderna started a trial in teenagers of 12-17 years of age, while Pfizer/BioNTech are running studies in 12-15-year-old children and pregnant women.
Source and image credit: Moderna


![Tuberculosis Diagnostics: The Promise of [18F]FDT PET Imaging Tuberculosis Diagnostics: The Promise of [18F]FDT PET Imaging](https://res.cloudinary.com/healthmanagement-org/image/upload/c_thumb,f_auto,fl_lossy,h_184,q_90,w_500/v1721132076/cw/00127782_cw_image_wi_88cc5f34b1423cec414436d2748b40ce.webp)